Abstract
Background
The contribution of long-term vs. recent-onset obesity to cardiometabolic risk in adolescence remains controversial. Here, we aimed to investigate the association of time of onset and length of obesity with the cardiometabolic profile of adolescence.
Methods
Prospective study in 678 16-year-olds. BMI was measured at birth–1–5–10–16 years and BMI trajectories were interpolated using cubic splines. BMI > 2 SD at <6 years was defined as early obesity. Waist circumference (WC), blood pressure, lipid and glucose profiles were measured at 16 years. A cardiometabolic risk score was computed (MetS_score). According to the BMI trajectory, four groups were defined: participants who were never obese (NOB), participants with obesity during adolescence (recent-onset obese (ROB)), participants who were obese in early childhood but transitioned to normal/overweight as preadolescents (formerly obese (FOB)), and participants who were obese in early childhood and remained obese (persistently obese (POB)).
Results
ROBs and POBs had significantly unhealthier cardiometabolic profile than NOBs. No differences were observed in the cardiometabolic profile of ROBs compared to POBs. Although FOBs had higher WC and MetS_score than NOBs, no differences were found in other biomarkers. FOBs were in healthier cardiometabolic condition than ROBs and POBs.
Conclusions
Both long-term and recent-onset obesity increase the cardiometabolic risk in adolescents.
Similar content being viewed by others
Introduction
Obesity is a major public health concern worldwide, contributing to a decline in healthy years of life years and significant economic costs.1 In Chile, the prevalence of obesity rose from 4.5% in the mid-1980s to 33.3% in 2017.2,3 Currently, about three-quarters of adults are overweight or obese,4 and 51% of preschoolers and first graders have some degree of excess weight.3
Childhood obesity is a significant predictor of the persistence of obesity and the development of cardiometabolic disorders in adulthood, including Metabolic Syndrome (MetS).5,6 Among the critical determinants for the development of long-lasting obesity, early adiposity rebound (AR) has been identified as the most important.7 Under normal circumstances, body mass index (BMI) increases throughout the first year of life and declines to a nadir around the age of 6 years. The second rise in BMI, following this minimum, marks the beginning of the AR.7 Obesity in early childhood, or AR before the age of 6 years, is associated with epigenetic programming of metabolism that allows adaptation to the external environment.8 Likewise, postnatal nutrition has been proposed to have long-term health effects by promoting growth acceleration,9 with AR now occurring at a younger age compared to several decades ago.10 Maternal influences (e.g., diabetes, tobacco use, and excessive weight gain during pregnancy) and the obesogenic environment (e.g., physical inactivity and excessive intake of proinflammatory foods) also contribute to an early onset of obesity and thus to a greater risk of cardiometabolic disorders.11,12
Longitudinal studies in children and adolescents from a variety of countries have found a relationship between early-onset obesity and later risk of obesity.7,13,14 A follow-up study conducted in France was the first to show that obesity before the age of 6 years was associated with higher adiposity levels in adolescence.7 In the UK, the Avondale Longitudinal Study of Parents and Children also found that obesity in the early childhood years was linked to increased BMI and fat mass in adolescence.13 Other studies report associations between early onset of obesity and greater risk of high blood pressure, dyslipidemia, impaired glucose tolerance, and diabetes in childhood, adolescence, or adulthood.15,16,17,18,19 However, not all studies agree. Evidence from cohort studies suggests that early cardiovascular risk in adolescents relates to magnitude and/or duration of obesity, independent of age at obesity onset.20,21,22 Differences in both frequencies of BMI assessment and methodologies to estimate BMI trajectories may explain, in part, disagreement between studies. Here we aimed to investigate the association of time of onset and length of obesity with the cardiometabolic profile of adolescence under the hypothesis that long-term and recent-onset obesity make a similar contribution to the cardiometabolic risk in the adolescent period.
Methods
Study design and population
We studied 678 16–17-year-old adolescents living in Santiago, Chile, of low-to-middle socioeconomic status (SES). Participants are from the Santiago Longitudinal Study and were recruited at 4 months in 1992–1996 from public health-care facilities to participate in research related to nutrition, growth, and development as infants with follow-up at 1, 5, 10, and 16 years.23 To be eligible, they had to be full-term singletons and weighing ≥3 kg at birth, free of acute or chronic health problems, thus none had genetic disorders associated with obesity. It is worth mentioning that participants were born during a dramatic nutritional transition from parents and/or grandparents who were exposed to child undernutrition. At 16 years, they were assessed for the presence of cardiovascular risk factors.24,25 Ethical approval was obtained by the IRBs of the University of Michigan, Institute of Nutrition and Food Technology (University of Chile), and the University of California, San Diego. Informed and written consent was provided according to the norms for Human Experimentation, Code of Ethics of the World Medical Association (Declaration of Helsinki, 1995).
Measurements
Anthropometric assessment
At 1, 5, 10, and 16 years, a research physician used standardized procedures to measure the height (cm) to the nearest 0.1 cm, using a Holtain stadiometer, and weight (kg) to the nearest 0.1 kg, using a scale (Seca 725 and 703, Seca GmbH & Co. Hamburg, Germany). Birth weight was gathered from health records. At 16 years, waist circumference (WC) was measured with a non-elastic flexible tape and recorded to 0.1 cm (Seca 201, Seca GmbH & Co. Hamburg, Germany). Measurements were taken twice, with a third measurement if the difference between the first two exceeded 0.3 kg for weight, 0.5 cm for height, and 1.0 cm for WC. BMI and BMI for age and sex (BMIz) were calculated. Obesity was diagnosed with 2006 and 2007 World Health Organization references according to age: BMIz > 2 SD. Obesity in early childhood was defined as having a BMIz > 2 SD before the age of 6 years. Total fat mass (TFM) was determined on dual X-ray absorptiometry (apparatus: Lunar Prodigy Corp., Madison, WI. Software: Lunar iDXA ENCORE 2011, Version 13.60.033) and total fat-free mass (TFFM) was estimated.
Additional cardiometabolic risk assessment
After 15 min at rest and before the other physical evaluations, systolic and diastolic blood pressures (SBP and DBP, respectively) were measured three times on the non-dominant arm using a standard mercury sphygmomanometer, according to the Second Task Force on Blood Pressure Control in Children (1987); the average value was used for analyses. Fasting serum total glucose, insulin, total cholesterol, triglycerides (TG), high-density lipoprotein (HDL)-cholesterol (HDL-chol), and high-sensitivity C-reactive protein (hs-CRP) were measured after 8–12 h overnight fast. Glucose was measured with an enzymatic colorimetric test (QCA S.A., Amposta, Spain), and radioimmunoassay (Diagnostic Products Corporation, Los Angeles, CA) was used for insulin determination. Cholesterol profile was determined by dry analytical methodology (Vitros®; Ortho Clinical Diagnostics Inc., Raritan, NJ), and hs-CRP was measured with a sensitive latex-based immunoassay. The homeostatic model assessment (HOMA) was used to quantify insulin sensitivity.26 HOMA-insulin resistance (IR) was estimated as the product of fasting glucose (mmol/l) and insulin (μU/ml) divided by the constant 22.5, with values ≥2.6 denoting IR.27 hs-CRP values of >1.0 mg/l were considered low-grade systemic inflammation, according to the AHA/CDC joint Statement on Markers of Inflammation and Cardiovascular Disease.28 To avoid abnormally high hs-CRP levels, participants who were sick (e.g., cold, viral infections, diarrhea, etc.) at least 10 days before the assessment had their appointment rescheduled. Participants with hs-CRP values of >9.0 were excluded from the analysis when inflammation was the outcome (n = 18 or 2.6%). MetS was diagnosed based on the 2009 AHA/NHLBI/IDF Joint Interim Statement. This includes having three of the following cardiometabolic biomarkers: abdominal obesity (WC ≥ 80 and 90 cm in females and males, respectively), high blood arterial pressure (SBP ≥ 130 mm Hg, DBP ≥ 85 mm Hg), hypertriglyceridemia (TG ≥ 150 mg/dl), low HDL (≤50 and ≤40 mg/dl in females and males, respectively), and fasting hyperglycemia (glucose ≥100 mg/dl).29 Additionally, a continuous score representing a composite cardiometabolic risk factor profile was computed according to Gurka et al.30 The equations provide a sex- and race-specific score measure for MetS risk based on standardized and log-transformed values for each component of the MetS. A lower composite metabolic risk score denotes a healthier cardiometabolic profile.
Other covariates
Sex and breastfeeding (BF) length were used as covariates since both have been reported as variables associated with obesity and cardiometabolic risk in this cohort.25,31 Information on breastfeeding duration was gathered in infancy by maternal self-report. For those receiving formula/cow milk supplementation before the age of 4 months, the date of the first bottle was recorded based on maternal recall. For all other infants, this information was asked prospectively, on a weekly basis. Mothers were also asked the date of the last BF. In Chile, at the time of this study, bottle feeding was equivalent to formula feeding, as feeding expressed breast milk by bottle was exceedingly rare. Almost the entire sample was initially breastfed, with only five participants never being breastfed. Using the date of the first bottle of milk/formula and date of the last BF, we created a variable denoting short exclusive BF or BF as the sole source of milk ≤3 months. Other variables, such as diet quality and sedentary behavior were not considered as covariates, because in previous works we found that both loose significance in predictive models of cardiometabolic risk due to the fact that unhealthy diets and reduced time allocation for exercise are highly prevalent in these participants.24,27
Data analysis
All variables were checked for distribution normality (Shapiro–Wilk test) before the analysis. WC, SBP, DBP, HOMA-IR, TG, and HDL were normalized by natural logarithm transformation. Statistical analysis was conducted using transformed data, but untransformed data are presented here. For each participant, we interpolated the BMI trajectory from birth to adolescence using cubic splines. Cubic polynomials is a mathematical numerical analysis to construct new data points within the range of a discrete set of known data points, which allows for modeling of smoothed, non-linear trajectories of BMI across different ages and do not require that all children have the same number of measurements or that the measurements be obtained at the same ages.32,33 By using this method, we fitted the models with data from all assessments (birth, 1, 5, 10, and 16 years), obtained 678 BMI trajectories from birth to adolescence, and got interpolated data for ages not measured during the anthropometric assessment. Once we obtained the full BMI trajectory, in those participants who were obese or had had obesity in early childhood we estimated the timing of obesity onset and the length of obesity, with a precision of weeks. Next, based on the observation of each interpolated BMI trajectory, we created four mutually exclusive groups: participants who were never obese (NOB), participants with obesity during adolescence (recent-onset obese (ROB)), participants who were obese in early childhood but transitioned to normal/overweight as preadolescents (formerly obese (FOB)), and participants who were obese in early childhood and remained obese (persistently obese (POB)). Analysis of variance was used for comparison of mean values of anthropometric and cardiometabolic variables. Pearson Chi-square analysis assessed differences in the prevalence of cardiometabolic risk factors and MetS. To determine the contribution of long-term and recent-onset obesity to the cardiovascular risk in adolescence, we conducted analysis of covariance. The first model was unadjusted and the second model controlled for sex and breastfeeding duration. Post hoc analyses were conducted with Bonferroni correction to assess further differences between groups. A P value of <0.05 denoted statistical significance. Data were analyzed using Stata for Windows version 15.0 (Lakeway Drive College Station, TX, USA) and Python 2.7.13 for GNU/Linux (The Python Software Foundation, Wilmington, DE, USA).
Results
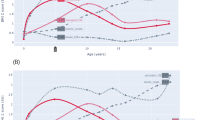
Mean age of participants (n = 678) in adolescence was 16.8 years (0.3 SD), 52.3% were males, and 24% (n = 162) had obesity in early childhood. In adolescence, the prevalence of obesity was 16.4% and 9.7% met the criteria for MetS. Of the participants, 69.2% (n = 470) were NOB, 6.8% (n = 46) were ROB, 16.6% (n = 112) were FOB, and 7.3% (n = 50) were POB. Figure 1 shows the BMI trajectory from birth to adolescence in the four groups. Mean age at obesity onset was 13.5 years (0.5 SD) in ROB adolescents, 1.5 years (0.7 SD) in FOB adolescents, and 1.9 years (0.9 SD) in POB adolescents. Likewise, the mean length of obesity was 3.3 years (0.4 SD) in ROB adolescents, 7.4 years (1.5 SD) in FOB adolescents, and 14.9 years in POB adolescents. FOB participants stopped being obese at 8.8 (1.0 SD) years of age.
Body mass index for age and sex (BAZ) from birth to adolescence in the Santiago Longitudinal Study (n = 678). NOB, participants who were never obese ROB, participants with obesity during adolescence (recent-onset obese); FOB participants who were obese in early childhood but transitioned to normal/overweigh as preadolescents (formerly obese); POB, participants who were obese in early childhood and remained obese (persistently obese)
Table 1 contains the anthropometric and cardiometabolic profile in the four groups. In the sample, sex was not related to an earlier onset or greater length of obesity. Comparison of the anthropometric profile in the adolescent period showed that ROB and POB adolescents had significantly higher values of BMI, BMIz, WC, TFM%, and lower values of TFFM% than NOB and FOB adolescents. Also, ROB and POB adolescents had similar values in all anthropometric markers assessed in the study, except for WC. Although FOB adolescents had higher BMI, BMIz, WC, TFM%, and MetS score, along with lower TFFM%, compared to the NOB group, they had lower values BMI, BMIz, WC, and TFM% and higher values of TFFM% compared to ROB and POB participants. Comparison of the anthropometric profile in infancy and childhood showed that all groups had similar weight at birth. BMIz at 1 and 5 years of age was higher in POB and FOB adolescents compared to NOB and ROB adolescents. ROB participants had higher BMIz at 5 years of age compared to NOB adolescents. BMIz at 10 years of age was similar among ROB and FOB participants, although both groups had higher values compared to NOB adolescents. At 10 years of age, POB participants had higher BMIz than NOB, FOB, and ROB adolescents. Comparison of the cardiometabolic profile in the adolescent period showed that POB and ROB participants had similar values in all biomarkers assessed. Likewise, no differences were found among NOB and FOB participants in all cardiometabolic markers assessed, except for MetS score. Last, POB and ROB adolescents had higher cardiometabolic values compared to NOB and FOB adolescents.
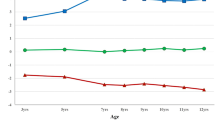
We also found that ROB and POB adolescents had a greater prevalence of abdominal obesity, high BP, hypertriglyceridemia, IR, and MetS compared to NOB and FOB adolescents (Fig. 2).
Cardiometabolic risk at 16 years by time of onset and persistence of obesity in the Santiago Longitudinal Study (n = 678). Metabolic Syndrome and its components diagnosed using the AHA/NHBLI/IDF joint statement. Insulin resistance (IR) diagnosed with homeostatic model assessment-IR values ≥2.6. NOB, participants who were never obese; ROB, participants with obesity during adolescence (recent-onset obese); FOB, participants who were obese in early childhood but transitioned to normal/overweigh as preadolescents (formerly obese); POB, participants who were obese in early childhood and remained obese (persistently obese)
Results of the regression analysis conducted to determine the contribution of recent-onset and long-term obesity to the cardiometabolic risk are contained in Table 2. After accounting for the effect of sex and breastfeeding length, we observed that ROB and POB adolescents had unhealthier anthropometric and cardiometabolic values compared to NOB adolescents (reference group) in all the biomarkers assessed. However, although FOB participants had significantly higher values of BMI (+0.56 SD), WC (+6.7 cm), TFM (+4.9%), and MetS score (+0.27) than NOB participants, no difference were found in SBP, DBP, glucose, insulin, HOMA-IR, TG, HDL, and hs-CRP.
A comparison of cardiometabolic markers between groups (post hoc analysis) showed that ROB and POB adolescents had a similar profile, except for the fact that POB participants had higher WC values (+6.0 cm) compared ROB adolescents (Fig. 3). Likewise, we found that FOB adolescents had significantly lower values of TFM, WC, SBP, TG, insulin, HOMA-IR, and hs-CRP, along with higher HDL-chol than ROB and POB participants (see also Tables S1 and S2 in Suplemmentary Material).
Association of age of onset and persistency of obesity with cardiometabolic profile in adolescence: Post hoc analysis for selected biomarkers (n = 678). NOB, participants who were never obese; ROB, participants with obesity during adolescence (recent-onset obese); FOB, participants obese in early childhood but transitioned to normal/overweigh as preadolescents (formerly obese); POB, participants who were obese in early childhood and remained obese (persistently obese). a Systolic blood pressure, b HDL cholesterol; c homeostatic model assessment-insulin resistance; d Metabolic Syndrome. (1) Significantly different from NOB. (2) Significantly different from ROB. (3) Significantly different from FOB. (4) Significantly different from POB
Discussion
This study shows that recent-onset obesity raises the risk of cardiometabolic disorders in adolescence as much as long-term obesity. In our sample, adolescents with recent-onset obesity had a similar cardiometabolic profile than adolescents who were obese since early childhood and remained obese through adolescence. Several follow-up studies agree that the relationship between obesity in early childhood and cardiovascular risk during adulthood is associated with the trajectory of BMI.34,35,36,37 Different BMI trajectories might be related to different health risks in adult life but not yet in adolescence. A retrospective cohort study comparing adults with and without MetS showed that BMI trajectories were similar in the first years of life but differed progressively since the age of 4 years.38 Yet another longitudinal study showed that increased birth weight in both preterm and term children was associated with early obesity that persists in the school stage.39 A review of several longitudinal studies showed that high BMI since birth was associated with both high lean and fat body masses in adulthood, whereas low or normal BMI at birth followed by an early AR and subsequent increase in BMI was associated with increased fat body mass rather than increased lean body mass in the adult years.14 This second model may be associated with greater health risks than the first model. In our sample, POB participants had a normal birth weight and a BMI trajectory of biological risk started around the age of 2 years, thus they are closer to the second model. Although in our study persistence of obesity was not related to greater cardiometabolic risk in adolescence compared to recent-onset obesity, we are unable to say whether POB adolescents will have similar cardiometabolic values than ROB adolescents at some point in adulthood.
A second major finding shows that obesity in early childhood led to increased cardiometabolic risk in adolescence only when obesity persisted through the years. In our sample, participants who were obese before the age of 6 years but transitioned to a normal/overweight in their preadolescent years did not show an unhealthy cardiometabolic profile at 16 years. This group showed a BMI trajectory of biological risk for about 7 years.
Although a strong body of evidence shows that obesity in early childhood is associated with higher cardiometabolic risk in future, these studies do not differentiate between subjects who were obese in early childhood and stopped being obese later and those who were obese in early childhood and remained obese. In our sample, almost 70% of participants who were obese before the age of 6 years were no longer obese in preadolescence or adolescence. At 16 years, this group had a cardiometabolic profile similar to participants who had normal weight since infancy and a much healthier cardiometabolic profile than recent-onset and POB participants. This suggests that the cardiometabolic risk associated with early obesity might be reversible when children are able to transition to normal/overweight. However, these children should be followed as they grow, because although FOB adolescents did not show an unhealthier cardiometabolic profile compared to NOB adolescents, the former did have significantly higher values of BMI, TFM%, and MetS score at 16 years than the latter.
Our findings highlight the importance of protecting lifestyles (i.e. diet and physical activity) in the early stages of life, paying special attention in children who had BMI trajectories of risk before the age of 6 years. In Chile, this is particularly relevant since the prevalence of obesity in the population aged <6 years is twice the prevalence of obesity in adolescents: 27% vs. 13%.3 Likewise, in the current obesogenic context it is quite more difficult to leave obesity behind, and thus children who had obesity since the early stages of life would be more exposed to remain obese later. A recent follow-up in >30,000 German adolescents aged between 15 and 18 years found that half of the obese adolescents in the sample had been overweight/obese since the age of 5 years. Moreover, nearly 90% of children who were obese at age 3 years were overweight/obese in adolescence.40
Limitations and strengths
This study has some limitations that should be considered while interpreting its results. Our study population is not representative of the Chilean adolescent population because it included adolescents of low-to-middle SES. However, our findings may be relevant because of several reasons. According to several population surveys, the prevalence of obesity and cardiometabolic risk factors is significantly higher in individuals of low-to-middle SES.3,4 Also, >50% of children aged <6 years from these SES levels are overweight or obese.3 Another major limitation has to do with the fact that all of our participants had normal birth weight, so our findings cannot be generalized to children with extreme birth weights for gestational age. This could underestimate the cardiometabolic risk associated with long-term and recent-onset obesity. Despite these limitations, our study has several strengths. This sample is made up of adolescents born in the 1990s, during the emergence of the obesity epidemic in Chile. Moreover, they are offspring of parents or grandparents who were exposed to childhood undernutrition. Thus our cohort is an exceptional setting to examine the impact of obesity since early childhood on subsequent cardiometabolic risk in a population exposed to the programming of a thrifty phenotype. A second major strength is the availability of health and growth data from birth to adolescence, with multiple evaluations that allows estimation of BMI trajectories since the very beginning, using mathematical models and computer algorithms. Still, we must admit that a model based on interpolated data has its limitations since interpolated data do not have the same reliability as measured data. Last, in multiple studies, the BMI trajectory since childhood has been modeled by linear spline; here we used polynomial functions that allow smoothing this trajectory and thus obtain a more realistic representation.33,34 Cubic splines provide a better fit for the data than linear splines because the cubic interpolator requires the function to be continuous, as well as the first and second derivatives of the function; this is what allows smoothing the trajectory between two discrete points. In healthy subjects, we know that changes in the BMI trend are not abrupt, and so they are better represented through a cubic function compared to a linear function. In contrast, the linear interpolator only requires that the function be continuous but not the derivatives. In the trajectory that is reflected in the fact that the line connecting two points abruptly changes its trajectory.
Conclusion
This cohort study showed that both long-term and recent-onset obesity increase the risk of cardiometabolic disorders in adolescence. In fact, recent-onset obesity raised the risk of cardiometabolic disorders in adolescence as much as long-term obesity. Also, we found that adolescents who had early obesity but were able to transition to normal/overweight had a healthier cardiometabolic profile compared to recent-onset and POB adolescents. Thus tackling obesity during critical stages of development such as infancy, childhood, and adolescence might be an effective strategy to prevent persistence of obesity and reduce the risk of cardiometabolic diseases in adulthood.
References
Ng, M. et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: a systematic analysis for the Global Burden of Disease Study. Lancet 384, 766–781 (2014).
Muzzo, S., Cordero, J., Ramírez, I. & Burrows, R. Trend in nutritional status and stature among school age children in Chile. Nutrition 20, 867–973 (2004).
Informe Mapa Nutricional 2017. National Board of Scholarship and School Aid (Ministry of Education, Santiago, 2018).
Encuesta Nacional de Salud 2016-2017. Resultados Preliminares (Department of Epidemiology, Ministry of Health, Santiago, 2017).
Faienza, M., Wang, D., Fruhbeck, D., Garruti, G. & Portincas, P. The dangerous link between childhood and adulthood predictors of obesity and metabolic syndrome. Intern. Emerg. Med. 11, 175–182 (2016).
Kuwahara, E. et al. Increased childhood BMI is associated with young adult serum uric acid levels: a linkage study from Japan. Pediatr. Res. 81, 293–298 (2017).
Rolland-Cachera, M. F. et al. Adiposity rebound in children: a simple indicator for predicting obesity. Am. J. Clin. Nutr. 39, 129–135 (1984).
Barker, D. J. The developmental origins of adult disease. J. Am. Coll. Nutr. 23, 588–595 (2004).
Singhal, A. & Lucas, A. Early origins of cardiovascular disease: is there a unifying hypothesis? Lancet 363, 1642–1645 (2004).
Doi, L., Williams, A. & Frank, J. How has child growth around adiposity rebound altered in Scotland since 1990 and what are the risk factors for weight gain using the Growing Up in Scotland birth cohort 1? BMC Public Health 16, 1081 (2016).
Campbells, K. Biological, environmental, and social influences on childhood obesity. Pediatr. Res. 79, 205–211 (2015).
Jacota, M. et al. Maternal weight prior and during pregnancy and offspring’s BMI and adiposity at 5–6 years in the EDEN mother-child cohort. Pediatr. Obes. 12, 320–329 (2017).
Hughes, A., Sherriff, A., Ness, A. & Reilly, J. Timing of adiposity rebound and adiposity in adolescence. Pediatrics 134, e1354–e1361 (2014).
Rolland-Cachera, M. F. & Péneau, S. Growth trajectories associated with adult obesity. World Rev. Nutr. Diet. 106, 127–134 (2013).
Koyama, S. et al. Adiposity rebound and the development of Metabolic Syndrome. Pediatrics 133, 114–119 (2014).
Bornhorst, C. et al. Associations between early body mass index trajectories and later metabolic risk factors in European children: the IDEFICS study. Eur. J. Epidemiol. https://doi.org/10.1007/s10654-015-0080-z (2015).
Pacheco, L. et al. Early onset obesity and risk of metabolic syndrome among Chilean adolescents. Prev. Chronic Dis. 14, 170132 (2017).
Eriksson, J. et al. Early adiposity rebound in childhood and risk of type 2 diabetes in adult life. Diabetologia 46, 190–194 (2003).
Boyne, M. S. et al. Age at adiposity rebound is a predictor of blood pressure and body composition in Jamaican children. Pediatr. Res. 53, 34A (2003).
Péneau, S., Thibault, H. & Rolland-Cachera, M. F. Massively obese adolescents were of normal weight at the age of adiposity rebound. Obesity 17, 1309–1310 (2009).
Koyama, S., Sairenchi, T., Shimura, N. & Arisaka, O. Association between timing of adiposity rebound and body weight gain during infancy. J. Pediatr. 166, 309–12 (2015).
Cole, T. Children grow and horses race: is the adiposity rebound a critical period for later obesity? BMC Pediatr. 4, 6 (2004).
Lozoff, B. et al. Behavioral and developmental effects of preventing iron-deficiency anemia in healthy full-term infants. Pediatrics 112, 846–854 (2003).
Burrows, R. et al. High cardiometabolic risk in healthy Chilean adolescents: association with anthropometric, biological and life style factors. Public Health Nutr. 19, 486–493 (2016).
Reyes, M., Burrows, R., Blanco, E., Lozoff, B. & Gahaghan, S. Greater early weight gain and shorter breastfeeding are associated with low adolescent adiponectin levels. Pediatr. Obes. 13, 77–284 (2018).
Wallace, T. M., Levy, J. C. & Matthews, D. R. Use and abuse of HOMA modeling. Diabetes Care 27, 1487–1495 (2004).
Burrows, R. et al. Healthy Chilean adolescents with HOMA-IR ≥ 2.6 have increased cardio-metabolic risk: association with genetic, biological and environmental factors. J. Diabetes Res. 2015, 783296 (2015).
Pearson, T. et al. Markers of inflammation and cardiovascular disease. application to clinical and public health practice. Circulation 107, 499–511 (2003).
Alberti, K. G. et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 120, 1640–1645 (2009).
Gurka, M., Ice, C., Sun, S. & DeBoer, M. A confirmatory factor analysis of the metabolic syndrome in adolescents: an examination of sex and racial/ethnic differences. Cardiovasc. Diabetol. 11, 128–137 (2012).
Khuc, K. et al. Adolescent metabolic syndrome risk is increased with higher infancy weight gain and decreased with longer breast feeding. Int. J. Pediatr. 2012, 478610 (2012).
Royston, P., Ambler, G. & Sauerbrei, W. The use of fractional polynomials to model continuous risk variables in epidemiology. Int. J. Epidemiol. 28, 964–974, https://doi.org/10.1093/ije/28.5.964 (1999).
Kiusalaas, J. in Numerical Methods in Engineering with Python 2nd edn Ch. 3 (Cambridge University Press, Cambridge, 2010).
Boyer, B., Nelson, J. & Holub, S. Childhood BMI trajectories predicting cardiovascular risk in adolescence. J. Adolesc. Health 56, 599–605 (2015).
Fabricius-Bjerre, S. et al. Impact of birth weight and early infant weight gain on insulin resistance and associated cardiovascular risk factors in adolescence. PLoS ONE 6, e20595 (2011).
Araújo, J. et al. Developmental trajectories of adiposity from birth until early adulthood and association with cardiometabolic risk factors. Int. J. Obes. 39, 1443–1449 (2015).
Besharat Pour, M. et al. Age at adiposity rebound and body mass index trajectory from early childhood to adolescence: differences by breastfeeding and maternal immigration background. Pediatr. Obes. 12, 75–84 (2017).
Giudic, K. et al. Body mass index growth trajectories associated with the different parameters of the metabolic syndrome at adulthood. Int. J. Obes. (Lond.) 41, 1518–1525 (2017)
Kapral, N., Miller, S., Scharf, R., Gurka, M. & DeBoer, M. Associations between birthweight and overweight and obesity in school-age children. Pediatr. Obes. 13, 333–341 (2018).
Geserick, M. et al. Acceleration of BMI in early childhood and risk of sustained obesity. N. Engl. J. Med. 379, 1303–1312 (2018).
Acknowledgements
This research was supported by grants from the: National Heart, Lung and Blood Institute (Grant no. HL088530; to S.G.); Consejo Nacional de Ciencia y Tecnología (CONICYT) (Chile) through Programa de Capital Humano Avanzado (grant #79140003; to P.C.) and Fondo Nacional de Investigaciones Científicas y Tecnológicas (FONDECYT) (grant #1160639 and #1190662; to J.R.) (grant #1160240; to P.C., R.B.); and Centro para la Nanociencia y la Nanotecnología (CEDENNA) through Financiamiento Basal para Centros Científicos y Tecnológicos de Excelencia (grant #FB0807; to J.R.). These organizations had no role in study design; collection, analysis, and interpretation of data; writing the manuscript; and the decision to submit it for publication.
Author contributions
Conceptualization, RB and PCB; methodology, R.B., P.C.B., J.R.; formal analysis, P.C.B., J.R., E.C.; investigation, R.B., P.C.B.; data curation, R.B.; writing—original draft preparation, R.B.; writing—review and editing, E.B., E.C., S.G., P.C.B., J.R.; supervision, R.B., S.G.; project administration, R.B., E.B.; funding acquisition, S.G., R.B., P.C.B., J.R.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Burrows, R., Correa-Burrows, P., Rogan, J. et al. Long-term vs. recent-onset obesity: their contribution to cardiometabolic risk in adolescence. Pediatr Res 86, 776–782 (2019). https://doi.org/10.1038/s41390-019-0543-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-019-0543-0
This article is cited by
-
Hypertension in obese children is associated with vitamin D deficiency and serotonin dysregulation
BMC Pediatrics (2022)
-
Resolving early obesity leads to a cardiometabolic profile within normal ranges at 23 years old in a two-decade prospective follow-up study
Scientific Reports (2021)
-
Validity assessment of the single-point insulin sensitivity estimator (spise) for diagnosis of cardiometabolic risk in post-pubertal hispanic adolescents
Scientific Reports (2020)