Abstract
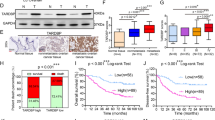
Thrombospondin 1 (TSP1) is known for its cell-specific functions in cancer progression, such as proliferation and migration. It contains 22 exons that may potentially produce several different transcripts. Here, we identified TSP1V as a novel TSP1-splicing variant produced by intron retention (IR) in human thyroid cancer cells and tissues. We observed that TSP1V functionally inhibited tumorigenesis contrary to TSP1 wild-type, as identified in vivo and in vitro. These activities of TSP1V are caused by inhibiting phospho-Smad and phospho-focal adhesion kinase. Reverse transcription polymerase chain reaction and minigene experiments revealed that some phytochemicals/non-steroidal anti-inflammatory drugs enhanced IR. We further found that RNA-binding motif protein 5 (RBM5) suppressed IR induced by sulindac sulfide treatment. Additionally, sulindac sulfide reduced phospho-RBM5 levels in a time-dependent manner. Furthermore, trans-chalcone demethylated TSP1V, thereby preventing methyl-CpG-binding protein 2 binding to TSP1V gene. In addition, TSP1V levels were significantly lower in patients with differentiated thyroid carcinoma than in those with benign thyroid nodule, indicating its potential application as a diagnostic biomarker in tumor progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data generated or analyzed in this study are included in this published article.
References
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. Ca-Cancer J Clin. 2022;72:7–33.
Kang MJ, Won YJ, Lee JJ, Jung KW, Kim HJ, Kong HJ, et al. Cancer Statistics in Korea: Incidence, mortality, survival, and prevalence in 2019. Cancer Res Treat. 2022;54:330–44.
Li RX, Hao JP, Zhu Z, Qiao XD, Wang L, Zhou ZB. Correlation between US-FNAC with BRAF V600E Mutation Analysis and Central Neck Lymph Node Metastasis in cN0 Papillary Thyroid Cancer. Biomed Res Int. 2021;2021:9937742.
Yoon RG, Baek JH, Lee JH, Choi YJ, Hong MJ, Song DE, et al. Diagnosis of thyroid follicular neoplasm: fine-needle aspiration versus core-needle biopsy. Thyroid. 2014;24:1612–7.
Stanek-Widera A, Biskup-Fruzynska M, Zembala-Nozynska E, Poltorak S, Snietura M, Lange D. Suspicious for follicular neoplasm or follicular neoplasm? The dilemma of a pathologist and a surgeon. Endokrynol Pol. 2016;67:17–22.
Hong Y, Lee J, Moon H, Ryu CH, Seok J, Jung YS, et al. Quercetin induces anticancer activity by upregulating Pro-NAG-1/GDF15 in differentiated thyroid cancer cells. Cancers (Basel). 2021;13:3022.
Fusco A, Grieco M, Santoro M, Berlingieri M, Pilotti S, Pierotti M, et al. A new oncogene in human thyroid papillary carcinomas and their lymph-nodal metastases. Nature. 1987;328:170–2.
Giordano TJ, Kuick R, Thomas DG, Misek DE, Vinco M, Sanders D, et al. Molecular classification of papillary thyroid carcinoma: distinct BRAF, RAS, and RET/PTC mutation-specific gene expression profiles discovered by DNA microarray analysis. Oncogene. 2005;24:6646–56.
Raman P, Koenig RJ. Pax-8-PPAR-γ fusion protein in thyroid carcinoma. Nat Rev Endocrinol. 2014;10:616–23.
Herrmann MA, Hay ID, Bartelt DH Jr, Ritland SR, Dahl RJ, Grant CS, et al. Cytogenetic and molecular genetic studies of follicular and papillary thyroid cancers. J Clin Invest. 1991;88:1596–604.
Marotta V, Guerra A, Sapio MR, Vitale M. RET/PTC rearrangement in benign and malignant thyroid diseases: a clinical standpoint. Eur J Endocrinol. 2011;165:499–507.
Grosso AR, Martins S, Carmo-Fonseca M. The emerging role of splicing factors in cancer. EMBO Rep. 2008;9:1087–93.
Garcia-Blanco MA, Baraniak AP, Lasda EL. Alternative splicing in disease and therapy. Nat Biotechnol. 2004;22:535–46.
Lazzereschi D, Nardi F, Turco A, Ottini L, D’Amico C, Mariani-Costantini R, et al. A complex pattern of mutations and abnormal splicing of Smad4 is present in thyroid tumours. Oncogene. 2005;24:5344–54.
Huang T, Sun L, Yuan X, Qiu H. Thrombospondin-1 is a multifaceted player in tumor progression. Oncotarget. 2017;8:84546–58.
Patwardhan S, Mahadik P, Shetty O, Sen S. ECM stiffness-tuned exosomes drive breast cancer motility through thrombospondin-1. Biomaterials. 2021;279:121185.
Mo DP, He F, Zheng JY, Chen HH, Tang L, Yan F. tRNA-Derived Fragment tRF-17-79MP9PP attenuates cell invasion and migration via THBS1/TGF-beta 1/Smad3 Axis in breast cancer. Front Oncol. 2021;11:656078.
Xiao JW, Zhang Y, Tang Y, Dai HF, OuYang Y, Li CC, et al. hsa-miR-4443 inhibits myocardial fibroblast proliferation by targeting THBS1 to regulate TGF-beta 1/alpha-SMA/collagen signaling in atrial fibrillation. Braz J Med Biol Res. 2021;54:e10692.
Yin Q, Wang PP, Peng R, Zhou H. MiR-19a enhances cell proliferation, migration, and invasiveness through enhancing lymphangiogenesis by targeting thrombospondin-1 in colorectal cancer. Biochem Cell Biol. 2019;97:731–9.
Duquette M, Sadow PM, Lawler J, Nucera C. Thrombospondin-1 Silencing down-regulates integrin expression levels in human anaplastic thyroid cancer cells with BRAF(V600E): New insights in the host tissue adaptation and homeostasis of tumor microenvironment. Front Endocrinol (Lausanne). 2013;4:189.
Soula-Rothhut M, Coissard C, Sartelet H, Boudot C, Bellon G, Martiny L, et al. The tumor suppressor PTEN inhibits EGF-induced TSP-1 and TIMP-1 expression in FTC-133 thyroid carcinoma cells. Exp Cell Res. 2005;304:187–201.
Rath GM, Schneider C, Dedieu S, Rothhut B, Soula-Rothhut M, Ghoneim C, et al. The C-terminal CD47/IAP-binding domain of thrombospondin-1 prevents camptothecin- and doxorubicin-induced apoptosis in human thyroid carcinoma cells. Biochim Biophys Acta. 2006;1763:1125–34.
Nucera C, Porrello A, Antonello ZA, Mekel M, Nehs MA, Giordano TJ, et al. B-RafV600 and thrombospondin-1 promote thyroid cancer progression. Proc Natl Acad Sci. 2010;107:10649.
Joseph JV, Magaut CR, Storevik S, Geraldo LH, Mathivet T, Latif MA, et al. TGF-beta promotes microtube formation in glioblastoma through thrombospondin 1. Neuro Oncol. 2022;24:541–53.
Wang H, Khor TO, Shu L, Su Z-Y, Fuentes F, Lee J-H, et al. Plants vs. cancer: A review on natural phytochemicals in preventing and treating cancers and their druggability. Anticancer Agents Med Chem. 2012;12:1281–305.
Oh J, Pradella D, Shao C, Li H, Choi N, Ha J, et al. Widespread alternative splicing changes in metastatic breast cancer cells. Cells. 2021;10:858.
Du JX, Luo YH, Zhang SJ, Wang B, Chen C, Zhu GQ, et al. Splicing factor SRSF1 promotes breast cancer progression via oncogenic splice switching of PTPMT1. J Exp Clin Cancer Res. 2021;40:171.
Murayama R, Kimura-Asami M, Togo-Ohno M, Yamasaki-Kato Y, Naruse TK, Yamamoto T, et al. Phosphorylation of the RSRSP stretch is critical for splicing regulation by RNA-Binding Motif Protein 20 (RBM20) through nuclear localization. Sci Rep. 2018;8:8970.
Wong JJL, Gao DD, Nguyen TV, Kwok CT, van Geldermalsen M, Middleton R, et al. Intron retention is regulated by altered MeCP2-mediated splicing factor recruitment. Nat Commun. 2017;8:15134.
Chatterjee B, Lin MH, Chen CC, Peng KL, Wu MS, Tseng MC, et al. DNA Demethylation by DNMT3A and DNMT3B in vitro and of Methylated Episomal DNA in Transiently Transfected Cells. Bba-Gene Regul Mech. 2018;1861:1048–61.
Kaczkowski B, Tanaka Y, Kawaji H, Sandelin A, Andersson R, Itoh M, et al. Transcriptome analysis of recurrently deregulated genes across multiple cancers identifies new pan-cancer biomarkers. Cancer Res. 2016;76:216–26.
Hsiao SJ, Nikiforov YE. Molecular approaches to thyroid cancer diagnosis. Endocr Relat Cancer. 2014;21:T301–T313.
Stenina OI, Ustinov V, Krukovets I, Marinic T, Topol EJ, Plow EF. Polymorphisms A387P in thrombospondin-4 and N700S in thrombospondin-1 perturb calcium binding sites. The FASEB Journal. 2005;19:1893–5.
Adolph KW. Detection of exon skipping and retained introns in transcription of the human Thrombospondin 2 Gene. Biochem Biophys Res Commun. 1999;259:527–32.
Hirose Y, Chiba K, Karasugi T, Nakajima M, Kawaguchi Y, Mikami Y, et al. A functional polymorphism in THBS2 that affects alternative splicing and MMP binding is associated with lumbar-disc herniation. Am J Hum Genet. 2008;82:1122–9.
Lee JH, Horak CE, Khanna C, Meng Z, Yu LR, Veenstra TD, et al. Alterations in Gemin5 expression contribute to alternative mRNA splicing patterns and tumor cell motility. Cancer Res. 2008;68:639–44.
Silva G, Marins M, Fachin AL, Lee SH, Baek SJ. Anti-cancer activity of trans-chalcone in osteosarcoma: Involvement of Sp1 and p53. Mol Carcinog. 2016;55:1438–48.
Komoto TT, Bernardes TM, Mesquita TB, Bortolotto LFB, Silva G, Bitencourt TA, et al. Chalcones repressed the AURKA and MDR proteins involved in metastasis and multiple drug resistance in breast cancer cell lines. Molecules. 2018;23:2018.
Xiao SH, Manley JL. Phosphorylation of the ASF/SF2 RS domain affects both protein-protein and protein-RNA interactions and is necessary for splicing. Genes Dev. 1997;11:334–44.
Duncan PI, Stojdl DF, Marius RM, Bell JC. In vivo regulation of alternative pre-mRNA splicing by the Clk1 protein kinase. Mol Cell Biol. 1997;17:5996–6001.
Cao W, Jamison SF, Garcia-Blanco MA. Both phosphorylation and dephosphorylation of ASF/SF2 are required for pre-mRNA splicing in vitro. RNA. 1997;3:1456–67.
Mathew R, Hartmuth K, Mohlmann S, Urlaub H, Ficner R, Luhrmann R. Phosphorylation of human PRP28 by SRPK2 is required for integration of the U4/U6-U5 tri-snRNP into the spliceosome. Nat Struct Mol Biol. 2008;15:435–43.
Ngo JC, Giang K, Chakrabarti S, Ma CT, Huynh N, Hagopian JC, et al. A sliding docking interaction is essential for sequential and processive phosphorylation of an SR protein by SRPK1. Mol Cell. 2008;29:563–76.
Zhong XY, Ding JH, Adams JA, Ghosh G, Fu XD. Regulation of SR protein phosphorylation and alternative splicing by modulating kinetic interactions of SRPK1 with molecular chaperones. Genes Dev. 2009;23:482–95.
Jiang K, Patel NA, Watson JE, Apostolatos H, Kleiman E, Hanson O, et al. Akt2 regulation of Cdc2-like kinases (Clk/Sty), serine/arginine-rich (SR) protein phosphorylation, and insulin-induced alternative splicing of PKCbetaII messenger ribonucleic acid. Endocrinology. 2009;150:2087–97.
Ghigna C, Valacca C, Biamonti G. Alternative splicing and tumor progression. Curr Genomics. 2008;9:556–70.
Timmer T, Terpstra P, van den Berg A, Veldhuis PM, Ter Elst A, Voutsinas G, et al. A comparison of genomic structures and expression patterns of two closely related flanking genes in a critical lung cancer region at 3p21.3. Eur J Human Genet.: EJHG. 1999;7:478–86.
Kobayashi T, Ishida J, Shimizu Y, Kawakami H, Suda G, Muranaka T, et al. Decreased RNA-binding motif 5 expression is associated with tumor progression in gastric cancer. Tumor Biol. 2017;39:1010428317694547.
Kobayashi T, Ishida J, Musashi M, Ota S, Yoshida T, Shimizu Y, et al. p53 transactivation is involved in the antiproliferative activity of the putative tumor suppressor RBM5. Int J Cancer. 2011;128:304–18.
Rintala-Maki ND, Goard CA, Langdon CE, Wall VE, Traulsen KE, Morin CD, et al. Expression of RBM5-related factors in primary breast tissue. J Cell Biochem. 2007;100:1440–58.
Lertpatipanpong P, Lee J, Kim I, Eling T, Oh SY, Seong JK, et al. The anti-diabetic effects of NAG-1/GDF15 on HFD/STZ-induced mice. Sci Rep-UK. 2021;11:15027.
Acknowledgements
We gratefully acknowledge the Korea Mouse Phenotyping Center (KMPC) for their technical support, especially Jongdoo Kim, PhD.
Funding
This work was supported by the Research Institute for Veterinary Science and BK21 PLUS Program for Creative Veterinary Science Research Center, Seoul National University, and by a National Research Foundation of Korea (NRF) grant funded by the Korean government (2020R1A2B2002923) to SJB. This work was also supported by a clinical research grant (NCC2211670-1) provided by the National Cancer Center to JSR, CHR, and SJB.
Author information
Authors and Affiliations
Contributions
YH, JR, and SJB. conceived of the study and designed the experiments. YH, IK, HM, JL, and PL. performed the experiments and collected data. CHR, YSJ, JS, YK, JR, and SJB. interpreted the results. YH, JR, and SJB. wrote the original and revised versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was approved by the Institutional Review Board of the National Cancer Center (NCC-2014-0003), mouse experiments were approved by the Institutional Animal Care and Use Committee of Seoul National University (SNU-210511-2-1), and all experiments were conducted in accordance with the relevant guidelines and regulations.
Consent for publication
All participants provided written informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hong, Y., Kim, I., Moon, H. et al. Novel thrombospondin-1 transcript exhibits distinctive expression and activity in thyroid tumorigenesis. Oncogene 42, 1832–1842 (2023). https://doi.org/10.1038/s41388-023-02692-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-023-02692-9