Abstract
Background
More than half of children with pediatric acute liver failure (PALF) experience hepatic encephalopathy (HE), which is related to poor outcomes; however, HE is difficult to diagnose in children. The objective of this study was to evaluate if heart rate variability (HRV), a continuous measure of autonomic nervous system function, was related to the presence and severity of HE as well as clinical outcomes in children with PALF.
Methods
We conducted a retrospective observational cohort study of 38 critically ill children with PALF to examine the association between HRV and HE severity and clinical outcome. HRV was estimated using the integer HRV (HRVi). Categorical variables were compared using the Fisher Exact test and continuous variables were compared using Kruskal–Wallis tests. Associations between grades of HE and minimum and median HRVi were evaluated with Pearson’s correlation, with p values <0.05 considered significant.
Results
A more negative median and minimum HRVi, indicating poorer autonomic nervous system function, was significantly associated with abnormal EEG findings, presence of HE, and poor outcomes (death or listing for transplant).
Conclusions
Heart rate variability may hold promise to predict outcomes in children with PALF, but these findings should be replicated in a larger sample.
Impact
-
The findings of our study suggest that heart rate variability is associated with clinical outcomes in children with acute liver failure, a cohort for which prognostics are challenging, especially in young children and infants.
-
Use of heart rate variability in the clinical setting may facilitate earlier detection of children with pediatric acute liver failure (PALF) at high risk for severe hepatic encephalopathy and poor outcomes.
-
Identification of children with PALF at high risk of decompensation may assist clinicians in making decisions about liver transplantation, an important, but resource-limited, treatment of PALF.
Similar content being viewed by others
Introduction
Pediatric acute liver failure (PALF) is a rare but devastating syndrome, with up to 45% of children dying or needing a liver transplantation (LT).1 At this time, the ability of existing scoring systems to predict PALF clinical outcomes and the need for LT is limited.2 Neurologic complications, specifically the development of hepatic encephalopathy (HE) and cerebral edema are key determinants of patient outcomes. More than half of children with PALF experience HE, and about 1 in 10 suffer moderate or severe HE.1 Identifying these children early in their clinical course is critical as severe HE (grade III or IV) or progression of HE has been associated with higher 21-day mortality.1,3,4 Unfortunately, clinical assessment of HE is subjective and often difficult in children, and HE may not be apparent until advanced stages of the disease process.1,5,6 Electroencephalogram (EEG) has been investigated as a more objective measure of HE and studies have found that PALF patients with moderate-to-severe abnormalities on EEG are more likely to die or undergo LT; however, EEG requires time for interpretation and expensive, limited equipment.7
Heart rate variability (HRV) is defined as the fluctuation in time between consecutive heartbeats. In this study, HRV was estimated using the integer HRV (HRVi), which is the standard deviation of the heart rate in beats-per-minute measured every second over a 5-min time window. It is a proxy for autonomic nervous system (ANS) function via the ANS’ effects on the cardiovascular system. Cardiovascular homeostasis requires variable levels of parasympathetic and sympathetic input, which result in a continuously changing heart rate under normal physiologic conditions.8 ANS dysfunction is a maladaptive response to physiologic stress that results in an imbalance between parasympathetic and sympathetic inputs to the heart. Specifically, ANS dysfunction in critical illness is often due to excessive sympathetic nervous system activation and inappropriate regulation of the parasympathetic nervous system that results in a reduced HRV.9,10 In children, decreased HRV has been associated with severity of hypoxic–ischemic encephalopathy and toxic encephalopathy.11,12 HRVi is correlated with the presence and degree of HE in adult patients with chronic liver disease, but the relationship between HRV and HE in children with PALF has yet to be explored.13
In this study, we conducted a retrospective observational cohort study of children with PALF admitted to a pediatric intensive care unit (PICU) to determine whether there was an association between HRV and HE severity and clinical outcomes.
Materials and methods
Study subjects and setting
Patients were included in the study if they were admitted to the PICU at Ann & Robert H. Lurie Children’s Hospital of Chicago with PALF between 06/09/2012 to 12/31/2020 and had continuous heart rate data recorded from the bedside monitor during the first 24 h of PICU admission (of note, our PICU has been routinely collecting heart rate data since 2012). PALF was defined using the criteria established by the Pediatric Acute Liver Failure Study Group.2 Patients were divided into two groups based on outcomes: (1) patients with unfavorable outcomes (died or were listed for transplant during hospital admission), and (2) patients with favorable outcomes (survived with their native liver and were not listed for transplant). The Institutional Review Board at Lurie Children’s Hospital approved this study with a waiver of informed consent (IRB #2019-2431).
Study data
Clinical data such as patient outcome, hospital length of stay, clinical interventions, laboratory values, variables for calculating the Pediatric Risk of Mortality (PRISM) III scores, and diagnoses were obtained from the electronic health record. The grade of HE was manually extracted from the neurocritical care consult note in the patient’s chart. In the case that a grade of HE was not available in the chart via chart review, a grade was assigned in accordance with published criteria for children by two members of our study team (T.R.P. and L.N.S.-P.) based on the clinical assessments recorded by the primary medical team.5 In these cases, Grade 1 HE was assigned to children who were inconsolable, inattentive, confused, or were experiencing mood changes. Children with Grade 2 HE not only had findings of Grade 1 HE but were also noted to be hyper-reflexic, disinhibited, dysarthric, or ataxic. Children with Grade 3 HE had findings of Grade 1 or 2 HE but were also described as somnolent, stuporous, combative, or only able to follow simple commands. Children with Grade 4 HE were comatose, aroused only with painful stimuli, had deceberate or decorticate posturing, or absent reflexes.14
HRVi was age-normalized using previously published norms.8 A negative value indicates measurements that are lower than the age-based median. The minimum and median age-normalized HRVi were calculated in three overlapping time windows from the time of PICU admission: 0–6, 0–12 h, and 0–24 h. Incremental windows were chosen to elucidate how long clinicians would need to monitor a child with PALF before HRVi was able to predict clinical outcomes. Both minimum and median HRVi captured during these windows were compared to patient outcomes, grade of HE, and presence of severe encephalopathy during the first 48 h of admission (defined as an HE grade III or IV). Quantitative EEG measures were obtained with Persyst 14 (Persyst, San Diego, CA) for EEGs that had full tracings available. Quantitative EEG measures included total anterior and posterior power, theta:delta, and alpha:delta ratios. Data were stratified by age based on previously published norms.6 Additionally, EEGs were qualitatively labeled as normal vs. abnormal by a neurologist (A.C.P.) based on the absence or presence of generalized slowing, abnormal or absent sleep, asymmetry, attenuation, or burst suppression.
Statistical analyses
Data were analyzed using R (R Project for Statistical Computing, version 3.6.3). Categorical variables were compared using the Fisher Exact test and continuous variables were compared using Kruskal–Wallis tests. Associations between grades of HE and minimum and median HRVi were evaluated with Pearson’s correlation. The performance of HRVi to discriminate outcomes was evaluated using the area under the receiver operator curves (AUC).
Results
Thirty-eight children met study inclusion criteria. Fourteen (36.8%) experienced an unfavorable outcome (death or listed for transplant); 7 (18.4%) died and 7 (18.4%) were listed for transplant. Patients with unfavorable outcomes were significantly younger than patients with favorable outcomes. There were no other significant differences in patient demographics between groups (Table 1). The most common etiology of PALF was acetaminophen toxicity in 16 (42.1%) children. There was a significant difference in etiology of liver failure between groups; acetaminophen toxicity was more frequent in children who experienced a favorable outcome (58.3 vs. 14.3% in patients who had an unfavorable outcome) and more patients with metabolic disorders experienced an unfavorable outcome (35.7 vs. 4.2% for patients who experienced a favorable outcome). Patients who experienced a poor outcome were more likely to have grade III–IV HE (85 vs. 8%), higher PRISM III scores (19.5 vs. 4), higher maximum INR (4.5 vs. 3.2), and lower maximum alanine aminotransferase levels (6077 vs. 1064) (Table 1).
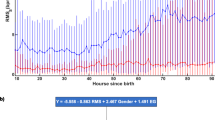
A more negative median and minimum HRVi, indicating poorer ANS function, was associated with poor outcomes (death or listing for transplant), higher grades of HE (Figs. 1 and 2), and abnormal EEG findings (Table 2).
HE hepatic encephalopathy, HRVi integer heart rate variability. *p < 0.05, **p < 0.01, ***p < 0.001. Favorable outcome: survived with native liver and were not listed for transplant. Unfavorable outcome: died or were listed for transplant during hospital admission. Presence of hepatic encephalopathy: HE grade III or IV. Absence of hepatic encephalopathy: HE grade II below.
At hours 6, 12, and 24, median HRVi and minimum HRVi were significantly associated with unfavorable outcomes (Figs. 3 and 4). Patients with Grade III or IV HE had lower median and minimum normalized HRVi measurements at all time points (Table 2). There was a moderate correlation between grade of HE at presentation and minimum HRVi up to 6 and 24 h and median HRVi of data collected before up to 12 and 24 h (Supplementary Table 1). There was a moderate correlation between highest grade of HE and minimum HRVi up to 6 and 24 h and median HRVi up to 12 and 24 h (Supplementary Table 1). The AUC curves demonstrated fair discrimination (AUC 0.70–0.80) of HE at 12 and 24 h using the minimum HRVi and at 6 and 12 h using the median HRVi. There was good discrimination of HE (AUC 0.80–0.90) at 6 h using the minimum HRVi at 24 h using the median HRVi. With respect to clinical outcomes, both median and minimum HRVi had fair discrimination between favorable and unfavorable outcomes at hours 12 and 24; there was fair discrimination between favorable and unfavorable outcomes at hour 6 for minimum HRVi, but not for median HRVi (Supplementary Table 2). Of note, the confidence intervals of the AUCs were wide for all measurements.
Quantitative EEG data during PICU admission were available for 21 of the 38 patients. Six of these patients had normal EEGs (Table 2). No patients with normal EEGs had HE, all patients with HE had an abnormal EEG. Because there are significant age-dependent differences in EEG measurements, the EEG data were separated into two cohorts: 9 patients aged 0–3 years and 12 patients aged 4–18 years.6 Patients with abnormal EEGs had significantly more negative minimum and median HRVi measurements collected before hours 6, 12, and 24. There were no significant correlations between anterior power, posterior power, theta:delta, or alpha:delta ratios and minimum HRVi and median HRVi for either age group (see Supplementary Tables 3 and 4).
Discussion
HE is a key clinical feature and determinant of outcomes in PALF, but early and accurate recognition can be difficult in pediatric patients. In this study, we show that HRVi is correlated with the presence and degree of HE, likelihood of poor outcomes, and the presence of abnormal EEG findings in patients with PALF.
HRV is representative of ANS function via its effects on cardiovascular homeostasis.10 ANS dysfunction may result in imbalanced parasympathetic and sympathetic signaling to the heart, which affects the time between consecutive heart beats. A lower HRVi (or a more negative normalized value as in our study) indicates more significant ANS dysfunction. The relationship between HRVi and PALF outcomes observed in our data is possibly mediated by HE, a condition characterized by global brain dysfunction, in part mediated by hyperammonemia, glutaminergic dysfunction, and inflammation.1,15,16 As liver function declines and HE worsens, more significant brain dysfunction ensues. Intracranial pressure also rises due to increased blood–brain barrier permeability and cerebral edema. While any of these physiologic insults could explain derangements in ANS function for patients with HE, the specific pathologic mechanism by which encephalopathy may cause ANS dysfunction remains an area of active study.15 A secondary mechanism connecting HRVi to HE and PALF outcomes could be ANS dysfunction related to the inflammatory and neuroendocrine response to a state of critical illness caused by severe PALF.8 This could be particularly important in children, as HE can appear late in the disease process, whereas significant associations between PALF outcomes and HRVi exist in the first day of PICU admission in our cohort.1
HRVi has shown to be useful in the detection of conditions with subtle or nonspecific clinical manifestations, such as organ dysfunction and septic shock.17,18,19,20 The impact that this information may have in clinical care is likely mediated by increased clinician awareness, particularly through the synthesis of very complex and dense data into a more succinct but still accurate measurement. For example, in a multi-site randomized control trial of low birth weight infants, patients whose heart rate characteristics (including HRV) were displayed to clinicians in the form of a score experienced a 22% relative reduction in mortality compared to controls.17 The mechanism by which display of HRV characteristics reduced mortality was not studied; however, the authors suspect that it is due to earlier recognition and treatment of sepsis by clinicians. Infants whose heart rate characteristics were displayed had >30% fewer deaths in the first 30 days after culture-proven sepsis. This study demonstrated the feasibility of incorporating HRV into routine clinical monitoring. If validated as a useful measurement, one could envision the use of continuous HRV measurements—along with other variables—to increase awareness and improve real-time stratification of children with PALF. Earlier recognition of brain dysfunction and deterioration in children with PALF could have significant impact in outcomes.
The incorporation of HRV data into existing workflows is especially feasible in the critical care units, where most patients have continuous heart rate monitoring during their admission. In resource- and personnel-limited settings, heart rate monitors may more accessible than EEG machines. Even in tertiary care centers, heart rate monitors are ubiquitous and the calculation of HRVi requires minimal time and expertise for interpretation as compared to EEG. Whereas a child with PALF may receive time-limited EEG monitoring for 24–48 h, their heart rate data are being collected throughout their entire hospital admission and HRVi can serve as a continuous, real-time marker of illness severity.
HRV could also be incorporated into existing predictive models. More effective prognostic tools could provide more accurate information for families and better direct organ allocation, the only known treatment for liver failure. Badke and colleagues developed the Heart Rate Variability Dysfunction (HRVD) score from data obtained from 7223 children and found that every one-point increase in the HRVD score was associated with a 25% increase in the odds of new or progressive multiple organ failure and a 51% increase in the odds of mortality.20 A potential next step would be to evaluate the addition of the HRVD score to a prognostic tool such as the Model for End-Stage Liver Disease (up to age 12 years), Pediatric End-Stage Liver Disease (children under age 12 years) to see if this improves prognostic accuracy.21
Limitations of this study include the small patient sample size and its retrospective design. Given the rare incidence of PALF, the study cohort is relatively small, and it was not possible to adequately adjust for potential confounders and produce reliable results. Interventions commonly employed in the PICU setting (e.g., mechanical ventilation) and medications used in the intensive care setting (e.g., beta-agonists, alpha-agonists, and inotropes) can affect HRVi.20 In a future study, analyses should be repeated in a larger cohort to evaluate the relationship between HE, PALF outcomes, and HRV while controlling for these variables. EEG data were only available for 21 patients during their PICU admission. Quantitative EEG norms are age-dependent, thus sub-analysis of a small number of subjects limits generalizations. There were no normal EEGs recorded in the younger age group, further limiting our analysis. Finally, findings from our single-center study may not be generalizable to all children with PALF.
Conclusion
Our findings suggest that decreased HRV, a noninvasive measure of ANS dysfunction, may facilitate earlier detection of children with PALF at high risk for severe encephalopathy and poor outcomes. This is of particular importance in young children and infants in whom a timely clinical determination of HE may present a challenge to clinicians. Further validation in a larger, multi-center cohort is warranted.
Data availability
De-identified data can be provided to researchers upon reasonable request. Requests can be made to the corresponding author.
References
Ciocca, M. et al. Prognostic factors in paediatric acute liver failure. Arch. Dis. Child. 93, 48–51 (2008).
Lu, B. R. et al. Evaluation of the liver injury unit scoring system to predict survival in a multinational study of pediatric acute liver failure. J. Pediatr. 162, 1010–1016 (2013).
Squires, R. H. Jr et al. Acute liver failure in children: the first 348 patients in the pediatric acute liver failure study group. J. Pediatr. 148, 652–658 (2006).
Ng, V. L. et al. Outcomes of children with and without hepatic encephalopathy from the Pediatric Acute Liver Failure (PALF) Study Group. J. Pediatr. Gastroenterol. Nutr. 63, 357 (2016).
Rivera-Penera, T. et al. Delayed encephalopathy in fulminant hepatic failure in the pediatric population and the role of liver transplantation. J. Pediatr. Gastroenterol. Nutr. 24, 128–134 (1997).
Press, C. A. et al. Spectral electroencephalogram analysis for the evaluation of encephalopathy grade in children with acute liver failure. Pediatr. Crit. Care Med. 18, 64–72 (2017).
Hussain, E. et al. EEG abnormalities are associated with increased risk of transplant or poor outcome in children with acute liver failure. J. Pediatr. Gastroenterol. Nutr. 58, 449–456 (2014).
Badke, C. M., Marsillio, L. E., Weese-Mayer, D. E. & Sanchez-Pinto, L. N. Autonomic nervous system dysfunction in pediatric sepsis. Front. Pediatr. 6, 280 (2018).
Dünser, M. W. & Hasibeder, W. R. Sympathetic overstimulation during critical illness: adverse effects of adrenergic stress. J. Intensive Care Med. 24, 293–316 (2009).
Schmidt, H. et al. The alteration of autonomic function in multiple organ dysfunction syndrome. Crit. Care Clin. 24, 149–163 (2008).
Goulding, R. M. et al. Heart rate variability in hypoxic ischemic encephalopathy: correlation with EEG grade and 2-y neurodevelopmental outcome. Pediatr. Res. 77, 681–687 (2015).
Yamada, Y. et al. Brainstem mechanisms of autonomic dysfunction in encephalopathy‐associated Shiga toxin 2 intoxication. Ann. Neurol. 45, 716–723 (1999).
Fleisher, L. A., Fleckenstein, J. F., Frank, S. M. & Thuluvath, P. J. Heart rate variability as a predictor of autonomic dysfunction in patients awaiting liver transplantation. Dig. Dis. Sci. 45, 340–344 (2000).
Squires, R. H. Acute liver failure in children. Semin. Liver Dis. 28, 153–166 (2008).
Rega, D. et al. Patients with minimal hepatic encephalopathy show altered thermal sensitivity and autonomic function. J. Clin. Med. 10, 239 (2021).
Felipo, V. Hepatic encephalopathy: effects of liver failure on brain function. Nat. Rev. Neurosci. 14, 851–858 (2013).
Moorman, J. R. et al. Mortality reduction by heart rate characteristic monitoring in very low birth weight neonates: a randomized trial. J. Pediatrics 159, 900–906 (2011).
Kasaoka, S., Nakahara, T., Kawamura, Y., Tsuruta, R. & Maekawa, T. Real-time monitoring of heart rate variability in critically ill patients. J. Crit. Care 25, 313–316 (2010).
Ahmad, S. et al. Continuous multi-parameter heart rate variability analysis heralds onset of sepsis in adults. PLoS ONE 4, e6642 (2009).
Badke, C. M., Marsillio, L. E., Carroll, M. S., Weese-Mayer, D. E. & Sanchez-Pinto, L. N. Development of a heart rate variability risk score to predict organ dysfunction and death in critically ill children. Pediatr. Crit. Care Med. 22, e437–e447 (2021).
Jain, V. & Dhawan, A. Prognostic modeling in pediatric acute liver failure. Liver Transplant. 22, 1418–1430 (2016).
Funding
None.
Author information
Authors and Affiliations
Contributions
All authors made substantial contributions to conception and design of this study as well as acquisition and interpretation of data. Data analysis was conducted by T.R.P. and Dr. L.N.S.-P. T.R.P. drafted this manuscript. All authors revised the manuscript critically for important intellectual content and approved the final version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The Institutional Review Board at Lurie Children’s Hospital approved this study with a waiver of informed consent (IRB #2019-2431). Patient consent was not required.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pendergrast, T.R., Chapin, C.A., Kriegermeier, A.A. et al. Heart rate variability is associated with encephalopathy and outcomes in pediatric acute liver failure. Pediatr Res 93, 1348–1353 (2023). https://doi.org/10.1038/s41390-022-02225-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-022-02225-2