Abstract
Background
The objective was to test the efficacy of a scalable, virtually delivered, diabetes-tailored weight management program on glycemic control in adults with type 2 diabetes (T2D).
Methods
This was a single arm, three-site clinical trial. Participants had baseline HbA1c between 7–11% and BMI between 27–50 kg/m2. Primary outcome was change in HbA1c at 24 weeks. Secondary outcomes were changes in body weight, waist circumference, the Diabetes Distress Scale (DDS), quality of life (IWQOL-L), and hunger (VAS). Generalized linear effects models were used for statistical analysis.
Results
Participants (n = 136) were 56.8 ± 0.8 y (Mean ± SEM), 36.9 ± 0.5 kg/m2, 80.2% female, 62.2% non-Hispanic white. Baseline HbA1c, weight, and total DDS score were 8.0 ± 0.09%, 101.10 ± 1.47 kg, and 2.35 ± 0.08, respectively. At week 24, HbA1c, body weight, and total DDS decreased by 0.75 ± 0.11%, 5.74 ± 0.50%, 0.33 ± 0.10 units, respectively (all p < 0.001). Also, at week 24, quality of life increased by 9.0 ± 1.2 units and hunger decreased by 14.3 ± 2.4 units, (both p < 0.0001).
Conclusions
The scalable, virtually delivered T2D-tailored weight management program had favorable and clinically meaningful effects on glycemic control, body weight, and psychosocial outcomes.
Similar content being viewed by others
Introduction
Diabetes is a debilitating, deadly, and costly disease [1, 2]. In 2012, the cost of care for diabetes totaled $245 billion, including $176 billion in direct medical costs, which is 2.3 times higher than costs in people without diabetes [3].
The American Diabetes Association (ADA) Standards of Care underscore the multiple benefits of weight management in the effective management of type 2 diabetes (T2D) [4]. Among those with T2D, the LookAHEAD study demonstrated that an intensive lifestyle intervention produced a 7.9% greater reduction in weight and a 0.5% greater reduction in hemoglobin A1c (HbA1c) than did a usual care approach at 1 year [5].
While clinic-based lifestyle interventions that reduce both weight and HbA1c are the most studied [6], their high cost and finite number, limit reach, accessibility, and impact for a large number of patients [7]. Community-based weight management programs are more affordable [8] and accessible than clinic-based modalities [9], and have been shown to be effective in promoting weight loss and improvements in glycemic control in adults [10] A randomized trial of a modified WeightWatchers (WW) program for patients with T2D showed improved glycemic control (HbA1c) and significant reductions in weight compared to standard of care diabetes nutrition counseling and education [11]. Here, we test the efficacy of a new WW program tailored for individuals with T2D and delivered virtually. We hypothesized that the program would result in clinically meaningful reductions in HbA1c [6].
Methods
This single-arm, three-site trial included Pennington Biomedical Research Center in Baton Rouge, LA, University of Florida in Gainesville, FL, and Virginia Commonwealth University in Richmond, VA. All participants were given verbal and written explanations about the study, provided written informed consent, and received incentives for data collection visits ($50 at 0 and 12 weeks, $125 at 24 weeks). Participants were recruited in cohorts, ranging from 7 to 31 participants (mean n = 17), between April–June 2021. The study was approved by the Institutional Review Boards at all three sites and registered at ClinicalTrials.gov (NCT04804774).
Participants had a reported diagnosis of T2D, were 18–70 y, objectively measured HbA1c between 7–11% and Body Mass Index (BMI) 27–50 kg/m2, on a stable medication regime for >3 months, and under a physician’s care for the management of T2D. The average length of time on T2D medication was 4 years. The number of participants on diabetes medication classes are as follows: (1) Rapid – Acting Insulin, n = 14; (2) Intermediate – Acting Insulin, n = 2; (3) Long – Acting Insulin, n = 21; (4) Combination Insulin, n = 1; (5) Biguanides, n = 40; (6) Sulfonylureas, n = 16; (7) Glucagon-like Peptide-1 agonists, n = 27; (8)Thiazolidinediones, n = 1; (9) Meglitinides n = 1; (10) DPP-4 Inhibitors, n = 11, (11) SGLT-2 Inhibitors, n = 16; and (12) Dopamine agonists, n = 1. Also, full exclusion criteria are listed below:
Exclusion criteria
-
1.
Participation in a weight control program within the past 3 months
-
2.
Weight loss of ≥5 kg in the previous 6 months
-
3.
Taking prescription or OTC weight loss medications within last 4 weeks
-
4.
History of a surgical procedure for weight loss at any time (e.g. gastroplasty, gastric by-pass, gastrectomy or partial gastrectomy, adjustable banding, gastric sleeve)
-
5.
History of major surgery within three months of enrollment
-
6.
Type 1 diabetes
-
7.
Renal insufficiency consisting of potassium over 5.5 (mmol/L) on a non-hemolyzed specimen, or a creatinine over 2.5 mg/dL
-
8.
Bilirubin over 3 (mg/dL) or an albumin less than 3 (g/dL)
-
9.
ALT > 3 (IU/L) times the upper limit of normal (normal range is 7–56)
-
10.
Evidence of more than 1 severe hypoglycemic event (episode requiring emergency medical services) in the past 12 months, unless the participant’s treating physician provides written clearance for participation
-
11.
Hemoglobinopathy that interferes with measurement of hemoglobin A1c
-
12.
Those on higher doses of diuretics (furosemide 40 mg or higher or comparable)
-
13.
Unstable heart disease (an ongoing workup or treatment for a cardiac symptom such as unstable angina, coronary ischemia)
-
14.
Presence of implanted cardiac defibrillator
-
15.
Blood pressure ≥180/100 mm Hg. If a potential participant has a BP above the inclusion criteria it is acceptable to re-test this potential participant within one week of the original test
-
16.
Thyroid disease for which the participant is untreated or has had treatment changed within the last 6 months. History of thyroid disease or current thyroid disease treated with a stable medication regimen for at least 6 months is acceptable
-
17.
Orthopedic limitations that would interfere with ability to engage in regular physical activity
-
18.
Uncontrolled gastrointestinal disorders including chronic malabsorptive conditions, peptic ulcer disease, Crohn’s disease, chronic diarrhea or active gallbladder disease
-
19.
Current cancer or cancer treatment, or a history of cancer or cancer treatment within the last 3 years. Persons with successfully resected non-melanoma carcinoma of the skin may be enrolled
-
20.
Dementia, psychiatric illness, or substance abuse that may interfere with adherence (e.g. illness that is currently unstable or resistant to first-line therapy; substance abuse in the past year)
-
21.
History of clinically diagnosed eating disorders including anorexia nervosa or bulimia nervosa
-
22.
Women who are pregnant, lactating, trying to become pregnant or unwilling to use an effective means of birth control
-
23.
Currently consuming >14 alcoholic drinks (1 drink = 12 fl oz beer, 4 fl oz wine or 1.5 fl oz liquor) per week and unwilling to limit intake to less than 3 drinks per drinking day during study participation
-
24.
Participation in another clinical trial within 30 days prior to enrollment
-
25.
Any other condition or factor which in the opinion of the study physician or investigator makes it inadvisable for the candidate to participate in the trial
As noted, specific drug exclusion criteria are as follows: (1) Anti-obesity medications (prescription or OTC weight loss medications) in the last 4 weeks including bupropion-naltrexone, liraglutide, phentermine, phentermine-topiramate, and orlistat. (GLP-1 Antagonists were acceptable in the lower dose for diabetics (if stable dosage for >3 months) but not at the higher dose which is FDA-approved for weight loss. (Tirzepatide was not FDA approved until May 2022, so no participants were taking the medication.) and (2) Diuretics with a dose that exceeds 40 mg (furosemide 40 mg or higher or comparable such as bumetanide, ethacrynic acid, and torsemide).
Participants completed 3 study visits including the screening/baseline visit and follow up visits at 12 (midpoint) and 24 weeks (post). All follow-up visits should have occurred within a ±7-day window but could occur within ±14 days. Participants were recruited in cohorts that ranged from 7 to 31 participants (mean n = 17) between April– June of 2021. Height, demographics, and medical history were assessed at screening/baseline. Key outcome measures included HbA1c, weight, waist circumference, blood pressure, diabetes distress (Diabetes Distress Scale; DDS) [12] and Impact of Weight on Quality of Life – Lite (IWQOL-L) [13], and hunger [14, 15].
For HbA1c, whole blood was collected via venipuncture. Trained research personnel measured height at baseline and body weight using a standardized digital weight scale, with participants wearing light clothing and shoes removed. Height was measured using a standardized height dynamometer. Waist circumference was measured in a horizontal plane around the abdomen at the level of the iliac crest. An average of the two closest measurements will be used for analyses. For height, weight, and waist circumference, measurement was performed twice, with a third measurement if the first 2 measurements deviate more than 0.5 cm or 0.5 kg.
The DDS is a measure of diabetes-related distress [12]. It consists of 17 items scored on a 1–6 scale, with higher scores indicating higher distress. The DDS comprises four subscales (emotional burden, physician-related distress, regimen-related distress, and interpersonal distress) and a total score. A total or subscale score > 2.0 (moderate distress) is considered clinically significant [16].
The IWQOL-L is a self-report measure of quality of life [13]. This measure is distinct from other measures of quality of life because it addresses this concept as it specifically relates to individuals with obesity. There are 31 items rated on a 1 (Never True) to 5 (Always True) point Likert scale with higher scores indicating more distress and a poorer quality of life. Five subscales are derived: physical function, self-esteem, sexual life, public distress, and work.
Retrospective Visual Analogue Scale (VAS) was used to measure average ratings of hunger that participants experienced over the past week. This method of collecting VAS data has been found to be consistent with daily assessments of satiety [15], and support has been found for the reliability and validity of VAS for measuring subjective states related to energy intake [14].
Following baseline assessments, eligible participants received the T2D-tailored WW program, which included access to weekly virtual workshops, weekly check-ins, the WW App, and a private online community. The program encouraged healthy habits with topics specific to T2D in the areas of food, activity, mindset, and sleep.
The intervention was delivered weekly via virtual group workshops. Each workshop lasted 30–60 min and included a new topic related to building healthy habits, behavioral skills to support behavior change, and group discussion. Through the WW app and website, participants were able to track their weights, dietary intake, physical activity; access progress reports; and complete weekly check-ins. The app also provided recipes, behavior change content, and T2D-specific information.
The core of the WW food program is the SmartPoints® system which assigns each food and beverage a SmartPoints® value per portion based on calories, protein, fiber, added sugar, saturated fat and unsaturated fat. In addition, foods that form the foundation of a healthy dietary pattern as recommended by the 2020–2025 Dietary Guidelines and global food-based dietary recommendations. (e.g., fruit, vegetables, whole grains, lean proteins) are assigned a SmartPoints® value of zero (ZeroPoint foods) and do not need to be measured or tracked. Based on glycemic control, foods higher in carbohydrates (e.g., fruit, yogurt, whole grains), are no longer ZeroPoints encouraging people with diabetes to weigh, measure, and track them. Participants were encouraged to focus on other zero point foods, such as lean proteins (e.g. skinless chicken and turkey breast), high fiber legumes (e.g. beans and peas), and healthy fats (e.g. avocado) that were lower in carbohydrates and/or higher in protein and less likely to impact blood sugar. Participants were provided with a personalized daily and weekly SmartPoints® budget which was designed to create an energy deficit of ~750 kcal/day using the Mifflin St Jeor equation [17].
Statistical analyses
Analyses adhered to the intent-to-treat principle; missing data were accounted for using maximum likelihood estimation. General linear mixed effect models adjusted for sex were used to evaluate changes over time in HbA1c and secondary outcomes (including percent change) at baseline, 12, and 24 weeks. Results are presented as mean±standard errors or overall percentages. Testing of differences employed either T-tests, or Chi-squared tests for percentages. Planned exploratory analysis looked at HbA1c subgroups of >8% or ≤8% HbA1c.
Power calculation
The sample size calculation was based on a previous WW study with T2D participants that found a 0.6 ± 1.4% decrease in HbA1c at 6 months [11]. To ensure adequate power, we anticipated a HbA1c reduction that was 20% less with a 20% higher standard deviation than previously found (0.5 ± 1.7%). The level of significance was 0.05 and was based on a two-sided one-sample t-test. Thus, a sample size of 120 subjects provided 90% power to detect this difference.
Results
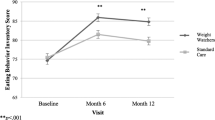
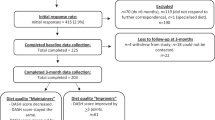
The flow of participants from initial screening through week 24 is shown in Fig. 1. Baseline characteristics and changes at 12 and 24 weeks are described in Table 1. HbA1c significantly decreased (0.75 ± 0.11%) at week 24. Also, those with an initial HbA1c ≥ 8 (8.95 ± 0.12%, 99.65 ± 1.87 kg; n = 52), significantly reduced HbA1c by 1.35 ± 0.16 (p ≤ 0.0001) and body weight by −4.97 ± 0.58% (p ≤ 0.0001), while those <8 (7.41 ± 0.10%, 103.45 ± 2.37 kg; n = 84) significantly reduced A1c by 0.38 ± 0.13 at 24 weeks (p = 0.0026) and body weight −5.31 ± 0.75% (p ≤ 0.0001). Body weight significantly decreased by 5.10 ± 0.46 kg (5.74 ± 0.50%) at week 24. The percent of participants who achieved ≥3%, ≥5%, and ≥10% weight loss at week 24 was 60%, 43%, and 15%, respectively. Participants experienced significant reductions in waist circumference (5.91 ± 0.53 cm) and diastolic blood pressure (2.8 ± 1.0 mmHg) at week 24. At week 24, there were significant reductions in overall DDS score (0.33 ± 0.10) as well as the Emotional Burden (0.33 ± 0.12) and Regimen Related Distress (0.62 ± 0.13) subscales, respectively (Table 2). Further, there were significant reductions in the IWQOL-L overall and in all subscales at week 24. Lastly, at week 24, hunger was significantly reduced (14 ± 2).
Discussion
The WW virtual weight loss and wellness program tailored for diabetes resulted in HbA1c reductions and improvements in diabetes distress similar to in-person trials [5, 18]. Of note, the >0.5% reduction in HbA1c, >5% weight loss, and decrease in DDS to from moderate to low levels of diabetes-related distress are all clinically meaningful [19,20,21]. The observed HbA1c reduction compares favorably to in-person community-based approaches, two of which included portion controlled meals [22, 23] as well as an earlier version of WW that included at least 2 individual sessions with a certified diabetes care and education specialist [11]. A recent meta-analysis found that more intensive interventions promoted reductions in HbA1c and DDS to those found in the current trial [24]. Another area of potential future study may be to evaluate the programming in comparison or addition to accredited diabetes self-management education/training (DSME/T).
Limitations
These promising results await replication in a randomized controlled trial. While it is possible that the observed effects were simply a regression to the mean, the baseline A1c of <8 suggest this is less likely. It is also unlikely that a nearly 6% weight loss occurs spontaneously in the absence of a structured weight management program.
Conclusion
A T2D-tailored virtual, widely available weight management program produced favorable change in glycemic control, weight, diabetes distress, quality of life, and CVD risk factors among people living with T2D and overweight/obesity. Moreover, since this program has been shown to be a cost-effective weight loss program [8] and is delivered via a digital platform, it has the potential to mitigate access and affordability barriers for adults with T2D seeking weight management to improve their glycemic control and other CVD risk factors.
References
Wright AK, Kontopantelis E, Emsley R, Buchan I, Sattar N, Rutter MK, et al. Life expectancy and cause-specific mortality in type 2 diabetes: a population-based cohort study quantifying relationships in ethnic subgroups. Diabetes Care. 2017;40:338–45.
American Diabetes A. Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917–28.
American Diabetes A. Economic costs of diabetes in the U.S. in 2012. Diabetes Care. 2013;36:1033–46.
American Diabetes Association Professional Practice C, Draznin B, Aroda VR, Bakris G, Benson G, Brown FM, et al. 8. Obesity and weight management for the prevention and treatment of type 2 diabetes: standards of medical care in diabetes-2022. Diabetes Care. 2022;45:S113–24.
Group LAR, Pi-Sunyer X, Blackburn G, Brancati FL, Bray GA, Bright R, et al. Reduction in weight and cardiovascular disease risk factors in individuals with type 2 diabetes: one-year results of the look AHEAD trial. Diabetes Care. 2007;30:1374–83.
Wilding JP. The importance of weight management in type 2 diabetes mellitus. Int J Clin Pract. 2014;68:682–91.
Porter GC, Schwab R, Hill JL, Bartee T, Heelan KA, Michaud TL, et al. Examining the feasibility and characteristics of realistic weight management support for patients: focus groups with rural, micropolitan, and metropolitan primary care providers. Prev Med Rep. 2021;23:101390.
Finkelstein EA, Kruger E. Meta- and cost-effectiveness analysis of commercial weight loss strategies. Obesity. 2014;22:1942–51.
Webb VL, Wadden TA. Intensive lifestyle intervention for obesity: principles, practices, and results. Gastroenterology. 2017;152:1752–64.
Chaudhry ZW, Doshi RS, Mehta AK, Jacobs DK, Vakil RM, Lee CJ, et al. A systematic review of commercial weight loss programmes’ effect on glycemic outcomes among overweight and obese adults with and without type 2 diabetes mellitus. Obes Rev. 2016;17:758–69.
O’Neil PM, Miller-Kovach K, Tuerk PW, Becker LE, Wadden TA, Fujioka K, et al. Randomized controlled trial of a nationally available weight control program tailored for adults with type 2 diabetes. Obesity. 2016;24:2269–77.
Polonsky WH, Fisher L, Earles J, Dudl RJ, Lees J, Mullan J, et al. Assessing psychosocial distress in diabetes: development of the diabetes distress scale. Diabetes Care. 2005;28:626–31.
Kolotkin RL, Crosby RD, Kosloski KD, Williams GR. Development of a brief measure to assess quality of life in obesity. Obes Res. 2001;9:102–11.
Flint A, Raben A, Blundell JE, Astrup A. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int J Obes Relat Metab Disord. 2000;24:38–48.
Womble LG, Wadden TA, Chandler JM, Martin AR. Agreement between weekly vs. daily assessment of appetite. Appetite. 2003;40:131–5.
Fisher L, Hessler DM, Polonsky WH, Mullan J. When is diabetes distress clinically meaningful?: establishing cut points for the Diabetes Distress Scale. Diabetes Care. 2012;35:259–64.
Mifflin MD, St Jeor ST, Hill LA, Scott BJ, Daugherty SA, Koh YO. A new predictive equation for resting energy expenditure in healthy individuals. Am J Clin Nutr. 1990;51:241–7.
Holland-Carter L, Tuerk PW, Wadden TA, Fujioka KN, Becker LE, Miller-Kovach K, et al. Impact on psychosocial outcomes of a nationally available weight management program tailored for individuals with type 2 diabetes: Results of a randomized controlled trial. J Diabetes Complications. 2017;31:891–7.
Lenters-Westra E, Schindhelm RK, Bilo HJ, Groenier KH, Slingerland RJ. Differences in interpretation of haemoglobin A1c values among diabetes care professionals. Neth J Med. 2014;72:462–6.
Little RR, Rohlfing CL. The long and winding road to optimal HbA1c measurement. Clin Chim Acta. 2013;418:63–71.
Jensen MD, Ryan DH, Apovian CM, Ard JD, Comuzzie AG, Donato KA, et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. J Am Coll Cardiol. 2014;63:2985–3023.
Foster GD, Wadden TA, Lagrotte CA, Vander Veur SS, Hesson LA, Homko CJ, et al. A randomized comparison of a commercially available portion-controlled weight-loss intervention with a diabetes self-management education program. Nutr Diabetes. 2013;3:e63.
Rock CL, Flatt SW, Pakiz B, Taylor KS, Leone AF, Brelje K, et al. Weight loss, glycemic control, and cardiovascular disease risk factors in response to differential diet composition in a weight loss program in type 2 diabetes: a randomized controlled trial. Diabetes Care. 2014;37:1573–80.
Mathiesen AS, Egerod I, Jensen T, Kaldan G, Langberg H, Thomsen T. Psychosocial interventions for reducing diabetes distress in vulnerable people with type 2 diabetes mellitus: a systematic review and meta-analysis. Diabetes Metab Syndr Obes. 2019;12:19–33.
Acknowledgements
We are grateful for the PBRC cores including intervention resources (IR), clinical chemistry, and medical records for assisting with the completion of this work.
Funding
This work was funded by WW International, Inc.
Author information
Authors and Affiliations
Contributions
JWA, JGL, SDA, FLG, TB, and GDF contributed to the development of the study concept and design. All authors contributed to data acquisition and interpretation. RAB performed the analysis. JWA and MIC contributed to drafting of the manuscript. All authors contributed to critical revision of the manuscript of important intellectual content. JWA is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Corresponding author
Ethics declarations
Competing interests
JWA: Grants NIH NIDDK and NIMHD, NSF, and USDA. JGL: Grants NIH NIDDK. FLG: Consultant: Ed Med Resources, NovMeta Pharma, GNC, Basic Research, Scientific Advisory Board: Altimune, Pfizer, Nutraceutical Corporation, Stock/stock options: UR Labs, Plensat, Ketogenic Health Systems, Slim Health Nutrition, Energesis, Rejuvenate Bio, Patents: Melior Discoveries (Tolimidone/menthol). Grants: NovMeta Pharma. GDF: Employee and Shareholder at WW. MIC: Grants: National Institute of Health NHLBI and NIDDK. Stock/Stock Options: WW International, Inc. Employee at WW International, Inc. The authors declare no other competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Apolzan, J.W., LaRose, J.G., Anton, S.D. et al. A scalable, virtual weight management program tailored for adults with type 2 diabetes: effects on glycemic control. Nutr. Diabetes 13, 3 (2023). https://doi.org/10.1038/s41387-023-00234-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41387-023-00234-6