Abstract
Background
Infants born preterm are known to be at risk for abnormal brain development and adverse neurobehavioral outcomes. To improve early neurodevelopment, several non-pharmacological interventions have been developed and implemented in the neonatal intensive care unit (NICU). Sensory-based interventions seem to improve short-term neurodevelopmental outcomes in the inherently stressful NICU environment. However, how this type of intervention affects brain development in the preterm population remains unclear.
Methods
A systematic review of the literature was conducted for published studies in the past 20 years reporting the effects of early, non-pharmacological, sensory-based interventions on the neonatal brain after preterm birth.
Results
Twelve randomized controlled trials (RCT) reporting short-term effects of auditory, tactile, and multisensory interventions were included after the screening of 1202 articles. Large heterogeneity was identified among studies in relation to both types of intervention and outcomes. Three areas of focus for sensory interventions were identified: auditory-based, tactile-based, and multisensory interventions.
Conclusions
Diversity in interventions and outcome measures challenges the possibility to perform an integrative synthesis of results and to translate these for evidence-based clinical practice. This review identifies gaps in the literature and methodological challenges for the implementation of RCTs of sensory interventions in the NICU.
Impact
-
This paper represents the first systematic review to investigate the effect of non-pharmacological, sensory-based interventions in the NICU on neonatal brain development.
-
Although reviewed RCTs present evidence on the impact of such interventions on the neonatal brain following preterm birth, it is not yet possible to formulate clear guidelines for clinical practice.
-
This review integrates existing literature on the effect of sensory-based interventions on the brain after preterm birth and identifies methodological challenges for the conduction of high-quality RCTs.
Similar content being viewed by others
Introduction
Preterm infants (infants born before 37 weeks of gestation) are at increased risk for mortality and long-term morbidity. Complications related to preterm birth are the leading cause of mortality in the neonatal period.1 Moreover, prematurity is associated with a specific risk to the maturation of sensory systems,2 the organization of brain structure and function3,4,5 and long-term developmental outcome.6,7,8,9 During the last months of gestation, the human brain undergoes a rapid sequence of functional and structural maturational changes, for which both endogenous and exogenous or environmental stimuli are fundamental.10,11 In congruence, research shows that preterm infants exhibit delayed maturation of white matter integrity, brain growth, and cortical morphology compared to term-born infants.12,13,14 Because we diagnose less overt brain lesions in preterm infants,15,16 this has led to new thinking about current preterm brain injury: not secondary to overt tissue loss, but rather because of a series of “dysmaturational” events leading to altered white matter connectivity.17,18 During the time preterm infants spend on the neonatal intensive care unit (NICU) (which would be the third trimester in pregnancy), it is the white matter that has been shown to be especially vulnerable.18
Although clinical perinatal variables (e.g., inflammation, hypoxia, hyperoxia, etc.) have a major impact on neurodevelopmental outcome,19,20,21 the type and quality of sensory experiences obtained during the prenatal and early postnatal period are also crucial for the maturation of the developing brain.22,23 In utero, the human fetus experiences temporally organized, cyclic and multimodal stimulation across the senses.24 After a preterm birth, infants are exposed to the environment of a NICU where duration, complexity, and intensity of sensory exposure is radically distinct to that experienced in utero.24,25 A preterm infant is exposed to relatively loud noises from monitoring devices, daily physical examinations, diaper changes, intravenous line placements, medication, abnormal circadian cues (e.g., 24-h light exposure), respiratory support, etc. Consequently, preterm infants need to cope, during a critical developmental period, with sensory stimulation that is not age-appropriate and for which they are yet immature. All these NICU variables may affect both spontaneous and evoked brain activity during the NICU period. This could potentially present long-lasting consequences for the development of the brain.26,27,28
In this line, the impact of both intense sensory exposure and sensory abatement in the NICU environment has become a topic of concern for an infant’s neurodevelopment. In particular, it has been suggested that sensory characteristics of the NICU environment can significantly affect the developing brain.29,30 Sensory-based interventions aiming at reducing neonatal stress by modulation of the NICU environment have been a fundamental part of developmental care programs implemented in the NICU.28,29 Adapting the early environment to support the needs of the preterm infant includes strategies that modulate the sensory input received by the infants and that target one or more sensory systems, such as cycled light, noise reduction and music interventions, positioning, skin-to-skin contact, and support of parental care.31,32 These types of interventions have been related to physiological stability, sleep–wake cycling, shorter NICU stay, and early development in preterm infants.33,34,35,36 Recent animal and human studies also suggest that sensory interventions could impact the stress response, epigenetics pathways, and, consequently, long-term neurodevelopment.37,38,39,40 A recent integrative review by Pineda et al.35 showed that early positive sensory experiences in very preterm infants are associated with improved infant and maternal outcomes in the NICU, although there is yet little evidence to suggest there are improved long-term outcomes.
Enhancing preterm infants’ sensory experience during this critical window of development could enhance the quality of care of the vulnerable preterm population and improve their long-term neurodevelopmental outcomes. Pioneer studies have shown that neurodevelopmental supportive programs in the vulnerable period following preterm birth can have a positive impact on brain structure and function.23,41 Although increasingly more practitioners consider implementing sensory interventions as environmental enrichment in the NICU, little is known about the effects of early sensory-based interventions in the neonatal brain. An improved understanding of the effects that non-pharmacological, sensory-based interventions may have on the developmental trajectories of the preterm brain is of extreme importance. To address this issue, our aim was to systematically analyze the current literature on the effects of non-pharmacological, sensory-based interventions on brain development in preterm neonates.
Methods
Aim
The aim of this systematic review is to explore available evidence on the impact of non-pharmacological, sensory-based interventions implemented in the NICU on the development of the neonatal brain after preterm birth.
Literature search and design
A systematic search for studies was conducted following the criteria of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement.42 The literature search was performed employing the online bibliographic databases PubMed, EMBASE, and the Cochrane Library. Search terms and an example of syntax utilized are included in Table 1. The protocol for the search was registered in the International Prospective Register of Systematic Reviews (PROSPERO) (CRD42018090909) and an update of the original search was performed in October 2020.
Two reviewers screened studies for inclusion. Studies were screened first by title, and when necessary, abstracts were retrieved for review. The full-text articles of potentially relevant studies were reviewed for final inclusion. When the relevance of the studies was unclear, it was resolved through discussion among the reviewers.
Inclusion criteria for study selection included (a) reports published in a peer-reviewed journal in the past 20 years, (b) population preterm infants <37 weeks gestational age (GA), (c) non-pharmacological, unimodal, or multimodal sensory interventions taking place in the NICU and performed by health care workers or parents, (d) primary or secondary outcomes related to brain development reported using neurobiological plausible measures of brain maturation such as ultrasound, near-infrared spectroscopy, magnetic resonance imaging (MRI), functional MRI (fMRI), diffusion tensor imaging (DTI) and electroencephalogram, and (e) randomized controlled trial (RCT) design.
Studies analyzing the effects of unimodal or multimodal sensory-based interventions targeting the auditory, olfactory/gustatory, tactile, and visual sensory systems were included. Studies of programs promoting parental support and involvement in the NICU were included as long as the main focus was set on a sensory exchange between parents and infants.
A preliminary exploration of the literature indicated that the effects of head positioning interventions in the development of brain injury in preterm infants have been previously and extensively addressed.43,44,45 Therefore, it was decided to focus on other types of sensory interventions and interventions addressing exclusively the vestibular and/or proprioceptive systems were excluded from this review. In addition, studies including only kinaesthetic-based interventions were also excluded in order to avoid studies reporting effects of specific physiotherapy treatments. Given the multiplicity and diversity of areas of interventions in the framework of developmental support programs (such as NIDCAP—for a review see ref. 46) developmental support-related studies were excluded from this review. Lastly, to improve the strength of the conclusions, pilot or feasibility studies with a sample size of ≤10 in the intervention group were excluded.
One reviewer performed cross-referencing on the included studies, which yielded no additional studies that met the inclusion criteria described above.
Data extraction and risk of bias assessment
Data were extracted from included studies using a predetermined form. Blinding of authorship was not performed.
Assessment of the studies was performed independently by two authors using the Cochrane Risk of Bias (RoB 2.0) tool for RCTs.47 Disagreements regarding the critical assessment were resolved by discussion until consensus was reached. In situations where consensus could not be reached, consultation with a third author took place. Critical assessments for each study were conducted qualifying five domains of bias separately; judgments were expressed for each domain as “low risk”, “high risk,” or “some concerns” of bias.
Results
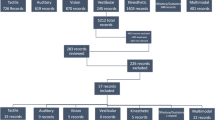
In total, 1900 records were identified after the initial database search. An additional three articles from other sources were included. Duplicate removal resulted in 1202 studies to be assessed for eligibility based on title and abstract. The resulting 42 studies were read the full text. A further 30 articles were excluded for non-adherence to inclusion criteria, leaving 12 RCTs for qualitative synthesis.48,49,50,51,52,53,54,55,56,57,58,59 The PRISMA flow diagram, which quantitatively summarizes this process and elaborates on reasons for exclusion, is included below (Fig. 1).
Due to the heterogeneous nature of the trials, comprising a diversity of interventions and outcomes, study findings were qualitatively summarized and a meta-analysis was not deemed appropriate. Assessment of risk of bias across studies is reported in Fig. 2.
No studies were found reporting effects of specific visual, olfactory, or gustatory interventions and matching the inclusion criteria of this review. Studies were grouped according to the type of interventions, which led to the identification of three areas of focus: auditory, tactile, and multisensory interventions. Interventions based on auditory input consisted of exposure to recorded mother’s voice and heartbeat sounds,50 exposure to live music by an expert therapist,58 and exposure to recorded music.55,56,59 Regarding tactile-based interventions, two studies reported on orocutaneous stimulation,53,54 one study reported on massage therapy,52 and one study reported on skin-to-skin contact.51 An additional three studies were identified reporting effects of a multimodal intervention in the frame of a parental support program in the NICU. These studies were conducted in the same base population using the family nurture intervention (FNI).48,49,57 The design involved a long-term intervention program (from birth or 30–32 weeks PMA and throughout hospitalization), in which the mother was trained and stimulated to perform kangaroo care, scent exchange, vocalizations, and other types of sensory and affective interactions with her infant during admission in the NICU.
Characteristics of respective studies and interventions are summarized in Table 2. A summary of the type of outcomes and main findings for each study can be found in Table 3.
Auditory interventions
In the study of Webb et al., stimulation was conducted with voice and biological sounds (heartbeat) recording. Based on ultrasound measurements, authors reported an increase in the size of both the left and right auditory cortex after daily exposure to recorded mother’s voice and heartbeat sounds (p = 0.015 and p = <0.001, for left and right cortex, respectively).50
Haslbeck et al.58 found that though structural brain connectivity appears to be largely unaffected by Creative Music Therapy, functional connectivity (fc) was higher in the left precentral gyrus and partly the left supplementary motor area for the group receiving the intervention.
To assess the effects of a recorded music intervention, MRI and fMRI imaging at term-equivalent age (TEA) were used for outcome analysis.55,56,59 Sa de Almeida et al.59 reported results of DTI scalars, tractography, and amygdala volumetric analyses. Significant differences were found between groups for FA, mean diffusivity (MD), axial diffusivity (AD), and radial diffusivity (RD) in regions of interest (ROIs) after music exposure (see Table 3 for further details). Additionally, the authors found that amygdala volumes were smaller for the preterm group that had not received the music intervention. The study of Lordier et al.55 processed fMRI data identifying 14 functional resting-state networks (RSNs) used as ROIs. Fc was assessed by means of a nonparametric estimator (accordance) reflecting coupling between two ROIs. The authors first identified a circuitry of interest composed by three network modules interconnected by the salience network. This circuit showed reduced functional connectivity in the preterm control group when compared with the full-term group. In this same circuit, the preterm infants exposed to the music intervention showed significantly higher RS-fc than the control group. RSN coupling was particularly increased between the auditory, sensory-motor, and medial superior frontal networks with the salience network, between the auditory and the medial superior frontal networks, and between the salience and thalamus networks and salience and precuneus networks. In a second study, Lordier et al.56 used a psychophysiological interaction approach to fMRI. During MRI scanning, five conditions of music stimuli were presented: Silence, Original music (as provided to preterm intervention group), Tempo music, Transposed music, and Background music. Their results showed that when exposed to the music received during the music intervention period, the preterm intervention group displayed increased connectivity between the right primary auditory cortex and right thalamus, left middle cingulate cortex (MCC) and caudate nucleus compared to the preterm control group. Additionally, connectivity between the left primary auditory cortex region and the left superior temporal gyrus and the left MCC was also increased in the preterm intervention group when compared to the full-term group.
Tactile interventions
Two studies, performed in the same base population, used pulsed stimulation delivered to the baby’s oral sensorium (soft tissues of the infant’s lips–anterior tongue–intraoral mucosa–jaw) through a silicone pacifier.53,54 The intervention was delivered at a young age (around 32 weeks PMA) and at regular intervals and EGG was measured during the intervention. EEG leads were placed in the C3, C4, P3, and P4 positions and recordings were done after stimulation blocks. Song et al.53 investigated spectral edge frequency, fc = 90% (SEF-90), derived from nine sequential epochs at 1-min intervals for both intervention and control groups. They observed reorganization of SEF-90 in both the left and right hemispheres (p = 0.005 and p < 0.0001, respectively). The authors reported a significant difference between hemispheres on the polarity of the frequency shift in infant cortical SEF during oral somatosensory stimulation. An after-effect was also reported for the intervention group. Barlow et al.54 measured amplitude-integrated EEG (aEEG) margins and range-EEG (rEEG) amplitude bands measured at 1-min intervals. aEEG and rEEG amplitude measures were compared between four stimulus conditions (sham/pacifier, pacifier in mouth/pacifier out of mouth). Bands were defined as A: 0–10 μV; B: 10–25 μV; C: 25–50 μV; D: 50–100 μV; and E: >100 μV. These authors found that aEEG maxima, mean, and minima in the left hemisphere (p = <0.0001) and aEEG maxima and mean in the right hemisphere (p < 0.001 and p = 0.015, respectively) were affected by the intervention, as well as crosshead measures of aEEG maxima and mean (p < 0.0001). Significant reorganization of rEEG amplitude bands was identified in both hemispheres. Significant Bonferroni pairwise contrasts between the intervention and control groups were found for bands B, C, D, and E in the left hemisphere (p < 0.001 for C, D, and E bands, p = 0.020 for B band), bands C and D in the right hemisphere (p < 0.001), and bands C and E for the crosshead amplitude bands (p < 0.001). Cortical asymmetry was observed, and authors reported an apparent shift of rEEG power in the intervention group from the E and D bands to the C band.
Guzzetta et al.52 reported on a massage intervention administered upon the infant reaching 10 postnatal days. EEG recordings were made before and after the intervention period (at around 1 and 4 weeks of age, T0 and T1, respectively) for ~40 min including all stages of sleep. Active sleep was selected for further analysis. EEG signal was obtained from eight leads (fp1, fp2, C3, C4, T3, T4, O1, and O2) plus a reference electrode. The average of the values of all electrodes determined the global absolute power, while the average of the values of paired homotopic monopolar derivations in each lobe determined local absolute power. Fast Fourier transformation was used and spectrum bands were classified as delta (0.5–4.0 Hz), theta (4.5–7.5 Hz), alpha (8.0–11.0 Hz), and beta (15.5–20 Hz). The authors reported significant variations in global absolute power in the alpha and delta band over the period T0–T1 for infants in the control group. In the intervention group, significant differences between t0 and t1 were also found for local power, with an increase of power on the central regions for delta and theta bands and a decrease in the temporal regions for delta and alpha bands. Significant interactions between time and participant group were found for the global absolute power in the delta band and for the local power in central leads of the beta and the delta band Differences between groups were mostly due to the significant decrease in EEG spectral power found between t0 and t1, especially in the delta band, in the control group, and not in the intervention group.
Ludington-Hoe et al.51 analyzed the effects of a single session of skin-to-skin contact using electroencephalographic/polysomnographic measures of neonatal sleep organization. Sleep status was analyzed through analysis of behavioral and EEG data. For data analysis, the authors compared the data from the full-test period with the full-pretest period. Authors determined sleep status (quiet (QS), active (AS), and indeterminate sleep (IS)) by means of visual analysis and scoring of EEG continuity, discontinuity, and arousals. Changes in discontinuity within QS, rapid eye movement (REM) counts within AS, arousals, mean duration of the cycle, and percentages of QS, AS, and IS were measured. Arousal was defined as test–pretest changes in the percentage of time of EEG micro-arousal and extended arousal within the respective time periods. Results showed that the percentage of time of arousals was significantly lower for the intervention group as compared to controls across the study period and during quiet and active sleep. The control group showed increased arousals during the test period, while REM counts were significantly lower in the intervention group during active sleep and over the study. Changes in the EEG β/α ratio and EEG left/right hemisphere correlation were also assessed by the authors, although no significant differences in these outcome measures were reported between the control and the intervention group.
Multisensory interventions
Three studies were identified reporting effects of a multisensory intervention in the frame of a parental support program in the NICU.48,49,57
In the study by Welch et al.,48 EEG power was computed for each of the 125 electrodes. Infants assigned to the FNI group showed a significant increase in EEG power in the high-frequency bands during both quiet and active sleep (>10 Hz, p < 0.01 and p < 0.05, respectively). At term age, no significant differences between frontal power were found between groups, although, over the developmental trajectory, significant age-by-group interactions were found for some brain regions. A significant interaction between twin/singleton status and group was found.
In a posterior study, Welch et al.57 performed a follow-up analysis on this population. EEG power was computed in active and quiet sleep in ten frequency bands (1–48 Hz) for ten brain regions. Rates of change in EEG power per week of age within each region and each frequency band were calculated. A percent change in power/week from the preterm age to the near to term age was computed. The authors reported that the developmental rate of change in EEG power was increased in the intervention group in 132/200 tests (p < 0.05).
In the study by Myers et al.,49 EEG coherence was computed between all possible pairs of electrodes for ten frequency bands. Electrodes were grouped to define regions. The EEG setup consisted of 124 leads. Infants in the FNI group showed lower EEG coherence within the left frontal polar region for frequencies between 4 and 18 Hz and lower EEG coherence within the right frontal polar regions for frequencies between 0 and 12 Hz during quiet sleep (p < 0.01). In both quiet and active sleep, multiple significant reductions in coherence between regions (also across hemispheres) were found (p < 0.01). A reduction in coherence between the left and right frontal polar regions at a frequency of 10–12 Hz was the most notable finding (p = 0.00011, quiet sleep).
Discussion
In this systematic review, we included RCTs exploring non-pharmacological, sensory-based interventions in the NICU and their effect on the neonatal brain after preterm birth. The included studies examined auditory, tactile, and multisensory interventions. Outcomes were based on measures derived from EEG, MRI/fMRI, and cranial ultrasound. The results of the included studies would support the notion that environmental enrichment using sensory-based interventions may be beneficial for the development of the brain after preterm birth. However, two issues are fundamental for further analysis and will be addressed in this discussion, (a) although reported results seem promising, the heterogeneity of interventions, methods, and outcomes measures challenges the possibility of drawing integrative and reliable conclusions, and (b) the risk of bias of the reviewed reports was globally high and reflects the existing methodological challenges for the implementation of intervention RCTs in the NICU.
The main findings of auditory-based intervention studies were increased size of the auditory cortex in the first month upon exposure to maternal voice and biological sounds50 and changes in microstructural white matter and functional connectivity in specific circuits of interest after music-related interventions.55,56,58,59 Exposure to recorded music showed increased functional connectivity between the salience network and regions underlying sensory and higher-order cognitive functions together with structural maturation on auditory and emotional processing neural pathways upon exposure to the intervention.55,56,59 Creative music therapy shows effects on functional connectivity in networks implicated in higher-order cognitive and socio-emotional functions in preterm infants.58 These findings are in line with literature reporting short-term improvements in physiological stability in the preterm population have been reported after exposure to maternal biological sounds and maternal voice60,61,62,63 and after music interventions, using maternal singing or prerecorded music.64,65,66,67 Furthermore, interpretation of results can be done in the context of early brain network development, where preterm birth has been shown to affect maturational pathways.4,68 Neural activity plays a critical role in brain development69 and it has been well established that early experiences can sculpt the white matter wiring of the nervous system.70,71 Plasticity of brain connectivity is at its highest during early preterm brain development, in which pre-established white matter connections show a heightened sensitivity to endogenous and exogenous activity-induced modification. Although the results reported on the included RCTs provide initial evidence on the effect of music interventions in brain regions known to be altered by prematurity, a thorough analysis of the effects of the NICU auditory exposure and increasing levels of evidence on early auditory interventions is necessary. Especially when considering the social7 and language72,73 difficulties that premature children present in their development even in the absence of brain injury or major disabilities.
It should be noted that the identified studies differ not only in the type of auditory stimulation but also in the timing and duration of the intervention. Start time and duration of an auditory intervention in such a vulnerable population should be always carefully assessed, in reference to available evidence of the maturation of the auditory pathways (as in ref. 56) and close assessment of the physiological and behavioral responses of each infant. The same holds true for the type of stimuli, duration and intensity (in dBA), and methodology selected for stimuli administration. Live music, exposure to music via headphones, or via a player attached to the incubator may have very different acoustic characteristics that impact the preterm infant.74 Issues such as reverberation or signal-to-noise ratio must be considered for the study design and included in the reporting of results.
Tactile-based intervention studies differed in intervention and outcome type. A specific type of tactile input was used in included studies reporting orosensory stimulation.53,54 Both studies reported modulation of aEEG, reorganization of rEEG amplitude bands, and reorganization of SEF-90 in infants exposed to the intervention. Significant decrease in EEG spectral power during active sleep in massaged infants and lower arousals and REM counts in infants receiving skin-to-skin contact. These results would suggest a more mature neurophysiological sleep organization in infants exposed to this type of tactile stimuli. Some studies have also previously related tactile stimulation with improved behavioral measures of sleep in preterm infants, although evidence levels are variable.36 Advances in the field beyond the “state-of-the-art’ show that sleep has a very specific and crucial role during early brain development. From the early pioneering work of Roffwarg et al.75 to the current work of Blumberg et al.,76 is now becoming clear that “active sleep’ in the fetus has a very specific role: produce endogenous or spontaneous, “self-organized” network activity.77 Recent studies have put forward the close relationship between sleep and brain development in the preterm population,78,79 which highlights the impact that these sensory interventions promoting sleep could have on long-term outcomes.
It is clear that the behavioral activity of preterm infants in the incubator in the NICU is completely different from that of an aged-matched fetus in the womb. A preterm infant is exposed to NICU variables that may affect both spontaneous and evoked brain activity during the NICU period. Therefore, optimizing active sleep (endogenous brain activity) or stimulating wake activities (exogenous activity) can have a true impact on development as aEEG studies have shown a link between neural activity and brain volumes.80 The results of the tactile-based, interpersonal intervention studies add to an array of studies that have put forward the importance of affective interpersonal touch for human affiliative behaviors.81,82,83 Affective interpersonal touch is considered to constitute the neurobiological substrate for the development of the social brain and for the expression of social behavior.84,85 A neuroimaging study showed that the frequency of maternal touch positively predicted connectivity in brain regions of their children associated with social functioning.86 In the NICU, a recent publication on affective touch (administered with a brush to optimally stimulate C-tactile fibers) before painful procedures was reported to attenuate noxious-evoked brain activity in full-term newborns.71 Comforting touch and skin-to-skin (kangaroo care) contact have also been shown to have several beneficial effects on infants in the NICU.33,87 In particular, previous studies reported positive effects of kangaroo care on physiological stability, early neurobehavioral, and mother–infant attachment.88,89 Furthermore, previous studies also support the hypothesis that positive effects of comforting tactile interventions could translate into enhanced maturational patterns in the neonatal brain.90,91,92
Studies reporting results of multisensory interventions were based on a sensory exchange between parents and infants and also focused on EEG-related measures.48,49 Welch et al. found increased EEG power in high-frequency bands, while Myers et al. found lower EEG coherence within and between several brain regions. In a follow-up study, Myers et al.57 reported an increase in developmental rates (change/week) of EEG spectral power. Increased power and decreased coherence have been associated with cortical maturation.93 Both parameters have previously been connected to long-term outcomes. Increased frontal power in preterm infants, term infants, and older children was found to be positively predictive of developmental outcomes at a later age94,95 and lower coherence in infants was linked to improved joint attention at 18 months of age.96 These results are in line with research in both animals and humans, which suggests that early positive parental care can have positive effects in epigenetic programming, regulation of the stress system, and the development of the brain.97,98,99,100
Whether the specific parent-focused sensory intervention discussed above are also influential on long-term brain and developmental outcomes of preterm infants require further research. Another factor that should be considered when assessing the effects of these interventions is that it remains difficult to determine whether observed outcomes are a result of the specific attention for parental involvement or of other sensory-specific aspects of the intervention. The interventions of the included studies focused on parent–infant interactions such as scent-cloth exchange, eye contact, and skin-to-skin contact. Previous research linking parental involvement to improved neurobehavioral and neurodevelopmental outcomes mostly includes (advice on) touching and holding the infant and sensitivity training for parents23,101,102 and were not focused on sensory interventions, and therefore were not included in this review. It is noted that it can be next to impossible to separate these different aspects of certain sensory interventions in a research model, and even to do so would attempt against the ecological validity of the study. Hence, it remains a challenge to determine whether there is one aspect of the intervention to which the changes in the neonatal brain measures should be mainly attributed, or whether the changes are due to the multimodal/affective characteristics of the presented stimuli. In the scenario of these early interventions in the NICU, a dichotomic approach could be reductionist and it is needed to acknowledge the complex intertwinement between sensory and affective dimensions.
A major confounder in all these studies is the absence of exact measurements of the sensory intervention. Delivering on this issue can be challenging in the context of family interventions. To this end, comprehensive quantitative and qualitative reporting on the infants’ sensory experience both within and outside the intervention could be implemented and standardized for these types of studies. In addition, there is a clear need for a true behavioral and neural activity coupling to understand the mediating effect of each intervention to further optimize the NICU as a neurodevelopmental unit.
As shown in this review, a plethora of NICU interventions have been studied with the aim to optimize both endogenous and exogenous brain activity. All in all, there is still a lack of reliable evidence and many questions remain unanswered in terms of the effects of sensory-based interventions in the neonatal brain. The modulation of EEG early activity via sensory interventions, as reported on several studies included in this review, acquires particular relevance when contemplating the importance of early brain activity for brain morphology and microstructure103,104 and the effect of early cortical changes on later neurodevelopmental outcomes.20 In this context, the conduction of further research of high methodological quality and low bias is paramount.
Strength and limitations of the review and of included studies
Regarding limitations of the study, it is important to address that this review did not include non-English language studies or data published in non-article formats. This could have left out relevant literature. Although the exclusion of pilot studies and the inclusion of only one type of study design may have excluded relevant literature, the decision was made considering the vulnerability of the population and the importance of identifying potential evidence-based clinical practices. To ensure trustworthiness, two reviewers performed an independent selection of studies and quality assessment. These selection criteria taken together with a rigorous methodology constitute strengths of the current manuscript.
It should be noted that setting up an early intervention study in a neonatal intensive care unit environment poses many methodological and ethical challenges, especially since the conductance of the study should not interfere with standard care. Blinding of nurses, for example, may not be possible for many of the proposed interventions, and inclusion rates may be very low. A source of bias is that early intervention studies in the NICU can be especially prone to be a “spillover” effect. Parents in the unit that were made aware of the research or that observe the administration of, for example, an auditory stimulation protocol, could be inclined to increase this type of interaction with their infant. If these parents participate in the study in the intervention or control arm, this may introduce significant bias. Authors should pay special attention to this issue together with the reporting of sensory interactions outside the assessed intervention. The inclusion of a report on parental interactions for the participants of the study, or a cluster randomization design—if possible/appropriate—could therefore be considered. Furthermore, detailed reporting on study setup, data collection, and analysis, as well as on the study population, are essential in lending reliability to results obtained. Especially, interventions should be detailed with care and, ideally, reporting guidelines will be developed and updated. These may also aid in standardizing procedures and, consequently, reducing at least some of the heterogeneity among studies on early intervention. Finally, by implementing multimodal neural and behavioral monitoring in the NICU (e.g., smart video motion tracking, safe dry electrode EEG systems, etc.), the mediating impact of interventions on neural and behavioral activity can be better studied. In the age of innovative safe biosensors and big data analysis, adding quantitative unobtrusive neurobehavioral monitoring could determine the mediating effect of the studied interventions to make the NICU of the future into an evidence-based neurodevelopmental care unit.
Conclusion
To our knowledge, this is the first systematic review of the effects of multiple modes of sensory-based interventions in the NICU on neonatal brain development after preterm birth. Although reviewed RCTs present initial evidence on the impact of these interventions for neonatal brain development, it is not yet possible to suggest clear guidelines for clinical practice. In line with previous studies, we agree that sensory-based interventions should always be introduced in combination with expert opinion, parental values, and detailed attention to infants’ behavior. Considering the known relevance of sensory experience and exposure in critical periods of development, further research in this field is warranted. Further RCTs of sensory-based interventions in the NICU, addressing the aforementioned methodological challenges, are needed for the design of evidence-based recommendations for clinical practitioners.
References
Liu, L. et al. Global, regional, and national causes of under-5 mortality in 2000–15: an updated systematic analysis with implications for the Sustainable Development Goals. Lancet 388, 3027–3035 (2016).
Graven, S. N. & Browne, J. V. Auditory development in the fetus and infant. Newborn Infant Nurs. Rev. https://doi.org/10.1053/j.nainr.2008.10.010 (2008).
Keunen, K. et al. Brain tissue volumes in preterm infants: prematurity, perinatal risk factors and neurodevelopmental outcome: a systematic review. J. Matern. Neonatal Med. 25, 89–100 (2012).
Smyser, C. D. et al. Resting-state network complexity and magnitude are reduced in prematurely born infants. Cereb. Cortex 26, 322–333 (2016).
Dubois, J. et al. Mapping the early cortical folding process in the preterm newborn brain. Cereb. Cortex 18, 1444–1454 (2008).
Johnson, S. et al. Autism spectrum disorders in extremely preterm children. J. Pediatr. 156, 525–531.e2 (2010).
Montagna, A. & Nosarti, C. Socio-emotional development following very preterm birth: pathways to psychopathology. Front. Psychol. 7, 80 (2016).
Stene-larsen, K., Lang, A. M., Landolt, M. A., Latal, B. & Vollrath, M. E. Emotional and behavioral problems in late preterm and early term births: outcomes at child age 36 months. BMC Pediatr. https://doi.org/10.1186/s12887-016-0746-z, 1–7 (2016).
Pierrat, V. et al. Neurodevelopmental outcome at 2 years for preterm children born at 22 to 34 weeks’ gestation in France in 2011: EPIPAGE-2 cohort study. BMJ 358, j3448 (2017).
Volpe, J. J. Brain injury in premature infants: a complex amalgam of destructive and developmental disturbances. Lancet Neurol. 8, 110–124 (2009).
Rees, S. & Inder, T. Fetal and neonatal origins of altered brain development. Early Hum. Dev. 81, 753–761 (2005).
Keunen, K. et al. Brain volumes at term-equivalent age in preterm infants: imaging biomarkers for neurodevelopmental outcome through early school age. J. Pediatr. 172, 88–95 (2016).
Dubois, J. et al. Primary cortical folding in the human newborn: an early marker of later functional development. Brain 131, 2028–2041 (2008).
Ment, L. R., Hirtz, D. & Hüppi, P. S. Imaging biomarkers of outcome in the developing preterm brain. Lancet Neurol. 8, 1042–1055 (2009).
Marlow, N. et al. No change in neurodevelopment at 11 years after extremely preterm birth. Arch. Dis. Child. Fetal Neonatal Ed. https://doi.org/10.1136/archdischild-2020-320650 (2021)
Alterman, N. et al. Gestational age at birth and child special educational needs: a UK representative birth cohort study. Arch. Dis. Child. Fetal Neonatal Ed. https://doi.org/10.1136/archdischild-2020-320213 (2021)
Bouyssi-Kobar, M. et al. Third trimester brain growth in preterm infants compared with in utero healthy fetuses. Pediatrics 138, e20161640 (2016).
Volpe, J. J. Dysmaturation of premature brain: importance, cellular mechanisms, and potential interventions. Pediatr. Neurol. 95, 42–66 (2019).
Coviello, C. et al. Effects of early nutrition and growth on brain volumes, white matter microstructure and neurodevelopmental outcome in preterm newborns. Pediatr. Res. https://doi.org/10.1038/pr.2017.227, 1–32 (2017)
Kersbergen, K. J. et al. Longitudinal regional brain development and clinical risk factors in extremely preterm infants. J. Pediatr. https://doi.org/10.1016/j.jpeds.2016.08.024 (2016).
Kersbergen, K. J. et al. Relation between clinical risk factors, early cortical changes, and neurodevelopmental outcome in preterm infants. Neuroimage 142, 301–310 (2016).
Graven, S. N. & Browne, J. V. Sensory development in the fetus, neonate, and infant: introduction and overview. Newborn Infant Nurs. Rev. 8, 169–172 (2008).
Als, H., Duffy, F. & McAnulty, G. Early experience alters brain function and structure. Pediatrics 113, 846 (2004).
Lickliter, R. The integrated development of sensory organization. Clin. Perinatol. 38, 591–603 (2011).
Lickliter, R. Atypical perinatal sensory stimulation and early perceptual development: insights from developmental psychobiology. J. Perinatol. 20, S45–S54 (2000).
Kramaric, K. et al. The effect of ambient noise in the NICU on cerebral oxygenation in preterm neonates on high fl ow oxygen therapy. Signa Vitae 13, 52–56 (2017).
Bartocci, M. et al. Cerebral hemodynamic response to unpleasant odors in the preterm newborn measured by near-infrared spectroscopy. Pediatr. Res. 50, 324–330 (2001).
Gressens, P., Rogido, M., Paindaveine, B. & Sola, A. The impact of neonatal intensive care practices on the developing brain. J. Pediatr. 140, 646–653 (2002).
Pineda, R. G. et al. Alterations in brain structure and neurodevelopmental outcome in preterm infants hospitalized in different neonatal intensive care unit environments. J. Pediatr. 164, 52–60 (2014).
Smith, G. C. et al. NICU stress is associated with brain development in preterm infants. Ann. Neurol. 70, 541–549 (2011).
Aj, S. & Pinelli, J. Developmental care for promoting development and preventing morbidity in preterm infants (Review). Cochrane Database Syst. Rev. https://doi.org/10.1002/14651858.CD001814.pub2.www.cochranelibrary.com (2009).
Lubbe, W., Van Der Walt, C. S. J. & Klopper, H. C. Integrative literature review defining evidence-based neurodevelopmental supportive care of the preterm infant. J. Perinat. Neonat. Nurs. 26, 251–259 (2012).
Conde-Aguedelo, A. & Díaz-rossello, J. L. Kangaroo mother care to reduce morbidity and mortality in low birthweight infants (Review). Cochrane Database Syst. Rev., 1–149 https://doi.org/10.1002/14651858.CD002771.pub4.www.cochranelibrary.com (2016)
Morag, I. & Ohlsson, A. Cycled light in the intensive care unit for preterm and low birth weight infants. Cochrane Database Syst. Rev. https://doi.org/10.1002/14651858.CD006982.pub4.www.cochranelibrary.com (2016).
Pineda, R. et al. Enhancing sensory experiences for very preterm infants in the NICU: an integrative review. J. Perinatol. 16, 1–10 (2016).
van den Hoogen, A. et al. How to improve sleep in a neonatal intensive care unit: a systematic review. Early Hum. Dev. 113, 78–86 (2017).
Anderson, D. E. & Patel, A. D. Infants born preterm, stress, and neurodevelopment in the neonatal intensive care unit: might music have an impact? Dev. Med. Child Neurol. https://doi.org/10.1111/dmcn.13663 (2018).
Fumagalli, M. et al. From early stress to 12-month development in very preterm infants: preliminary findings on epigenetic mechanisms and brain growth. PLoS Med. 13, 1 (2018).
Montirosso, R. & Provenzi, L. Implications of epigenetics and stress regulation on research and developmental care of preterm infants. J. Obstet. Gynecol. Neonatal Nurs. 44, 174–182 (2015).
Kentner, A. C., Scalia, S., Shin, J., Migliore, M. M. & Rondon-Ortiz, A. N. Targeted sensory enrichment interventions protect against behavioral and neuroendocrine consequences of early life stress. Psychoneuroendocrinology 98, 74–85 (2018).
Als, H. et al. NIDCAP improves brain function and structure in preterm infants with severe intrauterine growth restriction. J. Perinatol. 32, 797–803 (2012).
Moher, D. et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 6, 7 (2009).
De Bijl-Marcus, K. A., Brouwer, A. J., De Vries, L. S. & Van Wezel-Meijler, G. The effect of head positioning and head tilting on the incidence of intraventricular hemorrhage in very preterm infants: a systematic review. Neonatology 111, 267–279 (2017).
Romantsik, O., Calevo, M. G. & Bruschettini, M. Head midline position for preventing the occurrence or extension of germinal matrix-intraventricular hemorrhage in preterm infants. Cochrane Database Syst. Rev. 7, 7 (2017).
de Bijl-Marcus, K., Brouwer, A. J., De Vries, L. S., Groenendaal, F. & van Wezel-Meijler, G. Neonatal care bundles are associated with a reduction in the incidence of intraventricular haemorrhage in preterm infants: a multicentre cohort study. Arch. Dis. Child. Fetal Neonatal Ed. 105, 419–424 (2020).
Ohlsson, A. & Jacobs, S. E. NIDCAP: a systematic review and meta-analyses of randomized controlled trials. Pediatrics 131, e881–e893 (2013).
Sterne, J. et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366, l4898 (2019).
Welch, M. G. et al. Electroencephalographic activity of preterm infants is increased by family nurture intervention: a randomized controlled trial in the NICU. Clin. Neurophysiol. 125, 675–684 (2014).
Myers, M. M. et al. Family nurture intervention in preterm infants alters frontal cortical functional connectivity assessed by EEG coherence. Acta Paediatr. 104, 670–677 (2015).
Webb, A. R. et al. Mother’ s voice and heartbeat sounds elicit auditory plasticity in the human brain before full gestation. Proc. Natl Acad Sci. USA 112, 3152–3157 (2015).
Ludington-Hoe, S. M. Neurophysiologic assessment of neonatal sleep organization: preliminary results of a randomized, controlled trial of skin contact with preterm infants. Pediatrics 117, e909–e923 (2006).
Guzzetta, A. et al. The effects of preterm infant massage on brain electrical activity. Dev. Med. Child Neurol. 53, 46–51 (2011).
Song, D. et al. Modulation of EEG spectral edge frequency during patterned pneumatic oral stimulation in preterm infants. Pediatr. Res. 75, 85–92 (2014).
Barlow, S. M. et al. Amplitude-integrated EEG and range-EEG modulation associated with pneumatic orocutaneous stimulation in preterm infants. J. Perinatol. 34, 213–219 (2014).
Lordier, L. et al. Music in premature infants enhances high-level cognitive brain networks. Proc. Natl Acad. Sci. USA 116, 12103–12108 (2019).
Lordier, L. et al. Music processing in preterm and full-term newborns: a psychophysiological interaction (PPI) approach in neonatal fMRI. Neuroimage 185, 857–864 (2019).
Welch, M. G. et al. Family nurture intervention in preterm infants increases early development of cortical activity and independence of regional power trajectories. Acta Paediatr. Int. J. Paediatr. 106, 1952–1960 (2017).
Haslbeck, F. B. et al. Creative music therapy to promote brain function and brain structure in preterm infants: a randomized controlled pilot study. Neuroimage. Clin. 25, 102171 (2020).
Sa de Almeida, J. et al. Music enhances structural maturation of emotional processing neural pathways in very preterm infants. Neuroimage 207, 116391 (2020).
Doheny, L. et al. Exposure to biological maternal sounds improves cardiorespiratory regulation in extremely preterm infants. J. Matern. Neonatal Med. 25, 1591–1594 (2012).
Parga, J. J., Harper, R. M., Karp, H., Kesavan, K. & Zeltzer, L. Low frequency rhythmic womb-like sounds modify autonomic activity in premature neonates. J. Investig. Med. 64, 159–160 (2016).
Sajjadian, N., Mohammadzadeh, M. & Alizadeh, P. Positive effects of low intensity recorded maternal voice on physiologic reactions in premature infants. Infant Behav. Dev. 46, 59–66 (2017).
Filippa, M. et al. Systematic review of maternal voice interventions demonstrates increased stability in preterm infants. Acta Paediatr. 106, 1220–1229 (2017).
Loewy, J., Stewart, K., Dassler, A. M., Telsey, A. & Homel, P. The effects of music therapy on vital signs, feeding, and sleep in premature infants. Pediatrics 131, 902–918 (2013).
Wirth, L. et al. Effects of standardized acoustic stimulation in premature infants: a randomized controlled trial. J. Perinatol. 36, 486–492 (2016).
Olischar, M., Shoemark, H., Holton, T., Weninger, M. & Hunt, R. W. The influence of music on aEEG activity in neurologically healthy newborns ‡ 32 weeks’ gestational age. Acta Paediatr. 670–675 https://doi.org/10.1111/j.1651-2227.2011.02171.x (2011).
Shoemark, H., Hanson-Abromeit, D. & Stewart, L. Constructing optimal experience for the hospitalized newborn through neuro-based music therapy. Front. Hum. Neurosci. 9, 1–5 (2015).
Keunen, K., Counsell, S. J. & Benders, M. J. The emergence of functional architecture during early brain development. Neuroimage, 1–13 https://doi.org/10.1016/j.neuroimage.2017.01.047 (2017)
Hooks, B. M. & Chen, C. Critical periods in the visual system: changing views for a model of experience-dependent plasticity. Neuron 56, 312–326 (2007).
Tortora, D. et al. Early pain exposure influences functional brain connectivity in very preterm neonates. Front. Neurosci. 13, 1–11 (2019).
Gursul, D. et al. Stroking modulates noxious-evoked brain activity in human infants. Curr. Biol. 28, R1380–R1381 (2018).
van Noort-van der Spek, I. L., Franken, M.-C. J. P. & Weisglas-Kuperus, N. Language functions in preterm-born children: a systematic review and meta-analysis. Pediatrics 129, 745–754 (2012).
Vandormael, C., Schoenhals, L., Hüppi, P. S., Filippa, M. & Tolsa, C. B. Language in preterm born children: atypical development and effects of early interventions on neuroplasticity. Neural Plast. 2019, 6873270 (2019).
Kuhn, P. et al. Infants born very preterm react to variations of the acoustic environment in their incubator from a minimum signal-to-noise ratio threshold of 5 to 10 dBA. Pediatr. Res. 71, 386–392 (2012).
Roffwarg, H. P., Muzio, J. N. & Dement, W. C. Ontogenetic development of the human sleep-dream cycle. Science 152, 604 LP–604619 (1966).
Blumberg, M. S., Lesku, J. A., Libourel, P., Schmidt, M. H. & Rattenborg, N. C. What is REM sleep? Curr. Biol. 30, R38–R49 (2020).
Cao, J., Herman, A. B., West, G. B., Poe, G. & Savage, V. M. Unraveling why we sleep: quantitative analysis reveals abrupt transition from neural reorganization to repair in early development. Sci. Adv. 6, eaba0398 (2020).
Tokariev, A. et al. Large-scale brain modes reorganize between infant sleep states and carry prognostic information for preterms. Nat. Commun. 10, 2619 (2019).
Bennet, L., Walker, D. W. & Horne, R. S. C. Waking up too early—the consequences of preterm birth on sleep development. J. Physiol. 596.23, 5687–5708 (2018).
De Wel, O. et al. Relationship between early functional and structural brain developments and brain injury in preterm infants. Cerebellum https://doi.org/10.1007/s12311-021-01232-z (2021).
Ellingsen, D. M. et al. In touch with your emotions: oxytocin and touch change social impressions while others’ facial expressions can alter touch. Psychoneuroendocrinology 39, 11–20 (2014).
Suvilehto, J. T. et al. Topography of social touching depends on emotional bonds between humans. Proc. Natl Acad. Sci. USA 112, 45 (2015).
Herstentein, M., Verkamp, J. M., Kerestes, A. M. & Holmes, R. M. The communicative functions of touch in humans, nonhuman primates, and rats: a review and synthesis of the empirical research. Genet. Soc. Gen. Psychol. Monogr. 132, 5–94 (2006).
Walker, S. C. & McGlone, F. P. The social brain: neurobiological basis of affiliative behaviours and psychological well-being. Neuropeptides 47, 379–393 (2013).
Morrison, I., Löken, L. & Olausson, H. The skin as a social organ. Exp. Brain Res. 204, 305–314 (2010).
Brauer, J., Xiao, Y., Poulain, T., Friederici, A. D. & Schirmer, A. Frequency of maternal touch predicts resting activity and connectivity of the developing social brain. Cereb. Cortex 26, 3544–3552 (2016).
Smith, J. R. Comforting touch in the very preterm hospitalized infant: an integrative review. Adv. Neonatal Care 12, 349–365 (2012).
Boundy, E. O. et al. Kangaroo mother care and neonatal outcomes: a meta-analysis. Pediatrics 137, x–16 (2016).
Feldman, R., Weller, A., Sirota, L. & Eidelman, A. I. Skin-to-skin contact (kangaroo care) promotes self-regulation in premature infants: sleep–wake cyclicity, arousal modulation, and sustained exploration. Dev. Psychol. 38, 194–207 (2002).
Korraa, A. A., El Nagger, A. A. I., Mohamed, R. A. E.-S. & Helmy, N. M. Impact of kangaroo mother care on cerebral blood flow of preterm infants. Ital. J. Pediatr. 40, 83 (2014).
Maitre, N. L. et al. The dual nature of early-life experience on somatosensory processing in the human infant brain. Curr. Biol. 27, 1048–1054 (2017).
Guzzetta, A. et al. Massage accelerates brain development and the maturation of visual function. J. Neurosci. 29, 6042–6051 (2009).
Meijer, E. et al. Functional connectivity in preterm infants derived from EEG coherence analysis. Eur. J. Paediatr. Neurol. 18, 780–789 (2014).
Scher, M. S., Steppe, D. A. & Banks, D. L. Prediction of lower developmental performances of healthy neonates by neonatal EEG-sleep measures. Pediatr. Neurol. 14, 137–144 (1996).
Gou, Z., Choudhury, N. & Benasich, A. A. Resting frontal gamma power at 16, 24 and 36 months predicts individual differences in language and cognition at 4 and 5 years. Behav. Brain Res. 220, 263–270 (2011).
Mundy, P., Mundy, P., Card, J. & Fox, N. EEG correlates of the development of infant joint attention skills. Dev. Psychobiol. 36, 325–338 (2016).
Weaver, I. C. G. et al. Epigenetic programming by maternal behavior. Nat. Neurosci. 7, 847–854 (2004).
Champagne, D. L. et al. Maternal care and hippocampal plasticity: evidence for experience-dependent structural plasticity, altered synaptic functioning, and differential responsiveness to glucocorticoids and stress. J. Neurosci. 28, 6037–6045 (2008).
Kommers, D., Oei, G., Chen, W., Feijs, L. & Bambang Oetomo, S. Suboptimal bonding impairs hormonal, epigenetic and neuronal development in preterm infants, but these impairments can be reversed. Acta Paediatr. 105, 738–751 (2016).
Gunnar, M. R., Hostinar, C. E., Sanchez, M. M., Tottenham, N. & Sullivan, R. M. Parental buffering of fear and stress neurobiology: Reviewing parallels across rodent, monkey, and human models. Soc. Neurosci. 10, 474–478 (2015).
Reynolds, L. C. et al. Parental presence and holding in the neonatal intensive care unit and associations with early neurobehavior. J. Perinatol. 33, 636–641 (2013).
Milgrom, J. et al. Early sensitivity training for parents of preterm infants: impact on the developing brain. Pediatr. Res. 67, 330–335 (2010).
Benders, M. J. et al. Early brain activity relates to subsequent brain growth in premature infants. Cereb. Cortex 25, 3014–3024 (2015).
Tataranno, M. L. et al. Changes in brain morphology and microstructure in relation to early brain activity in extremely preterm infants. Pediatr. Res. 83, 834–842 (2018).
Funding
This research did not receive any financial support from funding agencies in the public, commercial, or not-for-profit sectors. The publication was made possible by the ESPR.
Author information
Authors and Affiliations
Contributions
Substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data: M.I.B., J.D., T.M.d.J., and A.v.d.H. Drafting the article or revising it critically for important intellectual content; and final approval of the version to be published: all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent statement
Patient consent was not required.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Beltrán, M.I., Dudink, J., de Jong, T.M. et al. Sensory-based interventions in the NICU: systematic review of effects on preterm brain development. Pediatr Res 92, 47–60 (2022). https://doi.org/10.1038/s41390-021-01718-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-021-01718-w
This article is cited by
-
Multisensory stimulation bundles on sleep and neurobehavioral development in the first year after birth in very preterm infants: a randomized crossover controlled study protocol
Trials (2023)
-
Intelligent wearable allows out-of-the-lab tracking of developing motor abilities in infants
Communications Medicine (2022)