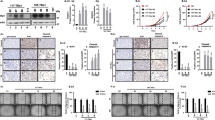
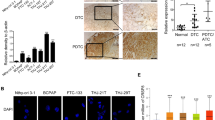
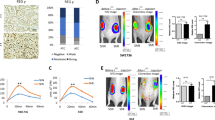
Abstract
Increasing numbers of cancer stem cell markers have been recently identified. It is not known, however, whether a member of the nuclear receptor superfamily, thyroid hormone receptor β (TRβ), can function to regulate cancer stem cell (CSC) activity. Using anaplastic thyroid cancer cells (ATC) as a model, we highlight the role of TRβ in CSC activity. ATC is one of the most aggressive solid cancers in humans and is resistant to currently available therapeutics. Recent studies provide evidence that CSC activity underlies aggressiveness and therapeutic resistance of ATC. Here we show that TRβ inhibits CSC activity by suppressing tumor-sphere formation of human ATC cells and their tumor-initiating capacity. TRβ suppresses the expression of CSC regulators, including ALDH, KLF2, SOX2, b-catenin, and ABCG2, in ATC cell-induced xenograft tumors. Single-cell transcriptomic analysis shows that TRβ reduces CSC population in ATC-induced xenograft tumors. Analysis of The Cancer Genome Atlas (TCGA) database demonstrates that the inhibition of CSC capacity by TRβ contributes to favorable clinical outcomes in human cancer. Our studies show that TRβ is a newly identified transcription regulator that acts to suppress CSC activity and that TRβ could be considered as a molecular target for therapeutic intervention of ATC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bonnet D, Dick JE. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med. 1997;3:730–7.
Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci. 2003;100:3983–8.
Ricci-Vitiani L, Lombardi DG, Pilozzi E, Biffoni M, Todaro M, Peschle C, et al. Identification and expansion of human colon-cancer-initiating cells. Nature. 2007;445:111–5.
Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T, et al. Identification of human brain tumour initiating cells. nature. 2004;432:396–401.
Ayob AZ, Ramasamy TS. Cancer stem cells as key drivers of tumour progression. J Biomed Sci. 2018;25:20.
Tuttle RM, Haugen B, Perrier ND Updated American Joint Committee on cancer/tumor-node-metastasis staging system for differentiated and anaplastic thyroid cancer: what changed and why? Mary Ann Liebert, Inc. 140 Huguenot Street, 3rd Floor New Rochelle, NY 10801 USA, 2017.
Smallridge RC. Approach to the patient with anaplastic thyroid carcinoma. J Clin Endocrinol Metab. 2012;97:2566–72.
Tiedje V, Stuschke M, Weber F, Dralle H, Moss L, Führer D. Anaplastic thyroid carcinoma: review of treatment protocols. Endocr-Relat cancer. 2018;25:R153–R161.
Molinaro E, Romei C, Biagini A, Sabini E, Agate L, Mazzeo S, et al. Anaplastic thyroid carcinoma: from clinicopathology to genetics and advanced therapies. Nat Rev Endocrinol. 2017;13:644–60.
Smallridge RC, Ain KB, Asa SL, Bible KC, Brierley JD, Burman KD, et al. American Thyroid Association guidelines for management of patients with anaplastic thyroid cancer. Thyroid. 2012;22:1104–39.
Subbiah V, Kreitman RJ, Wainberg ZA, Cho JY, Schellens JHM, Soria JC, et al. Dabrafenib and trametinib treatment in patients with locally advanced or metastatic BRAF V600-mutant anaplastic thyroid cancer. J Clin Oncol. 2018;36:7–13.
Lee WK, Lee SG, Yim SH, Kim D, Kim H, Jeong S, et al. Whole exome sequencing identifies a novel hedgehog-interacting protein g516r mutation in locally advanced papillary thyroid cancer. Int J Mol Sci. 2018;19:2867.
Ando S, Sarlis NJ, Oldfield EH, Yen PM. Somatic mutation of TRβ can cause a defect in negative regulation of TSH in a TSH-secreting pituitary tumor. J Clin Endocrinol Metab. 2001;86:5572–6.
Silva JM, Domínguez G, González-Sancho JM, García JM, Silva J, García-Andrade C, et al. Expression of thyroid hormone receptor/erbA genes is altered in human breast cancer. Oncogene. 2002;21:4307.
Aranda A, Martínez-Iglesias O, Ruiz-Llorente L, García-Carpizo V, Zambrano A. Thyroid receptor: roles in cancer. Trends Endocrinol Metab. 2009;20:318–24.
Kim WG, Cheng S-Y. Thyroid hormone receptors and cancer. Biochimica et Biophysica Acta (BBA)-Gen Subj. 2013;1830:3928–36.
Suzuki H, Willingham MC, Cheng S-Y. Mice with a mutation in the thyroid hormone receptor β gene spontaneously develop thyroid carcinoma: a mouse model of thyroid carcinogenesis. Thyroid. 2002;12:963–9.
Li Z, Meng ZH, Chandrasekaran R, Kuo W-L, Collins CC, Gray JW, et al. Biallelic inactivation of the thyroid hormone receptor β1 gene in early stage breast cancer. Cancer Res. 2002;62:1939–43.
Iwasaki Y, Sunaga N, Tomizawa Y, Imai H, Iijima H, Yanagitani N, et al. Epigenetic inactivation of the thyroid hormone receptor β1 gene at 3p24. 2 in lung cancer. Ann surgical Oncol. 2010;17:2222–8.
Joseph B, Ji M, Liu D, Hou P, Xing M. Lack of mutations in the thyroid hormone receptor (TR) α and β genes but frequent hypermethylation of the TR β gene in differentiated thyroid tumors. J Clin Endocrinol Metab. 2007;92:4766–70.
Kim WG, Zhu X, Kim DW, Zhang L, Kebebew E, Cheng S-Y. Reactivation of the silenced thyroid hormone receptor β gene expression delays thyroid tumor progression. Endocrinology. 2013;154:25–35.
Jazdzewski K, Boguslawska J, Jendrzejewski J, Liyanarachchi S, Pachucki J, Wardyn KA, et al. Thyroid hormone receptor β (THRB) is a major target gene for microRNAs deregulated in papillary thyroid carcinoma (PTC). J Clin Endocrinol Metab. 2011;96:E546–E553.
Lee C-H, Yu C-C, Wang B-Y, Chang W-W. Tumorsphere as an effective in vitro platform for screening anti-cancer stem cell drugs. Oncotarget. 2016;7:1215.
Park JW, Zhao L, Cheng S-Y. Inhibition of estrogen-dependent tumorigenesis by the thyroid hormone receptor β in xenograft models. Am J cancer Res. 2013;3:302.
Hu Y, Smyth GK. ELDA: extreme limiting dilution analysis for comparing depleted and enriched populations in stem cell and other assays. J immunological methods. 2009;347:70–78.
Plaks V, Kong N, Werb Z. The cancer stem cell niche: how essential is the niche in regulating stemness of tumor cells? cell stem cell. 2015;16:225–38.
Mabbott NA, Baillie JK, Brown H, Freeman TC, Hume DA. An expression atlas of human primary cells: inference of gene function from coexpression networks. BMC genomics. 2013;14:1–13.
Jean E, Laoudj‐Chenivesse D, Notarnicola C, Rouger K, Serratrice N, Bonnieu A, et al. Aldehyde dehydrogenase activity promotes survival of human muscle precursor cells. J Cell Mol Med. 2011;15:119–33.
Tanei T, Morimoto K, Shimazu K, Kim SJ, Tanji Y, Taguchi T, et al. Association of breast cancer stem cells identified by aldehyde dehydrogenase 1 expression with resistance to sequential Paclitaxel and epirubicin-based chemotherapy for breast cancers. Clin cancer Res. 2009;15:4234–41.
Agrawal N, Akbani R, Aksoy BA, Ally A, Arachchi H, Asa SL, et al. Integrated genomic characterization of papillary thyroid carcinoma. Cell. 2014;159:676–90.
Li W, Reeb AN, Sewell WA, Elhomsy G, Lin RY. Phenotypic characterization of metastatic anaplastic thyroid cancer stem cells. PLoS One. 2013;8:e65095.
Bolf EL, Gillis NE, Davidson CD, Rodriguez PD, Cozzens L, Tomczak JA, et al. Thyroid hormone receptor beta induces a tumor-suppressive program in anaplastic thyroid cancer. Mol cancer Res: MCR. 2020;18:1443–52.
Haghpanah V, Fallah P, Naderi M, Tavakoli R, Soleimani M, Larijani B. Cancer stem-like cell behavior in anaplastic thyroid cancer: a challenging dilemma. Life Sci. 2016;146:34–39.
Lee WK, Cheng S-Y. Targeting transcriptional regulators for treatment of anaplastic thyroid cancer. J Cancer Metastasis Treat. 2021;7:27–45.
Carr FE, Tai PW, Barnum MS, Gillis NE, Evans KG, Taber TH, et al. Thyroid hormone receptor-β (TRβ) mediates runt-related transcription factor 2 (Runx2) expression in thyroid cancer cells: a novel signaling pathway in thyroid cancer. Endocrinology. 2016;157:3278–92.
Ravi N, Yang M, Mylona N, Wennerberg J, Paulsson K. Global RNA expression and DNA methylation patterns in primary anaplastic thyroid cancer. Cancers. 2020;12:680–91.
Guigon C, Kim D, Willingham M, Cheng S. Mutation of thyroid hormone receptor-β in mice predisposes to the development of mammary tumors. Oncogene. 2011;30:3381–90.
Furumoto H, Ying H, Chandramouli G, Zhao L, Walker RL, Meltzer PS, et al. An unliganded thyroid hormone β receptor activates the cyclin D1/cyclin-dependent kinase/retinoblastoma/E2F pathway and induces pituitary tumorigenesis. Mol Cell Biol. 2005;25:124–35.
Kim WG, Zhao L, Kim DW, Willingham MC, Cheng S-Y. Inhibition of tumorigenesis by the thyroid hormone receptor β in xenograft models. Thyroid. 2014;24:260–9.
López-Mateo I, Alonso-Merino E, Suarez-Cabrera C, Park JW, Cheng S-Y, Alemany S, et al. Thyroid hormone receptor β inhibits self-renewal capacity of breast cancer stem cells. Thyroid. 2020;30:116–32.
Jerzak KJ, Cockburn JG, Dhesy-Thind SK, Pond GR, Pritchard KI, Nofech-Mozes S, et al. Thyroid hormone receptor beta-1 expression in early breast cancer: a validation study. Breast cancer Res Treat. 2018;171:709–17.
Marlow LA, D’Innocenzi J, Zhang Y, Rohl SD, Cooper SJ, Sebo T, et al. Detailed molecular fingerprinting of four new anaplastic thyroid carcinoma cell lines and their use for verification of RhoB as a molecular therapeutic target. J Clin Endocrinol Metab. 2010;95:5338–47.
Enomoto K, Zhu X, Park S, Zhao L, Zhu YJ, Willingham MC, et al. Targeting MYC as a therapeutic intervention for anaplastic thyroid cancer. J Clin Endocrinol Metab. 2017;102:2268–80.
Lee WK, Kim WG, Fozzatti L, Park S, Zhao L, Willingham MC, et al. Steroid receptor coactivator-3 as a target for anaplastic thyroid cancer. Endocr-Relat cancer. 2020;27:209–20.
Lee WK, Lee J, Kim H, Lee SG, Choi SH, Jeong S, et al. Peripheral location and infiltrative margin predict invasive features of papillary thyroid microcarcinoma. Eur J Endocrinol. 2019;181:139–49.
Acknowledgements
We thank Drs. Zachary Rae, Michael Kelly, and Kimia Dadkhah for carrying out single-cell RNA sequencing at the Single Cell Analysis Facility, Cancer Research Technology Program, Frederick National Laboratory (Leidos Biomed), NCI. We also thank Joelle Mornini, NIH Library, for manuscript editing assistance.
Funding
This research is supported by the Intramural Research Program of the Center for Cancer Research, National Cancer Institute, National Institutes of Health.
Author information
Authors and Affiliations
Contributions
Conception and designs were performed by SC and WKL; Development of methodology and acquisition of data were carried out by WKL, XZ, SP, LZ, and JZ; Analysis and interpretation of data were performed by WKL, XZ, SP, JZ, PM, and SC; Writing, review, and/or revision were performed by SC and WKL. Administrative, technical, or material support were performed by LZ, PM, and SC.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Lee, W.K., Zhu, X., Park, S. et al. Regulation of cancer stem cell activity by thyroid hormone receptor β. Oncogene 41, 2315–2325 (2022). https://doi.org/10.1038/s41388-022-02242-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-022-02242-9
This article is cited by
-
Tumor cell plasticity in targeted therapy-induced resistance: mechanisms and new strategies
Signal Transduction and Targeted Therapy (2023)