Abstract
Background
Perinatal events may alter psychosexual development. We aimed to assess whether a preterm birth at very low birth weight (VLBW; <1500 g) or antenatal synthetic glucocorticoids (sGC) given to the mother are associated with altered sex-typical behavior in childhood.
Methods
Sex-typical behavior was assessed using the Pre-school Activities Inventory (PSAI) at the mean age of 4.9 years (SD 1.6) in 879 children, of whom 143 were preterm with VLBW (PT <1500 g, all exposed to sGC), 282 were preterm with birth weight ≥1500 g (PT ≥1500 g, 171 exposed to sGC), and 454 were full term (FT, 166 exposed to sGC).
Results
Antenatal sGC was not associated with PSAI scores in either sex. PT <1500 g boys had less male-typical PSAI scores than other boys, even in multivariate model adjusting for age, maternal age, antenatal sGC, number of brothers and sisters, and motor or cognitive impairment. PT <1500 g girls had less female-typical PSAI scores than other girls in the multivariate model. The effect size was small (d = 0.03) for both sexes.
Conclusions
Preterm birth with VLBW is associated with reduced sex-typical behavior in childhood, which is in line with the previous data indicating altered psychosexual development in adults born preterm. Mechanisms underlying these observations are not fully understood.
Impact
-
Preterm birth is associated with reduced rates of marriage and reproduction in adulthood, but sex-typical behavior in children born preterm has not been studied before.
-
The results of this study indicate that preterm birth with very low birth weight <1500 g is associated with reduced sex-typical behavior in childhood in both sexes.
-
These observations are in line with the previous data indicating altered psychosexual development in adults born preterm. Mechanisms underlying these observations are not fully understood and require further studies.
Similar content being viewed by others
Introduction
Reduced rates of marriage and reproduction have been reported among adults born preterm, even in the absence of major disabilities.1 In several studies, preterm-born young adults have reported having challenges in their social relationships: they consider themselves less attractive, have less confidence in romantic relationships, are less likely to cohabit with a romantic partner, and are less sexually active than their full-term (FT) born peers.1,2,3,4,5 Most of these studies were performed on adults born before 32 weeks of gestation or with very low birth weight (VLBW; <1500 g), but similar findings have been reported in adults born with birth weight ≥1500 g and gestational age between 32 and 36 weeks.2 Moreover, in a recent study, a higher proportion of preterm-born young adults with extremely low birth weight (ELBW; <1000 g) identified themselves as nonheterosexual.6 These findings could indicate that prematurity or ELBW/VLBW is associated with altered psychosexual development.
Sexual orientation and core gender identity are among the human traits that have the highest sexual dimorphism, although smaller-scale differences between the sexes can be seen in many behavioral aspects.7 Some of these differences in sex-typical behavior, like toy and activity preferences, are already evident in early childhood.8 Preterm birth disrupts the normal intrauterine hormone milieu, and furthermore, most preterm infants are also exposed antenatally to synthetic glucocorticoids (sGC), given to the mother at the threat of premature labor. In animal studies, antenatal exposure to sGC has been associated with alterations in reproductive functions and sexual behavior.9,10,11,12 If prematurity and/or ELBW/VLBW is associated with a major alteration in psychosexual development, it can be hypothesized that these children already present differences in sex-typical behavior in childhood. However, to our knowledge, sex-typical behavior in children born preterm or at ELBW/VLBW has not been studied as of yet.
We hypothesized that children born preterm at VLBW show altered sex-typical behavior in childhood and that antenatal exposure to sGC also modulates sex-typical behavior in humans. This study tested these hypotheses by measuring sex-typical behavior in a large cohort of preterm and FT children, both with and without antenatal exposure to sGC.
Methods
The present cohort study was conducted among preterm (gestational age at birth <37 weeks) and FT children (gestational age at birth ≥37 weeks). The children were born during a 6-year period from 2003 to 2008 in Kuopio University Hospital, Finland and aged from 1.5 to 8 years at assessment. To identify eligible children, a hospital-based electronic birth record was used that includes data on the mother, pregnancy, exposure to antenatal sGC, and the newborn.
First, we aimed at recruiting all children who were exposed to antenatal sGC within three cohorts: children born preterm with VLBW at <1500 g (later PT <1500 g), children born preterm at birth weight ≥1500 g (later PT ≥1500 g), and children born FT at birth weight ≥1500 g (later FT). Second, we aimed at recruiting one or two randomly selected controls for each sGC-exposed child. The controls were not exposed to sGC, and they were matched for sex, gestational age (+/−6 days), and date of birth (+/−3 days).
The coverage of antenatal sGC had been virtually 100% at preterm birth before 35 gestation weeks; therefore, all PT <1500 g children (n = 263) were exposed to sGC, and no controls could be found for this group. Furthermore, 281 PT ≥1500 g children had been exposed to sGC, and for them, only 223 non-exposed matched controls could be identified. Additionally, there were 270 FT children who were exposed to sGC but then born FT, and 532 matched controls were identified for them. The living status and current address of all the 1569 eligible children were retrieved from the Finnish Population Registry Center; altogether 1500 children were alive and living with the biological parents.
The Pre-school Activities Inventory (PSAI) is a 24-item standardized questionnaire designed to discriminate gender-typical behavior within and between the sexes.13 The questions address three aspects of behavior: play with sex-typical toys (e.g., dolls, cars), engagement in sex-typical activities (e.g., ballgames, playing at cooking/cleaning), and sex-typical child characteristics (e.g., interest in snakes/spiders/insects, liking pretty things). Answers are given on a 5-point Likert-type scale from never to very often. Scores are combined so that a higher score indicates more male-typical/less female-typical behavior, with a theoretical scale of the score from −4.55 to 101.05. The test was initially standardized on >2000 children and validated against teacher’s ratings, and it has been used in various studies to assess the sex-typical behavior of mainly pre-school-aged children.14,15
A letter was mailed to parents that included a Finnish translation of the PSAI questionnaire and a questionnaire on basic demographic data. Both parents signed the informed consent. Among those who responded, data on motor or cognitive impairment were obtained from the hospital database (ICD10 codes for cerebral palsy, G80–G83, and for intellectual disability, F70–F79). The Ethics committee of the Northern-Savo Health Care District approved the study (decision number 47//2010).
Independent-samples t tests were used to examine differences in continuous variables between the groups; and chi-square tests were used to examine the proportions of single, twin, and triplet pregnancies between groups. Effect size d was calculated by dividing the difference of means by the pooled standard deviation. Because of the large numbers of twins and triplets in the study, linear mixed models were used as multivariate models assessing the association of different variables with the PSAI score. In these models, subject and twins and triplets were considered random effects, allowing for potential clustering effects. In addition, the following covariates were included in all the models as fixed effects: age, exposure to antenatal glucocorticoid, number of brothers, number of sisters, motor or cognitive impairment, and maternal age at delivery. The corrected age (i.e., the child’s chronological age minus the months the child was born preterm) was used for PT children up to the corrected age of 2 years. A separate analysis was performed to describe the PSAI scores among the ELBW (<1000 g) children.
Furthermore, we performed a separate age-adjusted linear mixed model to test the effect of birth weight as a continuous variable on the PSAI score combining all groups of boys and all groups of girls.
SPSS software (version 25.0; SPSS Inc.) was used for all statistical analyses.
Results
A total of 931 responses were received out of the 1500 postal requests for PSAIs, giving a response rate of 62.1% (68.1% in the PT <1500 g group; 57.6% in the PT ≥1500 g group; 56.8% in the FT group; p = 0.03; Fig. 1). The PSAI could not be scored in 52 children because of one or more missing responses. Thus, PSAI scores were available for 449 boys and 430 girls.
In total, 480 (54.7%) subjects were exposed to antenatal sGC, including all the children in the PT <1500 g group. Among the PT ≥1500 g girls and boys, those exposed to sGC were born at a significantly lower gestational age and weight than the non-exposed subjects (Tables 1 and 2). Among the PT ≥1500 g girls and boys, there were more twins or triplets in the sGC group. Among the PT ≥1500 g girls, those not exposed to sGC were older at PSAI assessment than those exposed (5.4 vs 4.9 years). Among the FT girls and boys, there were no differences in background factors between those exposed and those not exposed to sGC (Tables 1 and 2). Ten children (five boys in the PT <1500 g group, two boys in the PT >1500 g group, and one girl in the PT <1500 g, PT ≥1500 g, and FT groups) had been diagnosed with motor or a mild cognitive impairment.
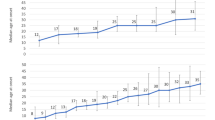
The mean PSAI score was 68.2 (SD 10.3) among all the boys and 28.4 (SD 10.9) among all the girls (p < 0.001). The PSAI score became more female-typical in girls and more male-typical in boys as their age increased (Fig. 2). The effect of age at assessment was statistically significant for boys but not for girls in the multivariate model (parameter estimate β = 1.8, p < 0.001 for boys; β = −0.48, p = 0.16 for girls; Table 3).
The effect of antenatal sGC on PSAI scores was assessed only in PT ≥1500 g and FT children, as all children born PT <1500 g were exposed to antenatal sGC. In the PT ≥1500 g and FT boys with antenatal sGC (n = 171), the mean PSAI score was 68.5 (SD 10.6), compared to 68.6 (SD 9.9) in boys without exposure to antenatal sGC (n = 213; p = 0.90, t test). In the PT ≥1500 g and FT girls, the mean PSAI score was 27.5 (SD 11.3) in the sGC group (n = 166) and 28.2 (SD 10.0) in the unexposed group (n = 186; p = 0.58). No effect of antenatal sGC was observed in the multivariate linear mixed model (β = 0.3, 95% confidence interval (CI), −1.7 to 2.3, p = 0.75 for boys; β = 1.3, 95% CI, −1.1 to 3.6, p = 0.29 for girls; Table 3).
Since exposure to antenatal sGC seemed not to modulate PSAI scores, at least in PT ≥1500 g and FT children, the univariate t tests between the PT <1500 g, PT ≥1500 g, and FT groups were performed after combining the results of children exposed and not exposed to sGC (separately for boys and girls) The PT <1500 g boys showed significantly less male-typical behavior than other boys (Fig. 3). The difference between PSAI in PT <1500 g boys (65.7) and other boys (PSAI 68.6) was small (effect size d = 0.3) but statistically significant (p = 0.04, t test). This less-masculine PSAI in PT <1500 g boys remained significant in the multivariate model, adjusting for age, maternal age at delivery, exposure to antenatal sGC, motor or cognitive impairment, and number of brothers and number of sisters (β = −3.0, 95% CI, −5.8 to −0.2; p = 0.04; Table 3). Among the boys born preterm at <1000 g (n = 23), the mean PSAI score was 65.6 (SD 9.9).
Like the PT <1500 g boys, the PT <1500 g girls displayed a tendency toward reduced sex-typical behavior, showing less female-typical PSAI scores than the other girls with a mean PSAI score of 30.8 (SD 12.0; Fig. 3). Compared to the other girls (PT ≥1500 g and FT girls combined, PSAI 27.9), the difference was again small (effect size d = 0.3) but significant (p = 0.03, t test). In the multivariate linear mixed model for girls, the parameter estimate associated with the PT <1500 g birth was 3.5 (95% CI, 0.5–6.5; p = 0.02; Table 3). This observation was primarily due to the high mean PSAI score of 35.0 (SD 12.5) among girls born preterm at birth weight <1000 g (n = 28).
In the multivariate linear mixed models that included all boys, other significant factors associated with PSAI scores were the child’s age at evaluation and the maternal age at delivery; the PSAI score became more male-typical with increasing age and with increased maternal age (Table 3). In the multivariate model for girls, none of the other factors was significantly associated with the PSAI score.
In a separate analysis, we also tested the effect of birth weight as a continuous variable on the PSAI score across all groups. In this age-adjusted linear mixed model that included all the children in the study, birth weight was significantly associated with more sex-typical PSAI score both in boys (β per 1000 g = 0.96, 95% CI, 0.04 to 1.8; p = 0.04) and in girls (β per 1000 g = −1.2, 95% CI, −2.3 to −0.84; p = 0.04).
Discussion
In this cohort study of 879 children aged 1.7–8.1 years, prematurity with VLBW <1500 g was associated with less-masculine PSAI scores in boys and with less-feminine PSAI scores in girls. In girls, this tendency was most prominent in children born preterm at birth weight <1000 g. In both sexes, the association of VLBW and reduced sex-typical behavior was small but remained significant after adjusting for multiple potential confounding factors. Exposure to antenatal sGC was not associated with altered sex-typical behavior in either sex.
To our knowledge, this is the first study to demonstrate reduced sex-typical behavior in children born PT at VLBW. Birth weight and gestational age both reflect developmental immaturity, and VLBW children can be seen as a special, well-documented group of the most immature newborns. However, birth weight and gestational age correlate strongly, and their effects are difficult to distinguish from each other, as most VLBW children are also very preterm. Birth weight per se is a more precise variable than gestational length, and due to the retrospective nature of the study, we chose to use birth weight in the analysis. In our study, it is not possible to say which aspect of immaturity is more associated with the noted effects.
Almost all very preterm infants are also exposed to antenatal sGC, representing one more variable whose effect is hard to distinguish from other aspects of prematurity. We tried to overcome this by also including a large number of infants who were exposed to sGC at the threat of prematurity but whose gestation then continued close to or completely to a FT delivery. The benefits of sGC are indisputable in preterm infants; however, many newborns are also exposed to sGC “in vain” (assessed in hindsight), due to imminent prematurity followed by birth at term.
In theory, sGC can have long-term effects of sex-typical behavior, as demonstrated by various animal studies.9,12 In humans, dexamethasone treatment started at gestational weeks 6–7 has been associated with reduced male-typical behavior in school-aged boys, when it was used to prevent virilization of female fetuses at risk of congenital adrenal hyperplasia before the sex of the fetus was known.16 On the other hand, gender development, sexual orientation, and sex-specific cognitive functioning were similar in individuals aged 20–22 years who were either exposed to antenatal sGC between 26 and 32 gestation weeks or not exposed.17 In our study, the exposure to antenatal betamethasone (intramuscular dose of 12 mg twice for the pregnant mother) between 23 and 34 gestation weeks was not associated with alterations in sex-typical behavior. Thus, although sGC in early pregnancy can have a de-masculinizing effect on male fetuses,16 our data, like those of Dessens et al.,17 suggest that antenatal sGC at threat of prematurity after mid-gestation are not likely to have a major effect on neurobehavioral sexual differentiation. The timing of the exposure might play a significant role as there is a masculinization programming window established in rats and assumed in humans between 8 and 14 weeks of gestation.18 During this window, androgens program the male reproductive tract, and theoretically, exposure to antenatal sGC could disrupt this process, whereas exposure later in gestation might not be as harmful.
The reproductive rate and the marriage rate in both men and women born preterm is lower than in the general population.19,20,21 Adults born at VLBW are also reported to have lower sexual activity in some5,22 though not all studies.23 Low birth weight has been also associated with homosexual tendencies in men, although in one study this was evident only if the subject had older brothers.24 The possible mechanisms for higher gender-atypical behavior in VLBW subjects remain unclear; however, it is known that VLBW children differ from the general population in many ways. VLBW infants, particularly those with ELBW, have high somatic morbidity and higher rates of behavioral and neurodevelopmental problems as compared to the general population.3,19,25 These could in theory lead to lower rates of reproduction, but it is hard to see that this could explain the differences in the sex-typical play behavior. There are also known differences in the personality traits among VLBW children, including higher conscientiousness and lower openness to experience.26 In addition, parents of VLBW children27 and preterm children28 are shown to be more controlling. Again, these factors could in theory associate with decreased sexual activity, but a link to altered sex-typical play behavior is not so evident. Furthermore, it must be emphasized that it is currently not known how the sex-typical play behavior translates to the psychosexual aspects in adulthood.
The hormonal factors of perinatal programming might exert long-lasting effects on reproduction as well as on psychosexual development in VLBW children. Prenatal testosterone is known to play a role in the sexual differentiation of the human brain,29,30 and now increasing evidence shows that testosterone in the early postnatal period may have effects as well.31,32,33 Clear differences can be seen in the perinatal hormonal milieu between premature (including VLBW) and FT infants. Premature infants’ exposure to maternal and placental steroids is shorter, and hormonal changes associated with birth take place at an earlier developmental age than in FT infants. For example, after birth there is a transient elevation in testosterone levels resulting from a transient pituitary–gonadal activation, known as minipuberty. In preterm infants, this minipuberty takes place at an earlier developmental age and is more vigorous, manifesting as higher testosterone levels in preterm boys compared to FT boys.34 However, these elevated testosterone levels should theoretically lead to a more masculinized behavior in preterm boys, and thus at least the magnitude of the testosterone surge in infancy does not explain our findings.
Prenatal stress offers one explanation for our results, at least in girls. In rodents, prenatal stress increases masculine-typical behavior in females35 and female-typical behavior in males.36 In human studies, this kind of association has been noted in girls but not significantly in boys.37,38 This effect is believed to be mediated by adrenal androgens.37 It is known that androgen levels are significantly higher in preterm girls than in FT girls during the first months of life.39
In our study, our group of VLBW infants contained many twins and triplets. One possible confounding factor that we could not account for is the sex of the co-twin/co-triplets. However, single/multiple pregnancy per se was not related to PSAI score in the multivariate model.
One limitation of our study is the assessment of sex-typed behavior using a single questionnaire in our study population with a wide age range. However, it has been previously shown that PSAI is a valid instrument in measuring sex-typed behavior in children. In those longitudinal studies, stability of the PSAI test results repeated in individuals at different ages has been high.14,40 Also, PSAI discriminated the sexes the better the older the child was, as in our study there was only minor overlap in the distributions of PSAI scores between boys and girls. To tackle the wide age range of the individuals, the age at assessment was used as a fixed factor in the linear mixed models. Another limitation is the rather low response rate of 62.1%, though this corresponds well to the median response rate of 60% in published mail surveys in medical journals.41 This could lead to bias in the results, however. The response rate was significantly higher in the VLBW group than in the other groups, possibly reflecting a better adherence to health care in this group of children and their families. It is probable that children and families without motor or cognitive impairment were more willing to participate than those with long-term impairment, because the rate of impairment in the final population was low.
In conclusion, our study indicates that prematurity with VLBW is associated with reduced sex-typical behavior in childhood in both sexes. Further studies are needed to verify these findings and to further examine the mechanisms underlying them.
References
Mendonça, M., Bilgin, A. & Wolke, D. Association of preterm birth and low birth weight with romantic partnership, sexual intercourse, and parenthood in adulthood. JAMA Netw. Open 2, e196961 (2019).
Männistö, T. et al. Independent living and romantic relations among young adults born preterm. Pediatrics 135, 290–297 (2015).
Hille, E. T. et al. Behavioural problems in children who weigh 1000 g or less at birth in four countries. Lancet 357, 1641–1643 (2001).
Darlow, B. A., Horwood, L. J., Pere-Bracken, H. M. & Woodward, L. J. Psychosocial outcomes of young adults born very low birth weight. Pediatrics 132, e1521–e1528 (2013).
Kajantie, E. et al. Young adults with very low birth weight: Leaving the parental home and sexual relationships-Helsinki study of very low birth weight adults. Pediatrics 122, e62–e72 (2008).
Saigal, S. et al. Health, wealth, social integration, and sexuality of extremely low-birth-weight prematurely born adults in the fourth decade of life. JAMA Pediatr. 170, 678 (2016).
Hines, M. Gender development and the human brain. Annu. Rev. Neurosci. 34, 69–88 (2011).
Hines, M., Brook, C. & Conway, G. S. Androgen and psychosexual development: core gender identity, sexual orientation, and recalled childhood gender role behavior in women and men with congenital adrenal hyperplasia (CAH). J. Sex. Res. 41, 75–81 (2004).
Piffer, R. C., Garcia, P. C. & Pereira, O. C. M. Adult partner preference and sexual behavior of male rats exposed prenatally to betamethasone. Physiol. Behav. 98, 163–167 (2009).
Dunn, E., Kapoor, A., Leen, J. & Matthews, S. G. Prenatal synthetic glucocorticoid exposure alters hypothalamic-pituitary-adrenal regulation and pregnancy outcomes in mature female guinea pigs. J. Physiol. 588, 887–899 (2010).
Oliveira, M. et al. Programming effects of antenatal corticosteroids exposure in male sexual behavior. J. Sex. Med. 8, 1965–1974 (2011).
Hauser, J. et al. Effects of prenatal dexamethasone treatment on postnatal physical, endocrine, and social development in the common marmoset monkey. Endocrinology 148, 1813–1822 (2007).
Golombok, S. & Rust, J. The measurement of gender role behavior in pre-school children: a research note. J. Child Psychol. Psychiatry 34, 805–811 (1993).
Golombok, S. et al. Developmental trajectories of sex-typed behavior in boys and girls: a longitudinal general population study of children aged 2.5-8 years. Child Dev. 79, 1583–1593 (2008).
Auyeung, B. et al. Fetal testosterone predicts sexually differentiated childhood behavior in girls and in boys. Psychol. Sci. 20, 144–148 (2009).
Hirvikoski, T., Lindholm, T., Lajic, S. & Nordenström, A. Gender role behaviour in prenatally dexamethasone-treated children at risk for congenital adrenal hyperplasia - a pilot study. Acta Paediatr. 100, e112–e119 (2011).
Dessens, A. B., Haas, H. S.-D. & Koppe, J. G. Twenty-year follow-up of antenatal corticosteroid treatment. Pediatrics 105, e77 (2000).
Welsh, M. et al. Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism. J. Clin. Investig. 118, 1479–1490 (2008).
Moster, D., Lie, R. T. & Markestad, T. Long-term medical and social consequences of preterm birth. N. Engl. J. Med. 359, 262–273 (2008).
Ekholm, K., Carstensen, J., Finnström, O. & Sydsjö, G. The probability of giving birth among women who were born preterm or with impaired fetal growth: a Swedish population-based registry study. Am. J. Epidemiol. 161, 725–733 (2005).
Swamy, G. K., Ostbye, T. & Skjaerven, R. Association of preterm birth with long-term survival, reproduction, and next-generation preterm birth. JAMA 299, 1429–1436 (2008).
Hack, M. et al. Outcomes in young adulthood for very-low-birth-weight infants. N. Engl. J. Med. 346, 149–157 (2002).
Cooke, R. W. I. Health, lifestyle, and quality of life for young adults born very preterm. Arch. Dis. Child. 89, 201–206 (2004).
Frisch, M. & Zdravkovic, S. Body size at birth and same-sex marriage in young adulthood. Arch. Sex. Behav. 39, 117–123 (2010).
Saigal, S. et al. Comparison of the health-related quality of life of extremely low birth weight children and a reference group of children at age eight years. J. Pediatr. 125, 418–425 (1994).
Pesonen, A.-K. et al. Personality of young adults born prematurely: the Helsinki study of very low birth weight adults. J. Child Psychol. Psychiatry 49, 609–617 (2008).
Pyhälä, R. et al. Parental bonding after preterm birth: Child and parent perspectives in the Helsinki study of very low birth weight adults. J. Pediatr. 158, 251.e1–256.e1 (2011).
Forcada-Guex, M., Pierrehumbert, B., Borghini, A., Moessinger, A. & Muller-Nix, C. Early dyadic patterns of mother-infant interactions and outcomes of prematurity at 18 months. Pediatrics 118, e107–e114 (2006).
Hines, M. Prenatal endocrine influences on sexual orientation and on sexually differentiated childhood behavior. Front. Neuroendocrinol. 32, 170–182 (2011).
Hines, M. Prenatal testosterone and gender-related behaviour. Eur. J. Endocrinol. 155, S115–S121 (2006).
Saenz, J. & Alexander, G. M. Postnatal testosterone levels and disorder relevant behavior in the second year of life. Biol. Psychol. 94, 152–159 (2013).
Hines, M. et al. The early postnatal period, mini-puberty, provides a window on the role of testosterone in human neurobehavioural development. Curr. Opin. Neurobiol. 38, 69–73 (2016).
Lamminmäki, A. et al. Testosterone measured in infancy predicts subsequent sex-typed behavior in boys and in girls. Horm. Behav. 61, 611–616 (2012).
Kuiri-Hänninen, T. et al. Increased activity of the hypothalamic-pituitary-testicular axis in infancy results in increased androgen action in premature boys. J. Clin. Endocrinol. Metab. 96, 98–105 (2011).
Sachser, N. & Kaiser, S. Prenatal social stress masculinizes the females’ behaviour in guinea pigs. Physiol. Behav. 60, 589–594 (1996).
Ward, I. L. Prenatal stress feminizes and demasculinizes the behavior of males. Science 175, 82–84 (1972).
Barrett, E. S., Redmon, J. B., Wang, C., Sparks, A. & Swan, S. H. Exposure to prenatal life events stress is associated with masculinized play behavior in girls. Neurotoxicology 41, 20–27 (2014).
Hines, M. et al. Prenatal stress and gender role behavior in girls and boys: a longitudinal, population study. Horm. Behav. 42, 126–134 (2002).
Kuiri-Hänninen, T. et al. Transient postnatal secretion of androgen hormones is associated with acne and sebaceous gland hypertrophy in early infancy. J. Clin. Endocrinol. Metab. 98, 199–206 (2013).
Golombok, S., Rust, J., Zervoulis, K., Golding, J. & Hines, M. Continuity in sex-typed behavior from preschool to adolescence: a longitudinal population study of boys and girls aged 3-13 years. Arch. Sex. Behav. 41, 591–597 (2012).
Asch, D. A., Jedrziewski, M. K. & Christakis, N. A. Response rates to mail surveys published in medical journals. J. Clin. Epidemiol. 50, 1129–1136 (1997).
Acknowledgements
The study was supported by the Foundation for Pediatric Research, The Kuopio University Hospital State Research Funding, The Finnish Medical Foundation, and The Päivikki and Sakari Sohlberg Foundation.
Author information
Authors and Affiliations
Contributions
A.L., T.K.-H., and U.S. have all completed the following criteria: substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; drafting the article or revising it critically for important intellectual content; and final approval of the version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Patient consent
Written informed consent was obtained from both parents.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lamminmäki, A., Kuiri-Hänninen, T. & Sankilampi, U. Sex-typical behavior in children born preterm at very low birth weight. Pediatr Res 89, 1765–1770 (2021). https://doi.org/10.1038/s41390-020-01133-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-020-01133-7