Abstract
Background
Bone health is remarkably affected by endocrine side effects due to paediatric cancer treatments and the disease itself. We aimed to provide novel insights into the contribution of independent predictors of bone health in young paediatric cancer survivors.
Methods
This cross-sectional multicentre study was carried out within the iBoneFIT framework in which 116 young paediatric cancer survivors (12.1 ± 3.3 years old; 43% female) were recruited. The independent predictors were sex, years from peak height velocity (PHV), time from treatment completion, radiotherapy exposure, region-specific lean and fat mass, musculoskeletal fitness, moderate-vigorous physical activity and past bone-specific physical activity.
Results
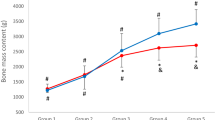
Region-specific lean mass was the strongest significant predictor of most areal bone mineral density (aBMD), all hip geometry parameters and Trabecular Bone Score (β = 0.400–0.775, p ≤ 0.05). Years from PHV was positively associated with total body less head, legs and arms aBMD, and time from treatment completion was also positively associated with total hip and femoral neck aBMD parameters and narrow neck cross-sectional area (β = 0.327–0.398, p ≤ 0.05; β = 0.135–0.221, p ≤ 0.05), respectively.
Conclusion
Region-specific lean mass was consistently the most important positive determinant of all bone parameters, except for total hip aBMD, all Hip Structural Analysis parameters and Trabecular Bone Score.
Impact
-
The findings of this study indicate that region-specific lean mass is consistently the most important positive determinant of bone health in young paediatric cancer survivors.
-
Randomised clinical trials focused on improving bone parameters of this population should target at region-specific lean mass due to the site-specific adaptations of the skeleton to external loading following paediatric cancer treatment.
-
After paediatric cancer diagnosis, years from peak height velocity (somatic maturity) is critical for bone development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are not publicly available due to the fact that the iBoneFIT project has not finished yet but are available from the corresponding author on reasonable request.
References
Miller, K. D. et al. Cancer statistics for adolescents and young adults, 2020. CA Cancer J. Clin. 70, 443–459 (2020).
Siegel, R. L. et al. Cancer statistics, 2023. CA Cancer J. Clin. 73, 17–48 (2023).
Wilson, C. L. & Ness, K. K. Bone mineral density deficits and fractures in survivors of childhood cancer. Curr. Osteoporos. Rep. 11, 329–337 (2013).
Marcucci, G. et al. Bone health in childhood cancer: review of the literature and recommendations for the management of bone health in childhood cancer survivors. Ann. Oncol. 30, 908–920 (2019).
Kelly, P.M. & Pottenger, E. Bone health issues in the pediatric oncology patient. Semin. Oncol. Nurs. 38, 151275. https://doi.org/10.1016/J.SONCN.2022.151275 (2022).
Weaver, C. M. et al. The National Osteoporosis Foundation’s position statement on peak bone mass development and lifestyle factors: a systematic review and implementation recommendations. Osteoporos. Int. 27, 1281–1386 (2016).
Gómez-Bruton, A., Matute-Llorente, Á., González-Agüero, A., Casajús, J. A. & Vicente-Rodríguez, G. Plyometric exercise and bone health in children and adolescents: a systematic review. World J. Pediatr. 13, 112–121 (2017).
Othman, F., Guo, C. Y., Webber, C., Atkinson, S. A. & Barr, R. D. Osteopenia in survivors of Wilms tumor. Int J. Oncol. 20, 827–833 (2002).
Chen, J. H., Liu, C., You, L. & Simmons, C. A. Boning up on Wolff’s Law: mechanical regulation of the cells that make and maintain bone. J. Biomech. 43, 108–118 (2010).
Jarfelt, M., Fors, H., Lannering, B. & Bjarnason, R. Bone mineral density and bone turnover in young adult survivors of childhood acute lymphoblastic leukaemia. Eur. J. Endocrinol. 154, 303–309 (2006).
Zymbal, V., Baptista, F., Letuchy, E. M., Janz, K. F. & Levy, S. M. Mediating effect of muscle on the relationship of physical activity and bone. Med. Sci. Sports Exerc. 51, 202–210 (2019).
Mostoufi-Moab, S. & Ward, L. M. Skeletal morbidity in children and adolescents during and following cancer therapy. Horm. Res. Paediatr. 91, 137–151 (2019).
Huncharek, M., Muscat, J. & Kupelnick, B. Impact of dairy products and dietary calcium on bone-mineral content in children: results of a meta-analysis. Bone 43, 312–321 (2008).
Rønne, M. S. et al. Bone mass development in childhood and its association with physical activity and vitamin D levels. The CHAMPS-Study DK. Calcif. Tissue Int. 104, 1–13 (2019).
Choudhary, A., Chou, J., Heller, G. & Sklar, C. Prevalence of vitamin D insufficiency in survivors of childhood cancer. Pediatr. Blood Cancer 60, 1237–1239 (2013).
Zhang, F. F., Saltzman, E., Must, A. & Parsons, S. K. Do childhood cancer survivors meet the diet and physical activity guidelines? A review of guidelines and literature. Int. J. Child Health Nutr. 1, 44–58. https://doi.org/10.6000/1929-4247.2012.01.01.06 (2012).
Vlachopoulos, D. et al. Determinants of bone outcomes in adolescent athletes at baseline: The PRO-BONE Study. Med. Sci. Sports Exerc. 49, 1389–1396 (2017).
Gil-Cosano, J. J. et al. The effect of an online exercise programme on bone health in paediatric cancer survivors (iBoneFIT): study protocol of a multi-centre randomized controlled trial. BMC Public Health 20, 1520 (2020).
Vandenbroucke, J. P. et al. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): explanation and elaboration. PLoS Med. 4, 1628–1654 (2007).
Moore, S. A. et al. Enhancing a somatic maturity prediction model. Med. Sci. Sports Exerc. 47, 1755–1764 (2015).
Gurney, J. G. et al. Bone mineral density among long-term survivors of childhood acute lymphoblastic leukemia: results from the St. Jude Lifetime Cohort Study. Pediatr. Blood Cancer 61, 1270–1276 (2014).
Migueles, J. H. et al. Comparability of published cut-points for the assessment of physical activity: Implications for data harmonization. Scand. J. Med. Sci. Sports 29, 566–574 (2019).
Evenson, K. R., Catellier, D. J., Gill, K., Ondrak, K. S. & McMurray, R. G. Calibration of two objective measures of physical activity for children. J. Sports Sci. 26, 1557–1565 (2008).
Weeks, B. K. & Beck, B. R. The BPAQ: a bone-specific physical activity assessment instrument. Osteoporos. Int 19, 1567–1577 (2008).
Ruiz, J. R. et al. Field-based fitness assessment in young people: the ALPHA health-related fitness test battery for children and adolescents. Br. J. Sports Med. 45, 518–524 (2011).
Ruiz, J. R. et al. Predictive validity of health-related fitness in youth: a systematic review. Br. J. Sports Med. 43, 909–923 (2009).
Julián Almárcegui, C. et al. Validity of a food-frequency questionnaire for estimating calcium intake in adolescent swimmers. Nutr. Hosp. 32, 1773–1779 (2015).
Bolek-Berquist, J. et al. Use of a questionnaire to assess vitamin D status in young adults. Public Health Nutr. 12, 236–243 (2009).
Shuhart, C. R. et al. Executive Summary of the 2019 ISCD Position Development Conference on Monitoring Treatment, DXA Cross-calibration and Least Significant Change, Spinal Cord Injury, Peri-prosthetic and Orthopedic Bone Health, Transgender Medicine, and Pediatrics. J. Clin. Densitom. 22, 453–471 (2019).
Johnson, J. & Dawson-Hughes, B. Precision and stability of dual-energy X-ray absorptiometry measurements. Calcif. Tissue Int. 49, 174–178 (1991).
Beck, T. J., Ruff, C. B., Warden, K. E., Scott, W. W. & Rao, G. U. Predicting femoral neck strength from bone mineral data. A structural approach. Invest. Radio. 25, 6–18 (1990).
Khoo, B. C. C. et al. In vivo short-term precision of hip structure analysis variables in comparison with bone mineral density using paired dual-energy X-ray absorptiometry scans from multi-center clinical trials. Bone 37, 112–121 (2005).
Hans, D., Goertzen, A. L., Krieg, M. A. & Leslie, W. D. Bone microarchitecture assessed by TBS predicts osteoporotic fractures independent of bone density: the Manitoba study. J. Bone Min. Res. 26, 2762–2769 (2011).
Pothuaud, L. et al. Evaluation of the potential use of trabecular bone score to complement bone mineral density in the diagnosis of osteoporosis: a preliminary spine BMD-matched, case-control study. J. Clin. Densitom. 12, 170–176 (2009).
Silva, B. C. et al. Trabecular bone score: a noninvasive analytical method based upon the DXA image. J. Bone Min. Res. 29, 518–530 (2014).
Shawwa, K. et al. Predictors of trabecular bone score in school children. Osteoporos. Int. 27, 703–710 (2016).
Del Rio, L., Di Gregorio, S. & Winzenrieth, R. WCO-IOF-ESCEO Seville 2014. Osteoporos. Int. 25(Suppl 2), 73–445 (2014).
Winzenrieth, R., Cormier, C., DiGregorio, S. & Del Rio, L. Influence of age and gender on spine bone density and TBS microarchitectural texture parameters in infants. Bone Abstracts 2, LB1. https://doi.org/10.1530/BONEABS.2.LB1 (2013).
Guagnelli, M. A. et al. Bone age as a correction factor for the analysis of trabecular bone score (TBS) in children. Arch. Osteoporos. 14, 26. https://doi.org/10.1007/S11657-019-0573-6 (2019).
Macdonald, H., Kontulainen, S., Petit, M., Janssen, P. & McKay, H. Bone strength and its determinants in pre- and early pubertal boys and girls. Bone 39, 598–608 (2006).
Davies, J. H., Evans, B. A. J. & Gregory, J. W. Bone mass acquisition in healthy children. Arch. Dis. Child 90, 373–378 (2005).
Agostinete, R. R. et al. The mediating role of lean soft tissue in the relationship between somatic maturation and bone density in adolescent practitioners and non-practitioners of sports. Int. J. Environ. Res. Public Health 18, 3008 (2021).
Stevens, J. P. Applied Multivariate Statistics for the Social Sciences (Lawrence Erlbaum, Mahwah, NJ, 2002).
Daly, R. M., Stenevi-Lundgren, S., Linden, C. & Karlsson, M. K. Muscle determinants of bone mass, geometry and strength in prepubertal girls. Med. Sci. Sports Exerc. 40, 1135–1141 (2008).
Högler, W. et al. Incidence of skeletal complications during treatment of childhood acute lymphoblastic leukemia: comparison of fracture risk with the General Practice Research Database. Pediatr. Blood Cancer 48, 21–27 (2007).
Lim, J. S. et al. Young age at diagnosis, male sex, and decreased lean mass are risk factors of osteoporosis in long-term survivors of osteosarcoma. J. Pediatr. Hematol. Oncol. 35, 54–60 (2013).
Muszynska-Roslan, K., Konstantynowicz, J., Krawczuk-Rybak, M. & Protas, P. Body composition and bone mass in survivors of childhood cancer. Pediatr. Blood Cancer 48, 200–204 (2007).
Muszynska-Roslan, K. et al. Bone mineral density in pediatric survivors of Hodgkin and non-Hodgkin lymphomas. Adv. Med. Sci. 59, 200–205 (2014).
Sioen, I., Lust, E., de Henauw, S., Moreno, L. A. & Jiménez-Pavón, D. Associations between body composition and bone health in children and adolescents: a systematic review. Calcif. Tissue Int. 99, 557–577 (2016).
Mostoufi-Moab, S. et al. Adverse fat depots and marrow adiposity are associated with skeletal deficits and insulin resistance in long-term survivors of pediatric hematopoietic stem cell transplantation. J. Bone Miner. Res. 30, 1657–1666 (2015).
Li, J., Kwong, D. L. W. & Chan, G. C. F. The effects of various irradiation doses on the growth and differentiation of marrow-derived human mesenchymal stromal cells. Pediatr. Transplant. 11, 379–387 (2007).
Baxter-Jones, A. D. G., Faulkner, R. A., Forwood, M. R., Mirwald, R. L. & Bailey, D. A. Bone mineral accrual from 8 to 30 years of age: an estimation of peak bone mass. J. Bone Min. Res. 26, 1729–1739 (2011).
Mogil, R. J. et al. Effect of low-magnitude, high-frequency mechanical stimulation on BMD among young childhood cancer survivors a randomized clinical trial. JAMA Oncol. 2, 908–914 (2016).
Elnaggar, R. K. & Mohamed, R. R. Aqua-plyometric exercises: potential implications for bone mineral density, functional capacity, and quality of life in survivors of childhood acute lymphoblastic leukemia. Semin. Oncol. Nurs. 37, 151225. https://doi.org/10.1016/j.soncn.2021.151225 (2021).
Dubnov-Raz, G. et al. Changes in fitness are associated with changes in body composition and bone health in children after cancer. Acta Paediatr. 104, 1055–1061 (2015).
Petit, M. A. et al. Proximal femur bone geometry is appropriately adapted to lean mass in overweight children and adolescents. Bone 36, 568–576 (2005).
Mostoufi-Moab, S. et al. Body composition abnormalities in long-term survivors of pediatric hematopoietic stem cell transplantation. J. Pediatr. 160, 122–128 (2012).
Mostoufi-Moab, S. et al. Bone density and structure in long-term survivors of pediatric allogeneic hematopoietic stem cell transplantation. J. Bone Miner. Res. 27, 760–769 (2012).
Mostoufi-Moab, S. & Halton, J. Bone morbidity in childhood leukemia: epidemiology, mechanisms, diagnosis, and treatment. Curr. Osteoporos. Rep. 12, 300–312 (2014).
Shawwa, K. et al. Predictors of trabecular bone score in school children. Osteoporos. Int. 27, 703–710 (2016).
Ubago-Guisado, E. et al. Longitudinal determinants of 12-month changes on bone health in adolescent male athletes. Arch. Osteoporos. 13, 106. https://doi.org/10.1007/S11657-018-0519-4 (2018).
Ireland, A. et al. Upper limb muscle-bone asymmetries and bone adaptation in elite youth tennis players. Med. Sci. Sports Exerc. 45, 1749–1758 (2013).
Acknowledgements
Herein, we would like to thank the disposal and consideration of all families involved in the investigation. The authors of this manuscript certify that they comply with the ethical guidelines for authorship and publishing in the Pediatric Research. The corresponding author affirms that all authors have contributed significantly to the work.
Funding
This study has been partially supported by the Spanish Ministry of Science and Innovation (ref: PID2020-117302RA-I00 financiado por MCIN/ AEI /10.13039/501100011033), La Caixa Foundation (ref: LCF/BQ/PR19/11700007), the University of Granada Plan Propio de Investigación 2021-Excellence actions: Unit of Excellence on Exercise, Nutrition and Health (UCEENS) and the CIBEROBN, Centro de Investigación Biomédica en Red (CB22/03/00058), Instituto de Salud Carlos III, Ministerio de Ciencia e Innovación and Unión Europea – European Regional Development Fund. A.M.-P. is the recipient of a predoctoral fellowship (FPU20/05530) by the Spanish Ministry of Education, Culture and Sport. E.U.-G. is supported by the Maria Zambrano fellowship by the Ministerio de Universidades y la Unión Europea—NextGenerationEU.
Author information
Authors and Affiliations
Contributions
A.M.-P., F.J.L.-C., E.U.-G., A.R.-S., J.J.G.-C., J.R.R. and L.G.-M. coordinated the study sites, including participant recruitment. A.M.-P., E.U.-G., A.R.-S., J.J.G.-C. and L.G.-M. wrote all the protocol versions, and obtained ethical approval and funding. L.G.-M. was the principal investigator. A.M.-P., F.J.L.-C., E.U.-G., A.R.-S., J.J.G.-C. and L.G.-M. were responsible for data collection, including clinical data. A.M.-P. conceptualised and designed the study with the support of E.U.-G., D.V. and L.G.-M. A.M.-P. drafted the initial manuscript and all co-authors were involved in the interpretation of data, revised the manuscript and approved the final manuscript as submitted.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent to participate
All parents and participants provided the required consent and assent, respectively.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Marmol-Perez, A., Ubago-Guisado, E., Llorente-Cantarero, F.J. et al. Determinants of bone parameters in young paediatric cancer survivors: the iBoneFIT project. Pediatr Res 94, 1538–1546 (2023). https://doi.org/10.1038/s41390-023-02645-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02645-8