Abstract
The interaction between social factors and alcohol addiction is complex, with potential for both positive and negative contributions to drug use and abstinence. Positive social connections are an important component in successful abstinence, and yet the social context of alcohol use can also lead to relapse. Recently it was shown that rats overwhelmingly choose social reward over methamphetamine, cocaine, and heroin in a discrete choice procedure, and that prolonged choice for social reward attenuates incubation of drug craving. The extent to which this effect generalises to rats trained to self-administer alcohol is not known. In this study we aimed to test the effect of social reward on choice for alcohol in male and female rats. We first validated social reward self-administration in both male and female Long-Evans rats, and found that 60 s access to a social partner of the same sex can serve as an operant reinforcer. Next we trained rats to self-administer both social reward and alcohol (20% ethanol in water), and then used discrete choice trial based tests to determine whether there is a choice preference for alcohol or social reward. Our main finding is that both male and female rats showed persistent choice for alcohol over social reward, with only minor differences between the sexes. We also show that choice for alcohol could be reduced via increased response requirement for alcohol, pre-choice alcohol exposure, and also decreasing the alcohol percentage. This study shows that preference for social rewards over drugs may not generalise to rats self-administering alcohol, and we describe several conditions where choice for social reward can be developed. This study highlights the important contribution of social factors to alcohol abuse, and future studies can investigate the neurobiology underlying a shift in preference from alcohol to social rewards.
Similar content being viewed by others
Introduction
The interaction between social factors and drug and alcohol addiction is complex, with potential for both positive and negative contributions to drug use and abstinence. Positive social connections are an important component in successful abstinence. For example, community-reinforcement approaches can be effective for achieving abstinence with social support from clinicians, family, colleagues and friends [1, 2]. However, the social context of alcohol use can also promote drug use and relapse. For example, social exclusion is a strong predictor of abuse liability [3], and certain social situations can increase stress leading to increased drug use [4]. Studying the interaction between social factors and alcohol addiction has the potential to reveal important moderating mechanisms for alcohol use disorder [5].
To study the protective effects of social factors on drug addiction, researchers have leveraged the fact that interaction with peers is reinforcing in rats [6]. Venniro et al. found that rats overwhelmingly choose social reward over methamphetamine in a discrete choice procedure, and that prolonged choice for social reward attenuates incubation of methamphetamine craving [7], heroin [8], and cocaine [9]. These studies have also found that rats that retain the ability to demonstrate deliberative choice for social reward despite also showing highly addictive behaviours such as escalation of drug use, resistance to punishment, and increased responding for drug despite non-reinforcement. This indicates that there is a profound protective effect of alternative choice for social reinforcement.
Currently, the role of discrete choice for social rewards in rats trained to self-administer alcohol has not been tested. Building on the foundational choice studies in rodents testing choice for cocaine versus sugar [10, 11], others have shown that rats typically prefer saccharine over alcohol, but that there is substantial variability [12]. Preference for saccharine can be reversed by alcohol dependence [13] and lesion of the subthalamic nucleus [14]. In this study we aimed to determine whether male and female rats will choose social reward over alcohol in a discrete choice paradigm. To our surprise, we found evidence for a strong preference for alcohol reward (20% ethanol in water) over social reward. We tested three different manipulations to determine whether this preference can be changed. In one group of rats, we increased the response requirement for alcohol to determine whether choice would shift to social reward as the amount of effort required for alcohol increases. In another group, we tested the effect of an alcohol self-administration session prior to choice, and the then tested the effect of decreasing the percentage of alcohol. Overall, the preference for alcohol over social reward was highly prevalent, with only minor differences between the sexes. This study shows that preference for social rewards over drugs may not generalise to rats self-administering alcohol, but that there are conditions where the choice for social reward can be developed. The results are discussed with relevance to the translational utility of this model.
Materials and methods
Subjects
We obtained 56 Long-Evans rats (28 male and 28 female), aged 10–12 weeks upon arrival, from Janvier, France. In compliance with Dutch law and Institutional regulations, all animal procedures were approved by the Centrale Commissie Dierproeven (CCD) and conducted in accordance with the Experiments on Animal Act. Experiments were approved by the local animal welfare body Animal Experiments Committee of the Vrije Universiteit, Amsterdam, The Netherlands. Behavioural tests were conducted during the dark phase of the rat’s diurnal cycle (12 h/12 h). Food and water were available ad libitum. In experiment 1 we used 24 rats (8 M/8 F experimental, 4 M/4 F social partners). In experiment 2 we used 32 rats (12 M/12 F experimental, 4 M/4 F social partners), but one male experimental rat was excluded during the experiment due to health issues.
Apparatus
Alcohol
We prepared 20% ethanol by diluting 70% ethanol (VWR International) with water. Both water and alcohol were prepared in standard home-cage water bottles.
Behavioural procedure
Experiment 1
Assessment of social reward as an operant reinforcer (2 Phases) (See Supplementary Methods for in depth details):
Housing conditions
Phase 1: Social reward self-administration
We first placed the social partner rat into the adjacent social holding component of the operant chamber, and then the experimental rat (of the same sex) was placed into the main component of the operant chamber. We trained rats in a 2 h session with the social-lever inserted into the chamber throughout. A single lever press on the social reward lever resulted in the opening of the guillotine door for 60 s, and the red cue-light above on for first 20 s.
Phase 2: Testing whether social reward self-administration requires the social partner and guillotine door opening
In the following 3 weeks we tested one of three different consequences: (1) Door/No_Partner (+/−): the door opening but the social partner not present; (2) No_Door/Partner (−/+): the door not opening, but the social partner is present; (3) No_Door/No_Partner (−/−): the door is not opening and the social partner is not present. These sessions were recorded for video analysis of the time spent in an area close to the social partner, the lever, or the remaining area on the other side of the chamber using EZTrack [15].
Experiment 2
Choice between alcohol and social reward (5 phases) (See Supplementary Methods for in depth details):
Housing conditions
Phase 1: Alcohol home-cage intermittent every other day access
We used an intermittent access (3–4 times/week) alcohol procedure [16, 17] in which rats received 12 × 24 h sessions of access to one bottle of 20% alcohol and one water bottle.
Phase 2
Self-administration (Social and alcohol) and initial choice tests.
Phase 2a: Social self-administration
See experiment 1.
Phase 2b: Alcohol self-administration
We first gave 1 h alcohol magazine training session with alcohol reward (0.1 mL) every 3 min (20 total), which coincided 20 s alcohol-associated cue (no lever press). Next we trained the rats to lever press for alcohol reward (FR1) for a total of 15 × 1 h sessions. The alcohol lever was inserted into the chamber throughout, and one lever press resulted in 0.1 ml alcohol, and 20 s alcohol cue. There was also a 20 s time out after the reinforced lever press where lever press resulted in no alcohol infusion.
Phase 2c: Choice tests
We tested choice between alcohol and social reward twice during the alcohol self-administration phase.
Alcohol seeking test (“d1”)
We tested alcohol seeking in a 60 min extinction test, where only the alcohol lever was inserted into the chamber. No alcohol was delivered but responses on the alcohol-paired lever were reinforced with 20 s presentation of the alcohol-paired cue.
Phase 3: Manipulation of alcohol FR during choice (19 sessions)
Throughout this phase the ratio requirement (FR) for social reward was 1. During the first four sessions alcohol was FR1, in the fifth session it was FR2, and we subsequently increased the FR for alcohol by 2 for every session until FR30.
Alcohol seeking test (“d21”)
We tested alcohol seeking again in a 60 min extinction test, identical conditions to the “d1” test.
Social seeking test (“d22”)
We tested social seeking in a 60 min extinction test, where only the social lever was inserted into the chamber. No social partner was present, and the door did not open, but responses on the social-paired lever were reinforced with 20 s presentation of the social-paired cue.
Phase 4: Pre-choice alcohol exposure prior to choice (8 sessions)
The first 15 min of the session were identical to alcohol self-administration (Phase 2b). After this, there was an 8 min time-out period and the choice test ran for another 2 h, using an identical design to Phase 2c.
Phase 5: Manipulation of alcohol percentage during choice (17 sessions)
During this phase we trained the rats on the choice test procedure identical to Phase 2c. We first used the standard 20% ethanol for 3 sessions, then 10% ethanol for 3 sessions, then 5% ethanol for 3 sessions, then 1% ethanol for 3 sessions, then 0% ethanol (i.e. water) for 2 sessions, then we returned to 20% ethanol for the final 3 sessions.
Experiment design
For the first experiment, all rats were given the same design, and there were no experimental groups. For the second experiment, after the self-administration phase we split the experimental rats into two groups because our original rationale was to test the effect of choice-imposed abstinence on incubation of alcohol seeking. One group of 12 rats (6 M, 6 F) went immediately into Phase 3 (Manipulation of alcohol FR), while the second group of 11 rats (5 M, 6 F) were kept in the home-cage for this period. At the end of Phase 3, we tested both groups for alcohol seeking in a non-reinforced test, and the next day tested social reward seeking in a non-reinforced test, and then gave all rats another social reward self-administration session (identical to Phase 2b). Following this, group 1 was no longer trained, and group 2 underwent phase 4 and Phase 5 in that order.
Statistics
Results
Experiment 1: Assessment of social reward as an operant reinforcer
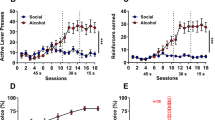
Fig. 1A shows the outline of Experiment 1, and the data from the initial social self-administration (Fig. 1B) and tests where we manipulated the presence of the social partner and the door opening (Fig. 1C). See Supplementary Results for analysis of the self-administration data. On the test sessions we used the within-subjects factors Door (Open, Closed) and Social Partner (Present, Not Present) and found a main effect of Door (F (1,14) = 69.9; p < 0.001) and Social Partner (F (1,14) = 4.7; p < 0.05). The interaction was not significant, but there was a main effect of Sex (F (1,14) = 6.8; p < 0.05).
A Outline of experiment 1. Phase 1: Rats were first trained to self-administer social access in 2 h sessions on an FR1 schedule of reinforcement. Phase 2: We then tested the effect of removing the response-contingent opening of the door, or the presence of the social partner in 1 h sessions. (+/+, Door opening, Social partner present; +/− Door opening, Social partner absent; −/+ Door closed, Social partner present; −/− Door closed, Social partner absent). B Mean ± SEM total rewards (left) and lever presses (right) during the social self-administration sessions. C Mean ± SEM, and individual data of total reinforced active lever presses during the four different test conditions. D Mean ± SEM, and individual data, time spent in the three different areas of the chamber during the four different test conditions.
Analysis of the video recordings (Fig. 1D) revealed two important findings. First, during standard social self-administration (i.e. Door Open, Partner Present: +/+), males spend on average 64.9%, and females 76.9% of the 60 s in close proximity to the social partner. ANOVA revealed a significant difference (F (1,14) = 8.6; p = 0.011), indicating females spend more time interacting with the social partner. Interestingly males spend significantly more time in the lever area during this period (F (1,14) = 24.2; p < 0.001). The second is a comparable pattern of results in the amount of time spent in the social area to the reinforced active lever presses (Fig. 1C). We found a main effect of Door (F (1,14) = 146; p < 0.001) and Social Partner (F (1,14) = 22.8; p < 0.05). The interaction was not significant, and there was a main effect of Sex (F (1,14) = 13.6; p < 0.01). Follow-up comparisons revealed a significant difference in time spent in the social area between the standard condition (i.e. Door Open, Partner Present: +/+) and all other conditions (ps < 0.05) in males. In female rats the comparison between the standard condition (+/+) and the door opening but no social partner condition (+/−) was not significant (p > 0.05), and the other two (−/+; −/−) were (ps < 0.001). See Supplementary Results for more comparisons.
These data show that, compared to the rate of responding during social self-administration, omitting both the door opening (cue still presented) and the social partner resulted in a significant decrease in the total number reinforced active lever presses, and significantly less time spent in the area of the chamber close to the guillotine door.
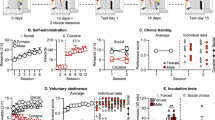
Experiment 2: Phase 2, self-administration phase and first 2 choice sessions
Fig. 2A describes the outline of experiment 2, and Fig. 2B describes the choice session procedure. See Supplementary Results for the analysis of the alcohol home-cage data and initial social and alcohol self-administration data. During the initial choice sessions (C1, C2; Fig. 3B), we found a significant effect of Choice (F (1,21) = 5.8; p < 0.05), and a significant Choice × Sex interaction (F (1,21) = 12.8, p < 0.01), but no effect of Session (F (1,21) = 3.9; p > 0.05). This reflects greater preference for alcohol in male rats compared to female rats, with no change in preference from C1 to C2. Statistical analysis on the preference score revealed no effect of Session (F (1,21) < 1; p > 0.05), and an overall effect of Sex (F (1,21) = 13.7; p = 0.001), again reflecting greater preference for alcohol in male rats compared to female rats. In addition, we found no difference between the two groups (home-cage, Alcohol FR manipulations) in their preference in C1 and C2 (Group (F (1,19) < 1; p > 0.05); Session × Group (F (1,19) = 2.2; p > 0.05).
A All rats were given home-cage access to alcohol for 4 weeks in group housing. Afterwards they were single-housed and operant training began, with separate training sessions for social and alcohol operant reward. Two choice tests were conducted (C1, C2) during the alcohol self-administration phase. We then tested alcohol seeking in a single extinction test session. Then one group (n = 12) were given successive choice sessions where we increased the alcohol FR, while the other group (n = 11) was first left in the home-cage during this time. We then tested both groups of rats for both alcohol and social reward seeking in extinction test session. After this, group 2 underwent the pre-choice alcohol exposure phase, and then the alcohol percentage manipulation phase. B Each choice session consisted of 15 × 8 min trials. A trial begins with onset of the discriminative stimuli (DS) for 10 s and then both levers are inserted. After the response requirement is met, both levers are retracted, both DS are turned off, and the relevant reward is delivered. If no response is made in 6 min then both levers are retracted, both DS are turned off, and there is an inter-trial-interval (ITI) of 1 min 50 s.
A The top row of graphs show the Mean ± SEM rewards (either 60 s social access or 0.1 mL alcohol) on the left y-axis, and for the alcohol self-administration sessions, g/kg per session is shown on the right y-axis. The bottom row of graphs show the Mean ± SEM total lever presses during these sessions. B Both the group data and individual data of the preference score from C1 and C2.
Experiment 2: Phase 3, alcohol FR manipulations
Figure 4A, B show the responses during the choice sessions where the FR requirement for alcohol increasing by 2 per session after the fifth session. During the FR1 choice sessions, male rats showed a higher preference for alcohol over social reward. While female rats showed no preference initially, preference shifted to alcohol by the fourth session. Statistical analysis across the sessions revealed a significant Response (Alcohol, Social) x Session interaction (F (18,180) = 16.8, p < 0.001), reflecting a change in choice behaviour in response to increased alcohol FR. Figure 4C shows the preference score for male and female rats in the FR1 and FR26–FR30 sessions. Statistical analysis revealed a significant effect of Session (F (1,10) = 24.6; p < 0.001), but no significant Session × Sex interaction (F (1,10) < 1; p > 0.05). Post-hoc test revealed significant difference within both male (p = 0.0059) and female (p = 0.0218) rats. These data indicate a significant change in preference as the FR requirement for alcohol increased, and that both male and female rats changed their preference in a comparable manner.
Choice responses during the increasing fixed ratio (FR) requirement for alcohol sessions are shown for male rats (A) and female rats (B). Choice response data are presented as Mean ± SEM. C Average preference scores for individual rats from the initial four FR1 sessions, and last three high FR sessions (FR26–FR30).
We also conducted correlation analyses between the Preference Score at low and high FR requirement for alcohol with measures of social or alcohol reward prior to choice to determine the relationship between these measures (see Supplementary Results).
Experiment 2: Alcohol and social reward seeking tests
See Supplementary Results for analysis of these tests. In summary, these results show evidence of a comparable rate of alcohol and social reward seeking during extinction tests.
Experiment 2: Phase 4, pre-choice alcohol exposure manipulations
Figure 5A, B show responses during the choice sessions from the pre-choice alcohol exposure sessions where we gave the rats 15 min of unrestricted alcohol self-administration (FR1) prior to each choice session. See Supplementary Results (Fig. S3C) for analysis of alcohol self-administration during these sessions. Figure 5C shows the mean preference score in the first 3 and last 3 pre-choice alcohol exposure choice sessions. We observed a significant effect of Session (F (1,9) = 8.1; p < 0.05), and a significant Session x Sex interaction (F (1,9) = 7.8; p < 0.05). Post-hoc test revealed significant difference between male (p = 0.0081) but not female (p > 0.05) rats. Overall, these results show that pre-choice alcohol exposure reduces choice for alcohol. This effect was sex dependent, female rats choice remained unbiased towards either option, but male rats showed a significant change in preference from alcohol preferring to social preferring.
Choice responses during the pre-choice alcohol sessions are shown for male rats (A) and female rats (B). C Average preference scores from the initial three pre-choice alcohol exposure sessions, and last three pre-choice alcohol exposure sessions. Choice responses during the decreasing alcohol percentage sessions are shown for male rats (D) and female rats (E). F Average preference scores from the initial three 20% alcohol sessions, and two zero percent alcohol sessions. Choice response data are presented as Mean ± SEM.
Experiment 2: Phase 5, alcohol percentage manipulations
Figure 5D, E show the responses during the choice sessions from the alcohol percentage manipulations sessions. To make reasonable interpretations of the statistical output, we only analysed sessions 1–14, where the alcohol percentage changes from 20% in sessions 1–3 to 0% in sessions 13 and 14. We show data from sessions 15–17, when alcohol percentage is returned to 20%, to illustrate the return of alcohol preference in both male and female rats. Analysis of the raw choice data revealed a significant Choice x Session interaction (F (13,117) = 7.6; p < 0.001) reflecting a change in choice for alcohol and social reward as the percentage of alcohol is reduced to 0% across the sessions. We observed no Choice × Session × Sex interaction (F (13,117) = 1.1; p > 0.05) reflecting no difference in choice behaviour across the sessions between the male and female rats. Figure 5F shows the mean preference score per subject in the initial three 20% sessions and the two 0% alcohol choice sessions. We observed a significant effect of Session (F (1,9) = 8.8; p = 0.015), but no Session x Sex interaction (F (1,9) < 1; p > 0.05), nor main effect of Sex (F (1,9) < 1; p > 0.05). Thus, we did find a significant change in preference in response to decreasing percentage of alcohol.
Discussion
In this study we show a strong preference for alcohol over social reward in both male and female rats. We first show that a social partner serves as an operant reinforcer. Next, we used a discrete choice [10] procedure and found that male and female rats show a choice preference for alcohol reward over social reward. We then tested three different manipulations to determine whether this preference can be changed. First we show that increasing the FR for alcohol, while social reward remains at FR1, decreased choice for alcohol. In another group of rats, we tested the effect of pre-choice alcohol exposure on choice, and found that male rats preference changed to social reward, while female rats choice for alcohol was reduced but there was no preference for social reward. Finally, we also reduced the percentage of alcohol over successive sessions, and found that both male and female rats reduced alcohol choice. Overall, this study shows that in a discrete choice procedure rats prefer alcohol over social reward. These findings suggest that choice for social reward over a drug alternative may not generalise to alcohol.
Effectiveness of a social partner to act as an operant reinforcer
In Experiment 1 we show that social reward self-administration is reduced when the social partner is not present and the guillotine does not open. These data demonstrate the importance of the presence of the social partner in this form of operant responding. However, surprisingly there was no reduction in the +/− condition (Fig. 1C). This finding is in contrast to a recent study showing a significant decrease in responding when the social partner is not present [18]. One possible explanation for this is that the door opening acts as a conditioned reinforcer, and in the three sessions we tested this behaviour did not extinguish. However we did not test this in naïve, non-trained rats. Another possibility is the presence of olfactory cues within the social box. We did not clean out this chamber between sessions, and emerging evidence (Marco Venniro, personal communication) indicates an important contribution of residual olfactory stimuli in social-reinforced behaviours.
We also observed a difference in the rate of social reward self-administration in male rats between Experiment 1 (Fig. 1B) and Experiment 2 (Fig. 3A). Although the experiments were not directly compared, we did find a significant difference between male and female rats in social reward self-administration in Experiment 2, but not Experiment 1. These cohorts were run separately, although under comparable conditions, as such we are unsure if this is a cohort effect. However, one critical difference between the experiments is the alcohol home-cage prior to social reward self-administration training in Experiment 2. Therefore it is possible this had a more significant impact of operant social reward in male rats than female rats. It remains an open and interesting question for future research as to whether alcohol exposure changes the propensity for social reinforcement in male rats.
Apparent lack of protective effect of social reward on alcohol self-administration
The most unexpected finding from this study is that both male and female rats choose alcohol over social reward. Although there is evidence in experiments with humans indicating that in a choice setting, drugs are often chosen over alternative rewards [19, 20], recent pre-clinical work has found that rats prefer social reward over methamphetamine, heroin, and cocaine [7,8,9]. Moreover, other alternative, non-social, rewards have been tested such as sugar, saccharine, and food pellets, and in each case these studies found that the majority of rats show a preference for the non-drug reward over drug [10, 21]. A comparable finding is also seen in rats self-administering alcohol, where the majority (80%) of rats were found to choose saccharine over alcohol in a discrete choice procedure [12]. However, in contrast to these findings, recently it was shown that rats will work harder to receive a food reward compared to a social reward [18, 22], and rats choose food pellets over social reward [22]. That rats show a choice preference for both food pellets and alcohol over social reward may indicate that orally consumed rewards are contrasted differentially to intravenous rewards in a choice setting with social reward.
It is possible that our implementation of social reward is not comparable to the experiments of Venniro et al. [7]. Our experiments were designed to replicate these studies however there were some important differences, such as the home-cage alcohol phase, and the social partners in our experiment were group housed. In experiment 1 we show that both male and female rats reduce their rate of social reward seeking when the social partner is not present and the guillotine door does not open. In experiment 2 we tested both alcohol and social reward seeking in extinction tests, and found a comparable rate of reward seeking. This observation provides some evidence that social reward is sufficiently motivating to yield a rate of seeking in extinction comparable to alcohol.
There are two possible experimental design factors that may have increased the value of alcohol relative to social reward in this study. The first is the 4 weeks of home-cage intermittent every other day access to alcohol [16], which may be sufficient to bias preference for alcohol over social reward. Although even longer periods of home-cage alcohol access are typically needed to develop ‘addiction-like’ phenotypes [23,24,25]. Consistent with this, repeated intermittent exposure to alcohol vapour, causing physical dependence, can shift preference in discrete choice to alcohol away from saccharine [13]. Venniro et al. showed that over 50 days of intravenous methamphetamine self-administration, which produced a 3 criteria distribution of rats [26], did not alter choice for drug reward over social reward [7]. Thus, preference for social reward has previously been shown to be unrelated to the amount of drug exposure. The second factor is the contribution of single-housed isolation, which can increase alcohol consumption [27,28,29,30,31,32]. We kept our rats individually housed in part to replicate previous studies [7], but also with the expectation that, if anything, this will increase motivation for social interaction. However in view of previous findings it is possible that social isolation increased motivation for alcohol.
Finally, the difference between intravenous and orally consumed rewards may explain why rats choose alcohol over social reward here, but other studies report choice for social reward over intravenously administered rewards [7,8,9]. The pattern of alcohol self-administration is qualitatively different to that of intravenous self-administration, where once dopamine levels in the nucleus accumbens decrease below a given threshold another reinforcement is taken [33, 34]. There is a delay in dopamine release between the action and the infusion. This delay makes choice procedures a competition between immediate reward (i.e. saccharine) and delayed reward (i.e. intravenous cocaine) [35]. A strong preference for immediate rewards can explain choice for saccharine over cocaine. Consistent with this, imposing a delay between either response and both outcomes leads to an increased preference for cocaine over saccharine [35], and also reduces choice for social reward over heroin [7].
Another key difference between orally consumed and intravenous rewards is the sensory properties of consumption. The pharmacokinetic properties of alcohol are different to intravenous drugs, and the implementation of choice in this experiment results in the rats being unable to achieve a pharmacological effect of alcohol. It is known that the internal drug state can shift choice preference [36], and consistent with this we show here that pre-choice alcohol decreases choice for alcohol. The taste of orally consumed drugs have been shown to function as conditioned reinforcers [20, 37, 38], and it is likely that the taste properties of alcohol serve as a conditioned reinforcer for the pharmacological effects of alcohol, an association that would have been well established during the home-cage alcohol phase. Thus, we propose that in a discrete choice paradigm choice is biased towards alcohol because a single reward every 8 min is insufficient to yield a pharmacological effect. This implies that the motivation for the pharmacological effects of alcohol, in part sustained by the conditioned reinforcing effects of alcohol taste, is stronger than the motivation for social interaction as implemented in this experimental setup.
Interactions between alcohol and social reward
In a manner with potential similarities to the human condition, in rodents there are social situations and factors that can promote drug use and relapse. For example, experimenter-controlled exposure to a social partner can increase alcohol consumption [39, 40], and can facilitate cocaine self-administration if that partner is also self-administering cocaine [41], and cocaine relapse can be induced by a conditioned social partner [42]. As such, it is also possible that the presence of the social partner during the choice procedures biases choice towards alcohol. In contrast to alcohol, cocaine self-administration is reduced in the presence of a social partner [43], which may also be related to the difference in social choice between cocaine [9] and alcohol. Social isolation leads to increased home-cage alcohol drinking compared to group-housed rats [31]. More complex social interactions such as social rejection or social play are also linked to alcohol use. For example, social rejection can elevate the risk of relapse in female, but not male, rats [44], and increased social play during adolescence is predictive of alcohol seeking in adverse situations [45]. While in this study we have not demonstrated an increase in alcohol consumption because of a social factor, we have shown that a strong preference for alcohol can be identified when placed in choice against social interaction.
We observed substantial individual variability in choice in response to the experimental interventions that we used to shift preference. Such a degree of individual variability is not present in the choice sessions where alcohol was at FR1. However, even during these sessions we found a negative correlation between preference for alcohol and the rate of self-administration of social reward (Fig. S3), as well as a positive correlation between preference in the high FR alcohol choice sessions and the day 1 alcohol seeking test. Thus, the propensity to choose social reward or alcohol is related to other measures of motivation. We have previously described individual variability with respect to punishment [46, 47], which others have shown both for alcohol [48,49,50,51], and intravenous drugs of abuse [52, 53]. Whether punishment-resistance is related to higher alcohol preference is not known. But the subjects who show a reduced shift in preference to social reward by increasing the effort to obtain alcohol, or decreasing the percentage of alcohol, may represent a more vulnerable sub-population that warrant further investigation.
Concluding remarks
In this study we tested a discrete choice model of addiction and observed a strong preference for alcohol over social reward. These findings stand in contrast to previous work investigating the effect of choice for social reward over intravenously administered drugs such as heroin, methamphetamine, and cocaine, but are consistent with a recent study showing preference for food pellets over social reward [22]. Overall, this study suggests that the effect of choice for social reward on drug taking and drug seeking in rodents may not generalise to alcohol. However, this study also describes the importance of social factors in alcohol abuse, and we highlight several different interventions which can shift preference away from alcohol towards social reward. It will be of interest in future studies to investigate the neurobiological mechanisms underlying both a strong preference for alcohol over social reward, and how this changes when preference shifts from alcohol to social rewards.
References
Azrin NH. Improvements in the community-reinforcement approach to alcoholism. Behav Res Ther. 1976;14:339–48.
Hunt GM, Azrin NH. A community-reinforcement approach to alcoholism. Behav Res Ther. 1973;11:91–104.
Havassy BE, Hall SM, Wasserman DA. Social support and relapse: commonalities among alcoholics, opiate users, and cigarette smokers. Addict Behav. 1991;16:235–46.
Marlatt GA, Baer JS, Donovan DM, Kivlahan DR. Addictive behaviors: etiology and treatment. Annu Rev Psychol. 1988;39:223–52.
Heilig M, Epstein DH, Nader MA, Shaham Y. Time to connect: bringing social context into addiction neuroscience. Nat Rev Neurosci. 2016;17:592–9.
Vanderschuren LJ, Achterberg EJ, Trezza V. The neurobiology of social play and its rewarding value in rats. Neurosci Biobehav Rev. 2016;70:86–105.
Venniro M, Zhang M, Caprioli D, Hoots JK, Golden SA, Heins C, et al. Volitional social interaction prevents drug addiction in rat models. Nat Neurosci. 2018;21:1520–9.
Venniro M, Russell TI, Zhang M, Shaham Y. Operant social reward decreases incubation of heroin craving in male and female rats. Biol Psychiatry. 2019;86:848–56.
Venniro M, Panlilio LV, Epstein DH, Shaham Y. The protective effect of operant social reward on cocaine self-administration, choice, and relapse is dependent on delay and effort for the social reward. Neuropsychopharmacology. 2021;46:2350–7.
Lenoir M, Serre F, Cantin L, Ahmed SH. Intense sweetness surpasses cocaine reward. PLoS ONE. 2007;2:e698.
Cantin L, Lenoir M, Augier E, Vanhille N, Dubreucq S, Serre F, et al. Cocaine is low on the value ladder of rats: possible evidence for resilience to addiction. PLoS ONE. 2010;5:e11592.
Augier E, Barbier E, Dulman RS, Licheri V, Augier G, Domi E, et al. A molecular mechanism for choosing alcohol over an alternative reward. Science. 2018;360:1321–6.
Russo M, Funk D, Loughlin A, Coen K, Le AD. Effects of alcohol dependence on discrete choice between alcohol and saccharin. Neuropsychopharmacology. 2018;43:1859–66.
Pelloux Y, Baunez C. Targeting the subthalamic nucleus in a preclinical model of alcohol use disorder. Psychopharmacology. 2017;234:2127–37.
Pennington ZT, Dong Z, Feng Y, Vetere LM, Page-Harley L, Shuman T, et al. ezTrack: an open-source video analysis pipeline for the investigation of animal behavior. Sci Rep. 2019;9:19979.
Simms JA, Steensland P, Medina B, Abernathy KE, Chandler LJ, Wise RA, et al. Intermittent access to 20% ethanol induces high ethanol consumption in Long–Evans and Wistar rats. Alcohol Clin Exp Res. 2008;32:1816–23.
Wise RA. Voluntary ethanol intake in rats following exposure to ethanol on various schedules. Psychopharmacology. 1973;29:203–10.
Baldwin AN, Banks ML, Marsh SA, Townsend EA, Venniro M, Shaham Y, et al. Acute pain-related depression of operant responding maintained by social interaction or food in male and female rats. Psychopharmacology. 2022;239:561–72.
Hart CL, Haney M, Foltin RW, Fischman MW. Alternative reinforcers differentially modify cocaine self-administration by humans. Behav Pharmacol. 2000;11:87–91.
Meisch RA. Oral drug self-administration: an overview of laboratory animal studies. Alcohol. 2001;24:117–28.
Caprioli D, Venniro M, Zeric T, Li X, Adhikary S, Madangopal R, et al. Effect of the novel positive allosteric modulator of mGluR 2 AZD8529 on incubation of methamphetamine craving after prolonged voluntary abstinence in a rat model. Biol Psychiatry. 2015;78:463–73.
Chow JJ, Beacher NJ, Chabot JM, Oke M, Venniro M, Lin DT, et al. Characterization of operant social interaction in rats: effects of access duration, effort, peer familiarity, housing conditions, and choice between social interaction vs. food or remifentanil. Psychopharmacology. 2022;239:2093–108.
Wolffgramm J, Heyne A. Social behavior, dominance, and social deprivation of rats determine drug choice. Pharmacol Biochem Behav. 1991;38:389–99.
Wolffgramm J, Heyne A. From controlled drug intake to loss of control: the irreversible development of drug addiction in the rat. Behavioural Brain Res. 1995;70:77–94.
Hopf FW, Chang SJ, Sparta DR, Bowers MS, Bonci A. Motivation for alcohol becomes resistant to quinine adulteration after 3 to 4 months of intermittent alcohol self-administration. Alcohol Clin Exp Res. 2010;34:1565–73.
Deroche-Gamonet V, Belin D, Piazza PV. Evidence for addiction-like behavior in the rat. Science. 2004;305:1014–7.
Lesscher HM, Spoelder M, Rotte MD, Janssen MJ, Hesseling P, Lozeman-van’t Klooster JG, et al. Early social isolation augments alcohol consumption in rats. Behav Pharmacol. 2015;26:673–80.
Bardo MT, Neisewander JL, Kelly TH. Individual differences and social influences on the neurobehavioral pharmacology of abused drugs. Pharmacol Rev. 2013;65:255–90.
Butler TR, Karkhanis AN, Jones SR, Weiner JL. Adolescent social isolation as a model of heightened vulnerability to comorbid alcoholism and anxiety disorders. Alcohol Clin Exp Res. 2016;40:1202–14.
Whitaker LR, Degoulet M, Morikawa H. Social deprivation enhances VTA synaptic plasticity and drug-induced contextual learning. Neuron. 2013;77:335–45.
Wukitsch TJ, Reinhardt EK, Kiefer SW, Cain ME. Voluntary ethanol consumption during early social isolation and responding for ethanol in adulthood. Alcohol. 2019;77:1–10.
Moench KM, Logrip ML. Housing condition differentially impacts escalation of alcohol intake, relapse-like drinking, anxiety-like behavior, and stress history effects by sex. Alcohol Clin Exp Res. 2021;45:480–9.
Wise RA, Leone P, Rivest R, Leeb K. Elevations of nucleus accumbens dopamine and DOPAC levels during intravenous heroin self-administration. Synapse. 1995;21:140–8.
Wise RA, Newton P, Leeb K, Burnette B, Pocock D, Justice JB Jr. Fluctuations in nucleus accumbens dopamine concentration during intravenous cocaine self-administration in rats. Psychopharmacology. 1995;120:10–20.
Canchy L, Girardeau P, Durand A, Vouillac-Mendoza C, Ahmed SH. Pharmacokinetics trumps pharmacodynamics during cocaine choice: a reconciliation with the dopamine hypothesis of addiction. Neuropsychopharmacology. 2021;46:288–96.
Vandaele Y, Cantin L, Serre F, Vouillac-Mendoza C, Ahmed SH. Choosing under the influence: a drug-specific mechanism by which the setting controls drug choices in rats. Neuropsychopharmacology. 2016;41:646–57.
Carroll ME. Oral self-administration of phencyclidine analogs by rhesus monkeys: conditioned taste and visual reinforcers. Psychopharmacol. 1982;78:116–20.
Carroll ME, Meisch RA. Concurrent etonitazene and water intake in rats: role of taste, olfaction, and auditory stimuli. Psychopharmacology. 1979;64:1–7.
Tomie A, Gittleman J, Dranoff E, Pohorecky LA. Social interaction opportunity and intermittent presentations of ethanol sipper tube induce ethanol drinking in rats. Alcohol. 2005;35:43–55.
Tomie A, Uveges JM, Burger KM, Patterson-Buckendahl P, Pohorecky LA. Effects of ethanol sipper and social opportunity on ethanol drinking in rats. Alcohol Alcohol. 2004;39:197–202.
Smith MA, Cha HS, Griffith AK, Sharp JL. Social contact reinforces cocaine self-administration in young adult male rats: the role of social reinforcement in vulnerability to drug use. Front Behav Neurosci. 2021;15:771114.
Meng S, Yan W, Liu X, Gong Y, Tian S, Wu P, et al. Social interaction with relapsed partner facilitates cocaine relapse in rats. Front Pharm. 2021;12:750397.
Smith MA. Peer influences on drug self-administration: social facilitation and social inhibition of cocaine intake in male rats. Psychopharmacology. 2012;224:81–90.
Surakka A, Vengeliene V, Skorodumov I, Meinhardt M, Hansson AC, Spanagel R. Adverse social experiences in adolescent rats result in persistent sex-dependent effects on alcohol-seeking behavior. Alcohol Clin Exp Res. 2021;45:1468–78.
Lesscher HMB, Achterberg EJM, Siviy SM, Vanderschuren L. Individual differences in social play behaviour predict alcohol intake and control over alcohol seeking in rats. Psychopharmacology. 2021;238:3119–30.
Marchant NJ, Campbell EJ, Kaganovsky K. Punishment of alcohol-reinforced responding in alcohol preferring P rats reveals a bimodal population: Implications for models of compulsive drug seeking. Prog Neuropsychopharmacol Biol Psychiatry. 2018;87:68–77.
Marchant NJ, Khuc TN, Pickens CL, Bonci A, Shaham Y. Context-induced relapse to alcohol seeking after punishment in a rat model. Biol Psychiatry. 2013;73:256–62.
Seif T, Chang SJ, Simms JA, Gibb SL, Dadgar J, Chen BT, et al. Cortical activation of accumbens hyperpolarization-active NMDARs mediates aversion-resistant alcohol intake. Nat Neurosci. 2013;16:1094–100.
Domi E, Xu L, Toivainen S, Nordeman A, Gobbo F, Venniro M, et al. A neural substrate of compulsive alcohol use. Sci Adv. 2021;7:eabg9045.
Giuliano C, Pena-Oliver Y, Goodlett CR, Cardinal RN, Robbins TW, Bullmore ET, et al. Evidence for a long-lasting compulsive alcohol seeking phenotype in rats. Neuropsychopharmacology. 2018;43:728–38.
Giuliano C, Belin D, Everitt BJ. Compulsive alcohol seeking results from a failure to disengage dorsolateral striatal control over behavior. J Neurosci. 2019;39:1744–54.
Pelloux Y, Everitt B, Dickinson A. Compulsive drug seeking by rats under punishment: effects of drug taking history. Psychopharmacology. 2007;194:127–37.
Torres OV, Jayanthi S, Ladenheim B, McCoy MT, Krasnova IN, Cadet JL. Compulsive methamphetamine taking under punishment is associated with greater cue-induced drug seeking in rats. Behav Brain Res. 2017;326:265–71.
Acknowledgements
The authors gratefully acknowledge Marco Venniro for providing Med-Associates programs for the choice procedure, and providing valuable insight throughout the initial phases of this experiment. We are also grateful to the peer reviewers for their constructive comments through the review process.
Author information
Authors and Affiliations
Contributions
NJM and TDV conceived of the experiments; RM, YvM, DS, AJM conducted the experiments; AJM analysed the video data. NJM wrote the first draft of the paper; All authors gave final approval of the submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Marchant, N.J., McDonald, A.J., Matsuzaki, R. et al. Rats choose alcohol over social reward in an operant choice procedure. Neuropsychopharmacol. 48, 585–593 (2023). https://doi.org/10.1038/s41386-022-01447-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-022-01447-6