Abstract
During evolution, the cerebral cortex advances by increasing in surface and the introduction of new cytoarchitectonic areas among which the prefrontal cortex (PFC) is considered to be the substrate of highest cognitive functions. Although neurons of the PFC are generated before birth, the differentiation of its neurons and development of synaptic connections in humans extend to the 3rd decade of life. During this period, synapses as well as neurotransmitter systems including their receptors and transporters, are initially overproduced followed by selective elimination. Advanced methods applied to human and animal models, enable investigation of the cellular mechanisms and role of specific genes, non-coding regulatory elements and signaling molecules in control of prefrontal neuronal production and phenotypic fate, as well as neuronal migration to establish layering of the PFC. Likewise, various genetic approaches in combination with functional assays and immunohistochemical and imaging methods reveal roles of neurotransmitter systems during maturation of the PFC. Disruption, or even a slight slowing of the rate of neuronal production, migration and synaptogenesis by genetic or environmental factors, can induce gross as well as subtle changes that eventually can lead to cognitive impairment. An understanding of the development and evolution of the PFC provide insight into the pathogenesis and treatment of congenital neuropsychiatric diseases as well as idiopathic developmental disorders that cause intellectual disabilities.
Similar content being viewed by others
PFC definition
There is little disagreement that the human cerebral cortex is the organ that enabled abstract thinking and the creation of civilization, including architecture, science and all types of art. Using a wide variety of methodologies, the size and cytoarchitecture of the frontal lobe, and more specifically the PFC, has been extensively studied over the years in various species. The PFC in humans and nonhuman primates can be divided into a collection of structurally and functionally different subdomains positioned anterior to the motor cortex; the medial (mPFC), lateral prefrontal cortex (lPFC) and orbitofrontal cortex (oFC). The lPFC is mostly involved in language and executive processing, while the oFC and mPFC are known to contribute to cognitive functioning and emotional control [1,2,3,4]. The mPFC can be further subdivided into the infralimbic (IL), the prelimbic (PL) and anterior cingulate cortex (ACC). The most ventral subdomain of the mPFC is the infralimbic cortex (IL) and is involved in coping with chronic stress eventually leading to structural changes and prefrontal dysfunction [5,6,7,8,9,10,11]. Interestingly, the PFC of rodent models such as mice is limited in size, containing medial, orbitofrontal and cingulate areas, but probably lacking the equivalent of the primate dorsolateral PFC. In humans, the PFC can be considered to have evolved disproportionally large and it is thought to be the last region of the brain to gain full maturity [12, 13].
Evolutionary view on PFC development
During mammalian evolution, the cerebral cortex not only increased in neuronal numbers and surface area but also acquired new cell types and cytoarchitectonic areas. Species-specific adaptations of prefrontal areas, steered by the environmental demands, can explain the differences in size of frontal areas over time. Among the most recent additions are several association areas, particularly the PFC, which has expanded enormously in primates culminating in humans [14]. In humans, the PFC occupies as much as about 30% of its surface. Although still debated, the human frontal lobe seems to have evolved three times larger than that of our closest living relatives, the great apes. In fact, it has been argued that the human brain possesses prefrontal regions that are both qualitatively and functionally exclusive [15]. It is, nevertheless, remarkable that we use the rodent model for most of the cellular and molecular neuroscientific studies, despite its lissencephalic brain which is clearly much simpler in both cytoarchitecture as well as function. A valid question still remains: Do rodents have a prefrontal cortex? [16, 17]. And if we were to focus more on the evolutionary aspects of prefrontal development in terms of structural organization and function, should we not include longitudinal neurodevelopmental studies on more species [18, 19]? Although the basic principles of cortical development may be similar in all mammals, the modifications of developmental events during millennia of primate evolution produce not only quantitative but also qualitative changes of its cellular structure and synaptic circuitry [13, 20]. The origin of species-specific distinctions can be traced either to the new or phylogenetically conserved genes that act at the time of the neural stem cell’s exit from the mitotic cycle and generate a different outcome, depending on the evolutionary context by interacting with a postmitotic neuron. Thus, the PFC as well as the Broca and Wernicke association areas in humans, which are formed in the frontal and temporal lobes, display a temporarily enriched gene expression pattern that is distinct from the mice or macaque cerebrum at the comparable prenatal stages (e.g., [21, 22]). More on evolution of the prefrontal cortex can be found in this volume, part I, chapter 1.
The early stages of PFC development
Genetic determination of the PFC
Still inside the womb, the generation of neural tissue (human, third gestational week) begins with the induction of ectoderm into neuroectoderm after which the neural tube will form through a process called neurulation [23]. The detailed analysis of a series of embryonic and fetal human postmortem brain tissue, as well as the evidence from experiments on animal models that range from rodents to nonhuman primates, showed that specific genes and regulatory elements are involved in evolutionary elaboration of the cranial part of the neural tube. More specifically, it is well documented that differential gene expression and the gradients of signaling molecules across the embryonic brain generate prospective subdivisions of the neocortex [24,25,26,27,28,29]. Work of Cholfin and Rubenstein in mice provide experimental evidence that the PFC can expand differentially and independently of the growth rate of the other areas [30, 31] and that its size can be regulated at early stages by the change of expression of specific growth factors before they receive the afferent axonal input [32]. Through regional specification in which the Fgf family plays a significant role, the (pre)frontal cortical area starts to expand [32]. The formation of the cytoarchitectonic map during evolution and individual development can be explained by the Protomap Hypothesis (PMH) of cortical parcellation [33]. This hypothesis postulates that intersecting gradients of molecules are expressed across the embryonic cerebral wall that guide and attract specific afferent systems to the appropriate position in the cortex where they can interact with a responsive set of cells [34]. The prefix “proto” indicates the malleable character of this primordial map, as opposed to the concept of equipotential cortical plate consisting of the undifferentiated cells that is eventually shaped and subdivided entirely by the instructions from those afferents [35, 36]. The PMH is at present universally accepted even by its initial opponents (e.g., [29]).
Prefrontal expansion and lamination
The structural development of the various subdomains of the PFC is a meticulous process starting with a massive expansion of the most proximal part of the developing neural tube. The first step in the expansion of the cortical surface during development starts with an increase in the number of symmetrical divisions of neural stem cells in the ventricular zone (VZ) before the onset of neurogenesis and the formation of the subventricular (SVZ), intermediate (IZ) and subplate (SPZ) zones and cortical plate (CP) below the marginal zone (MZ) [33, 37,38,39], for review see [34]. This initial cortical expansion is also supported by experimental studies in mice [40,41,42,43] and provides an explanation for the massive increase in cortical surface area during both individual development and evolution.
By the time the apical radial glial progenitors within the prefrontal subdomains start dividing asymmetrically, the number of neurons will increase rapidly and peaks between week 13 and 16 of gestation in human (E10-E15 rodents/E43-E50 primates), specifically in the dorsal telencephalon [44,45,46,47]. The labeling of dividing cells by the DNA replication markers tritiated thymidine (TdR) and bromodeoxyuridine (BrdU) showed that in nonhuman primate rhesus macaque, most cortical neurons, including those destined for the PFC, originate in the proliferative VZ near the cavity of the cerebral ventricle, between the 40th embryonic day (E40) and E100, during the 165-day-long gestational period in this species [48]. Genesis of neurons destined for the PFC is completed by E90, before completion of neurogenesis in the primary visual cortex at E90 (Fig. 1 and [49]). Through close interplay between cell-autonomous events and local as well as external cues, neurons generated close to the ventricle start migrating in radial columns [45, 46, 50, 51]. Gray matter continues to increase well into adolescence [52]. Astrocytes being the most abundant type of glial cells within cortical areas, are generated from the radial glial cells in the VZ and from the intermediate progenitors in the SVZ after the peak of neurogenesis [53]. The oligodendrocyte precursor cells, or OPCs, are generated within the medial ganglionic eminence and the anterior entopeduncular area and migrate toward the frontal cortical regions [54]. In the final stage of OPC production, this generation occurs in the cortical regions themselves. Microglia cells, on the other hand, are of mesodermal origin and migrate throughout the brain [55].
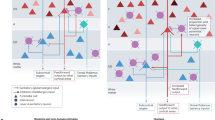
A Pen drawing of a macaque brain (side view) with the PFC indicated in pink. B Schematic overview of the time line for which [3H] thymidine ([3H]dT) injections were given at particular embryonic (E) time points indicated by green arrowheads. sac, sacrifice. C Relationship of time of origin and the final position of neurons destined for the PFC in macaque monkeys based on autoradiographic labeling of DNA replication by [3H] thymidine at various days of gestation (for details of the approach and methodology see Rakic [48]. Embryonic days are represented on the horizontal axis, vertical lines indicate the embryonic day on which an animal received a pulse of [3H]dT and the horizontal markers on the vertical lines represent the positions of heavily labeled neurons in the PFC. A schematic representation of the approximate position of layers I–VI and the white matter (WM) is indicated on the left (green rectangle). The data show that all neurons in PFC are generated between embryonic (E) day 40 and E90 within the 165-day-long gestational period in this primate species.
After the last cell division, postmitotic neurons migrate an increasingly long distance across the embryonic and fetal cerebral wall to their final positions in the cortex that develops below the pial surface [33, 56]. Although similar DNA labeling is not possible to perform in humans, examination of histological and Golgi silver impregnation methods of the embryonic and fetal human cerebrum indicate the existence of similar timing and sequence of these developmental events [37, 57, 58]. The pyramidal excitatory neurons born in the VZ and SVZ of the prefrontal subdomains, similar to other cortical areas, start to migrate radially toward the proper position in the CP under the influence of Fgfs [50, 59, 60]. Migrating neurons are guided over an increasingly long and curvilinear pathway by the elongated radial glial cell fibers that span the entire developing cerebral wall [61,62,63]. The radial glial processes that extend to the pial surface serve as a scaffold for the migrating neurons, which will settle themselves in an inside-out manner with the earlier-born neurons in the deeper layers and later born neurons in the more superficial ones [56, 64, 65]. Born in the ganglionic eminences, GABAergic interneurons migrate tangentially to the proper place within the prefrontal subdomains [66, 67]. Some recent findings in human and primates, such as the role of outer radial glia cells (oRGCs) and truncated glial cells, the diversity and complexity of cortical progenitors, the role of the subplate and the high specificity in axonal guidance events, again underline the complexity and evolution of cortical areas [68,69,70,71,72,73,74,75]. We now know from recent studies that it is the birth and migration of neurons derived from oRGCs that play a role in the development of the primary sulci (superior frontal, inferior frontal and precentral) in week 25–26 of gestation [23, 76]. After the process of migration is completed, RGCs retract their apical process and generate astrocytes and oligodendrocytes. In nonhuman primates and human, glial cells seem to somewhat outnumber neurons in the PFC, albeit with regional variation which is likely to contribute to the formation of secondary and tertiary gyri [77,78,79,80,81].
Contrary to some initial concepts and theories [35, 36], embryonic VZ and CP are not uniform and equipotential. The enlargement and introduction of the new cytoarchitectonic areas has been explained by the Radial Unit Hypothesis (RUH). According to this hypothesis, increasing the size and proliferative capacity of the neuronal stem cells in the proliferative zone enables initial enlargement of the cortex as well as formation of the distinct anatomical and functional cytoarchitecture areas in the mammalian evolution [33]. According to the RUH, tangential (horizontal) positions of cortical neurons are determined by the positions of their precursor cells, now called stem cells in the VZ, while their radial (vertical) position in the overlying cortex is determined by the time of their origin (Fig. 2). Therefore, the addition of the number of the radial columns increases the size of the cortical surface, whereas the number of cells within the columns determines its thickness.
A Three-dimensional reconstruction of postmitotic neurons migrating along radial glial fibers, based on electron micrographs of semi-serial sections of the monkey fetal cerebral cortex with permission from Rakic [56]. B Representation of the radial unit hypothesis based on Rakic [33] with permission from Silver et al. [396]. C Illustration of the dynamics of major developmental events and diversity of progenitors involved in the development of primate cerebral neocortex based on studies of Rakic, with permission from Silver et al. [396].
Differentiation and synaptogenesis
After neurons assume their final position, they begin to differentiate further and form synaptic connections. In humans, between 17 and 50 weeks of gestation (first to fourth postnatal week in rodents), the pyramidal and interneurons in the various cortical layers of the PFC will further mature and differentiate [82, 83]. The basal and apical dendritic length will increase, the spines will further develop, specifically in layer III and V, and their axons will extend to other cortical and subcortical targets [82, 84]. This is also the case for the inhibitory network where the interneurons mature extensively with a sharp increase in the dendritic spine formation but also in terms of their intrinsic as well as their network properties as was shown in mice [85, 86]. Prefrontal synaptogenesis starts prenatally and peaks postnatally followed by a process called pruning or refinement of synaptic connections, the removal of unused synaptic contacts [87]. When neurites to and from the PFC reach their final target position, an immature synapse is generated under the influence of, among others, cell adhesion molecules and reelin [88, 89]. Epigenetic regulatory factors such as microRNAs (miRNAs) play an important role in this process by modulating dendritic and synaptic maturation [90, 91]. The tempo and kinetics of synapse formation in the primate PFC closely resemble those described for other areas [92]. In young primate embryos, a precortical phase (E47-E78) is described when synapses are found only above and below, but not within, the CP. Following that, there is an early cortical phase, from E78 to E104, during which synapses accumulate within the cortical plate, initially exclusively on dendritic shafts. The next rapid phase of synaptogenesis begins at 2 months before birth and ends approximately at 2 months after birth, culminating with a mean density of 750 million synapses per cubic micrometer. This accumulation is largely accounted for by a selective increase in axospine synapses in the supragranular layers. Therefore, the early childhood PFC contains a 2–3 fold higher density of dendritic spines compared to the adult PFC. The period of overproduction of synapses is followed by a protracted plateau stage that lasts from 2 months to 3 years of age when synaptic density remains relatively constant. In humans, the PFC synaptic density spikes around 3.5 years of age (~4th postnatal week in rodents), which is relatively late compared to other cortical areas and almost double the net density of the adult PFC [82, 93, 94]. Examination of the course of synaptogenesis in the macaque PFC, by detailed quantitative electron microscopic analysis, showed that the number of synaptic contacts is initially grossly overproduced before declining to the normal adult level (Fig. 3 and [49]. Likewise, the axons of the corpus callosum, as well as other large axonal tracts in the macaque cortex, including PFC, are grossly overproduced before decreasing to the adult level [95,96,97]. A subpopulation of GABAergic neurons in the subplate zone also form transient synapses that are eventually eliminated [98, 99]. The period of synaptic decline in human PFC, which starts during childhood, is initially dramatic and continues during adolescence and extends at a slower, but statistically significant rate into the 3rd decade of life (Fig. 4 and [12]).
A Schematic representation of the site of the block dissection from the depth of the Sulcus Principalis (SP). On the right: The section of the cortex in the SP showing the vertical (radial) probes across layers I–VI, which were examined by electron microscopy. B The total number of synaptic contacts in each vertical probe as represented by the green dots. The semi-log plot in abscissa represents the number of days after conception. Adapted from Bourgeois et al. [49].
A Low-magnification photograph of the rapid Golgi-impregnated layer IIIc and V pyramidal cells in the dorsolateral PFC of a 16-year-old subject. B Neurolucida reconstruction of layer IIIc pyramidal neuron of a 49-year-old subject showing distal oblique (green), proximal oblique (blue) and basal dendrites (red). C Representative high-power magnification images of rapid Golgi-impregnated layer IIIc pyramidal neurons in a 1 month old infant, 2.5-year-old child, and 16-, 28-, and 49-year-old subjects. D Graphs representing number of dendritic spines per 50-μm dendrite segment on basal dendrites after the first bifurcation (red); apical proximal oblique dendrites originating within 100 μm from the apical main shaft (blue); and apical distal oblique dendrites originating within the second 100-μm segment from the apical main shaft (green) of layer IIIc (filled symbols) and layer V (open symbols) pyramidal cells in the human dorsolateral PFC. Squares represent males; circles represent females. The age in postnatal years is shown on a logarithmic scale. From Petanjek et al. [12].
The finding that synaptic density in the cerebral cortex is relatively stable from early adolescence through puberty (the plateau period) is indicative that in primates the final synaptic pattern is the result of selection and refinement of their higher number during the formative years when learning experiences are most intense. These discoveries led to the proposal that the Selective Elimination Hypothesis is a mechanism for tuning synaptic connections by interaction with the environment during the period of most intense learning [92]. These days, selective elimination or stabilization is commonly called “pruning”, and this refinement of the differentiating cortical network via pruning of dendritic branches, and/or efferent/afferent projections, is an important process to fine-tune the meticulous intricate prefrontal network [100, 101]. Within the rodent and primate PFC, this process of synaptic pruning, which is most dramatic in layer III, continues well into adolescence leading to a long-lasting decline in synaptic density across PFC subdomains [82, 102, 103]. It was furthermore discovered that major neurotransmitter receptors are also initially overproduced in all eight primate prefrontal regions examined [104, 105]. Moreover, during childhood the PFC myelination process starts (white matter volume increase) which continues into adulthood [106, 107].
Getting connected
The prolonged maturation of the PFC depends largely on the coordinated action of various external factors. Most neurotransmitter projections arrive in the prefrontal subdomains in two streams: within the marginal zone (MZ) and within the subplate zone (SPZ) which is thicker in the PFC compared to other cortical areas. A major change in development, which likely signals roots in the evolution of the cortex, is in the specificity in neurotransmitter systems alongside a boost in receptor type heterogeneity in primates and human [108,109,110,111,112]. In humans the thickening of the PFC subplate has evolved tremendously, suggesting playing a role in the extensive prefrontal circuitry [71, 102]. Vice versa, the multitude of pyramidal neurons in the various layers and PFC subdomains will connect to other cortical and subcortical targets by extending their axons, once they have reached their final position in the PFC (human: birth till end of first year/rodent first 2 postnatal weeks). The intricate timely integration of all these neurotransmitter systems is essential for prefrontal functioning. In this way, a unique and higher-order functional network capable of emotional processing and complex cognitive abilities is established.
Developing PFC connections - from the neurotransmitter perspective
Serotonin
The brain matures from the brainstem to the more frontal cortical regions, and it is therefore not surprising that serotonergic projections from both the dorsal as well as the medial Raphe nuclei (DRN, MRN respectively) are among the first to emerge and are set towards cortical regions where they arrive in the PFC around E16/E17 in rodent and week postnatal 10–13 in humans [113,114,115,116]. Most of the work on molecular and cellular underpinnings of serotonin functioning and guidance during early development have been investigated in rodents, although it is clear that the specificity of serotonergic prefrontal connectivity in primates and human increase tremendously in regional specificity [109]. In mice, the serotonergic projections toward the forebrain are predominantly guided by the Epha5/ephrina5 interaction of guidance cues [117]. Of note, early in the development of serotonergic signaling, molecules such as receptors and transporters are already expressed in the forebrain and an exogenous placental source of 5-HT has been considered to direct cortical development even before raphe-derived projections have reached the forebrain [118,119,120,121]. Once the serotonergic projections have arrived within cortical areas, they are able to make contacts with Cajal Retzius cells within the MZ, thereby raising the possibility of playing a role in neuronal migration [122,123,124]. It has become widely accepted that serotonin exerts a significant trophic and modulatory function in neurodevelopmental processes such as proliferation, migration and differentiation in cortical areas, including the PFC [119, 123, 125,126,127,128,129].
Noradrenalin
The Locus Coeruleus (LC) in the brainstem sends out its noradrenergic axonal projections to the PFC as early as E16/17 (rodent) and week 10–13 in human [130,131,132]. It appears to be a heterogeneous set of neurons innervating all aspects of the PFC subdomains [133,134,135]. Noradrenergic projections arrive in cortical areas before all cortical neurons have finished migrating and have adopted their final appearance [136]. During the embryological development of prefrontal areas specifically, noradrenaline plays a role in cell division, neuronal migration, differentiation as well as synaptogenesis [137,138,139,140,141]. Like serotonin, noradrenergic axons make contact with the Cajal Retzius cells in the marginal zone, suggesting a role in the laminar formation of cortical regions [132, 142, 143]. In addition, noradrenalin seems to have an effect on the development of dopaminergic projections in the PFC by providing a dopamine reuptake mechanism through the noradrenalin transporter [144, 145] as well as on GABAergic signaling in the PFC [146, 147]. Recent studies of rat and primate PFC showed that the α2-adrenoceptor and muscarinic M1 receptor modulate working memory via KCNQ potassium channel [148,149,150,151]. Reciprocal direct connections from the mPFC to the LC mature over time, and this system is involved in a variety of behaviors such as memory formation, attention, arousal, vigilance and coping with stress [152,153,154].
Dopamine
A subset of the medial part of the ventral tegmental area (VTA) starts to project to prefrontal subdomains around E15/E16 (rodent) and week 10–13 in human [155,156,157,158]. Steering dopaminergic projections from the VTA via the medial forebrain bundle toward forebrain regions mostly depend on a coordinated action of the guidance molecules Dcc and Netrin-1 mediated by microRNA miR-218 control of Dcc expression in the VTA [159,160,161], while Semaphorin3F is orchestrating their fasciculation, rostral growth and targeting within the various mPFC subdomains [156]. The dopaminergic innervation of the mPFC in rodent surges during adolescence hallmarked by massive changes in the organization, shape and density of the dopaminergic fibers [162,163,164]. A similar surge in regional-specific dopaminergic connectivity to the PFC can be observed in primates, including human modulating local microcircuits [165,166,167,168,169]. Of note here is that some of these dopaminergic neurons projecting to the various PFC subdomains are capable of co-releasing glutamate as well and have an exclusive excitatory effect on the GABAergic interneurons in the various layers of the PFC [170,171,172,173]. Eventually, the mature mesoprefrontal system is involved in attention, behavioral flexibility, action planning, sustainability of motivational and affective states, working memory and memory consolidation which is mediated in parallel by catecholaminergic pathways [169, 174,175,176,177,178]. In many neurodevelopmental disorders (NDDs) the developing dopamine system is affected playing a role in the diverse symptoms of these disorders [179].
GABA
Most of the GABAergic interneurons are born in the ganglionic eminences of the ventral telencephalon and migrate tangentially to the proper cortical areas and layers to form a network with the radially migrated pyramidal neurons [180,181,182]. Initially being excitatory through the GABAA receptors expressed on radial glia cells and migrating interneurons, GABA plays a role in proliferation, migration and synaptogenesis [183,184,185,186]. It has furthermore been shown that dopamine and GABA interactions can influence these processes [187, 188]. Around the second postnatal week in rodents (~first postnatal week in human), the depolarizing effect slowly transitions into an inhibitory net effect depending on place and time [189, 190]. A remarkable feature in GABA signaling from an evolutionary perspective is that in nonhuman primates and human there seems to be a cell-type specific expression of the GABA transporter GAT-1 in early childhood [191, 192]. Furthermore, it appears that nonhuman primates and humans have distinct populations of GABAergic neurons which originate in proliferative zones of the dorsal telencephalon [193, 194].
Glutamate
There are various sources of glutamatergic input projections including a subset of (non-)dopaminergic VTA neurons to GABAergic interneurons in the PFC [170,171,172,173]. The most prominent monosynaptic inputs of the PFC are derived from hippocampus, mediodorsal (MD) thalamus and amygdala [195,196,197,198,199,200,201]. In fact, the medial pulvinar part of the medial thalamus or PM, which evolutionary expanded alongside the association cortex in nonhuman primates and human, is characterized by a distinct prefrontal glutamatergic connectivity that seems to play a significant role in NDDs [202]. A multitude of cortical and subcortical targets are progressively innervated by developing glutamatergic projections from the PFC itself such as the various thalamic regions. Recently it was found that retinoic acid (RA) plays a critical role in PFC development and specifically in this thalamus-prefrontal connectivity [203]. The PFC furthermore sends out glutamatergic afferents to the VTA as well as to the nucleus accumbens modulating dopaminergic signaling [204,205,206]. In addition, DRN serotonergic neurons are controlled by glutamatergic projections from the PFC [152, 207,208,209].
Acetylcholine
Around birth, the numerous cholinergic projections arising from the basal forebrain nuclei innervate the primary cortical regions where they influence cortical ultrastructure [210,211,212,213]. Acetylcholine modulates primarily the prelimbic subdomain of the PFC during development targeting GABAergic interneurons [210]. But even before the cholinergic projections arrive in the cortical areas, the nicotinic and muscarinic receptors are expressed on neural progenitors playing a role in proliferation/differentiation and axonal guidance events [214,215,216,217]. In the PFC of nonhuman primates, the muscarinic M1 receptors modulate working memory via KCNQ potassium channels [151]. Alongside, a transient expression of the enzyme acetylcholinesterase seems to play a role in the thalamocortical circuit formation [218,219,220]. Within the PFC, the cholinergic innervation initially terminates in layers III and IV slowly losing laminar preference over time [221,222,223]. Key developing PFC circuitry is shaped by acetylcholine, and PFC pyramidal neurons depend on its proper signaling in terms of dendritic branching, spine formation and synaptogenesis [224,225,226].
Convergence of developing transmitter systems within the PFC
There is, furthermore, ample evidence now that during embryonic development there is convergence of the various neurotransmitter signaling pathways and influence each other’s development and functioning [153, 227,228,229,230,231]. These neurotransmitter systems can act as neurotrophic factors steering various neurodevelopmental events in their target areas. Serotonergic and dopaminergic markers are jointly present in their developmental origins, guidepost areas, as well as within the subdomains of the PFC, which is important for their intricate interaction later in life to establish higher cognitive functions [209, 232,233,234]. The same holds true for noradrenergic and dopaminergic projections towards forebrain regions as well as dopaminergic-glutamatergic and dopaminergic-cholinergic interactions controlling PFC maturation and functioning [227, 228, 231]. These neurotransmitter projections initially innervate prefrontal regions via two parallel paths; one via the subplate and one via the marginal zone where the Cajal Retzius cells reside [235]. Being in close proximity of the CR cells, it is likely that volume transmission is used to release the neurotransmitter. Receptors, transporters as well as synthesizing enzymes are already expressed (~E10 rodent and week 4–5 human) even when the actual axonal projections have not yet arrived in the PFC [236,237,238,239,240]. In fact, neurotransmitter receptors are found to be expressed by progenitor cells throughout development [231]. External neurotransmitter sources, such as the placenta, can play a role in this early shaping of cortical areas [114, 118, 241, 242]. All this is especially important in light of (anti-depressant) use of pharmacological drugs during pregnancy as they can interfere with these early signaling pathways and hamper the structural development of brain areas including the PFC.
PFC cognitive development
The PFC, as the seat of our higher-order cognitive functions, continues to develop into adulthood [52, 243]. It is among the latest brain regions to fully mature in humans as well as rodents [106, 159, 244, 245]. The primary somatosensory cortex, as well as the primary motor cortex, mature earlier, however the dendritic trees and the density of spines within the subdomains of the PFC seem to be more complex [246,247,248,249]. Cognitive abilities are shaped by experience over time and seem to be in synchrony with PFC structural changes such as synaptogenesis and pruning [250]. Following the ‘use it or lose it’ principle, the developing PFC dynamically rearranges incoming and outgoing wiring depending on usage and need [12]. Specific for the PFC, the non-coding microRNAs mir-128b and mir-30a-5p have shown to be involved in prefrontal-dependent cognitive maturation by affecting epigenetic mechanisms [251, 252]. The constantly developing cognitive and executive capabilities occur parallel to the neurophysiological changes within the PFC and its connected areas and seem to reach a plateau in teenagers (around 12 years in human, around P50 in rodents) [253]. Adolescence is typically characterized by changes in social interactions and cognitive abilities in order to gain independence and adult skills and competences [245]. In nonhuman primates this is characterized by risk-taking, novelty seeking, and increased vigilance; whereas in rodents by play behavior, increased exploratory activity and impulsivity are peaking [245]. Higher order cognitive functions, in which PFC plays a prominent role, such as language and intelligence, continue to develop into adulthood [254, 255]. More on the role of the PFC in cognitive control and executive functioning can be found in this volume in the reviews by Robbins and Friedman (I.6) and Menon and D’Esposito (I.7).
PFC development and mental illness
Although stress-induced structural changes in the PFC are equally important in their contribution to the pathophysiology of neuropsychiatric conditions [256,257,258,259,260,261,262] (see also part III of this volume), we focus in this paragraph on particular risk factors involved in the onset of NDDs) in which PFC functioning is affected. It has been speculated that, as the PFC takes so long to fully mature, it also has the largest critical window of all developing brain areas. The various risk factors, either genetic or environmental, can hamper the intricate developmental events and pose a risk in developing NDDs (Fig. 5).
Schematic overview of genetic and environmental risk factors during pregnancy to be the possible cause of NDDs (Clockwise: Genetic causes, Smoking, Drinking, prescription or recreational Drugs, certain combination of Nutrients, Physical factors such as UV, ultrasound or various radiations, Toxins, Virus infections). Possible causes for NDDs include specific genetic or environmental factors as well as a combination of both.
From a genetic point of view
The group of patients having a NDD is enormously heterogeneous. The genetic causes underlying NDDs are diverse ranging from single gene mutations, copy number variations to whole‐chromosome aberrations [263]. Even with monogenic causes, the severity and comorbidity of the symptoms can vary tremendously and neurological/neuropsychiatric symptoms are often accompanied by additional clinical features such as maldevelopment of organ systems. But there are also some clear examples of environmental risk factors that specifically hamper PFC development resulting in behavioral and cognitive deficits. Below we list specifically those NDDs where the structural development of the PFC is clearly affected.
Monogenic causes
Many of the Mendelian monogenic NDDs are characterized by intellectual disability and behavioral problems due to, in part, an altered prefrontal functionality. Fragile X syndrome (FX) is a NDD where the causative gene, Fragile X Mental Retardation Protein (FMRP), is completely absent causing a plethora of developmental abnormalities [264, 265]. It is clear that in FX the many behavioral and cognitive deficits can be attributed, at least in part, to prefrontal dysfunction. Some of these aspects could be rescued in an animal model where FMRP production was initiated in the mutant PFC [266]. In Rett syndrome, a severe NDD with specific cognitive and behavioral features, there is a strong PFC hypofunction with structural abnormalities [267,268,269]. Restoring Mecp2 levels within the PFC in mice via state-of-the-art techniques such as CRISPR-Cas9 or DREADDS can restore some of the endophenotypes such as social recognition deficits or long-term retrieval of auditory conditioned fear [267, 270, 271]. Other monogenic syndromes like Kleefstra, KBG, WitKoS, Angelman, Coffin-Sirris, Rubinstein-Taybi, Phelan-McDermid, Smith–Magenis Syndrome and Kabuki syndrome also have a clear prefrontal component in their behavioral and cognitive phenotype [272,273,274,275,276,277,278,279,280]. For some of these syndromes it has recently been shown that deficits in the structural development of the PFC underlie these problems [281,282,283,284,285,286,287].
Chromosomal abnormalities
In all human chromosomal aberration syndromes, including trisomies, monosomies (e.g., Turner syndrome, monosomy 1p36), polyploidies, disomies and imprinting errors or sex chromosome anomalies, structural abnormalities of (pre)frontal as well as many other areas are common [61, 288,289,290,291,292]. Trisomy of chromosome 21 or Down syndrome can be considered a NDD with significant developmental deficits. Cognitive abilities are affected due to a developmental delay including the maturation of brain areas such as the PFC [293]. Particular neurodevelopmental events are delayed in forebrain regions such as neurogenesis, migration and synaptogenesis eventually resulting in altered prefrontal circuitry [294,295,296]. Williams (WBS or WS) syndrome is a rare NDD with a deletion of approximately 25 genes on chromosome 7 and characterized by an unusual sociability and cognitive deficits [297]. The structural organization of prefrontal pyramidals, specifically their density and dendritic arboring, is severely affected [298, 299]. Prader-Willi syndrome (PWS) is a disorder in which imprinted genes on chromosome 15 are affected and is characterized by increased volume of prefrontal subdomains important in the reward circuitry [300, 301]. In the 22q11.2 deletion syndrome (or DiGeorge/Velo-Cardio-Facial syndrome), individuals are characterized by loss of executive function and working memory alongside other cognitive problems and MRI studies showed a clear loss of volume of the various PFC subdomains [302,303,304,305,306,307]
From an environmental point of view
Food/drugs
One of the most studied risks during pregnancy is the composition of our diet. Many food-derived molecules can reach the unborn baby in one form or another, and therefore could directly or indirectly influence brain development when crossing the immature blood-brain barrier and potentially affect PFC development [308]. It is therefore important to realize that with the change of our diet through the ages, having become more processed and high-fat and high-sugar in contents, this can have a dramatic effect on the development and functioning of the PFC. Particular consumption of high-fat and/or high-sugar during pregnancy, childhood and adolescence can result in structural changes in the PFC and deficits in executive functioning [308,309,310,311]. Maternal metabolic disorders including diabetes and obesity can pose another threat to the unborn child as placental dysfunctioning alters the prenatal exposure to nutrients and toxins [312,313,314]. In the early ‘70s it was found that women who abused alcohol during pregnancy may deliver children with severe developmental delays, smaller brains and cognitive problems called Fetal Alcohol Syndrome (FAS) [315]. These children often have various conditions that are collectively known as Fetal Alcohol Spectrum Disorder (FASD), which includes FAS and a condition known as Alcohol-related Neurodevelopmental Disorder (ARND). There is a clear correlation of children with FAS and prefrontal executive functioning [316]. MRI studies showed reductions in brainstem as well as cerebellum volume in a primate FAS model and a sex-dependent change in functional connectivity and metabolism in prefrontal areas in a rat FAS model [317, 318]. Structurally, the prefrontal cortical thickness is affected after prenatal alcohol exposure and it matures with a smaller number of excitatory neurons and more GABAergic ones disrupting the excitatory/inhibitory balance severely [319,320,321]. Similar structural and behavioral defects of the PFC can be observed in kids with prenatal exposure to opioids, cocaine, amphetamines and other drugs-of-abuse [322,323,324,325,326,327,328,329,330]. Similarly, we can find lead and other pollutants to be damaging to the developing brain and PFC [331,332,333,334]. Another field of recent study is the perinatal exposure to pharmaceuticals given to treat the pregnant mother. Perinatal HIV infections can alter the course of brain development (see below), on the other hand perinatal exposure to antiretroviral drugs such as Efavirenz (EFV) to treat HIV leads to an altered prefrontal cytoarchitecture [335, 336]. Although maternal stress itself can be detrimental to brain development in general and the developing PFC in particular (for review see [125]), treatments against maternal depression such as SSRIs can cause substantial structural damage to prefrontal subdomains [125, 128].
Viral infections
Traditionally, pregnant women were warned for TORCH (TOxoplasmosis, Rubella, Cytomegalovirus, and Herpes simplex viruses type 1 and 2) infections especially during the first two trimesters of pregnancy as they were shown to cause severe congenital abnormalities [337]. Later, the O was referring to Other infections such as syphilis, varicella-zoster, and parvovirus. Zika viral infection during pregnancy can cause microcephaly including severe structural damage to the prefrontal areas [338,339,340]. The Zika virus is able to infect neuroepithelial stem cells and cortical radial glial cells and to a lesser extent postmigratory neurons causing structural disorganization in these cells eventually leading to cell death [341,342,343]. Even a postnatal viral infection can lead to postnatal meningitis and neurodevelopmental problems due to structural and functional damage of frontal areas [344]. In the recent SARS-CoV-2 or COVID-19 viral outbreak, similar structural damage of frontal cortical areas could be observed, most likely due to an inflammatory response in which parenchymal cells and the choroid plexus are involved [345,346,347]. Little is known however on the short- and long-term effects of a COVID-19 infection during pregnancy and the possible neurodevelopmental changes it can make during corticogenesis leading to NDDs [348].
Other perinatal causes
Multiple fetuses per pregnancy, intrauterine growth restriction (IUGR, due to placental failure other than by causes described above), X-ray, UV, nuclear or cosmic radiation, (ultra)sound as well as high temperature, preterm birth or hypoxia whether or not by traumatic causes can pose serious threats to proper corticogenesis as well [349,350,351,352,353,354,355,356]. Recently it has become clear that IUGR is associated with an increase in impulsive behavior due to an altered dopamine signaling in the PFC [357, 358]. Perinatal hypoxia can furthermore change the expression of cytokine and ceramide metabolism genes in the PFC and hampers cognitive functioning in later life [359, 360]. In preterm birth, changes in white and gray matter including reductions in cortical surface area and cortical thickness of the PFC are described [361,362,363,364]. Disruptions in PFC network activity often further aggravates the already compromised neurocognitive development in these children [365, 366].
The multifactorial view
It is now generally accepted that the etiology of many NDDs is considered to be multifactorial. Often, comorbidity of two or more NDDs is observed. Variable environmental exposure to risk factors combined with variable genetic background makes it hard to pinpoint possible causes. Yet, as the PFC takes the longest to fully mature, we can argue that it is most vulnerable to any risk factor when presented early enough. We will here review three of the ‘classical’ NDDs that are considered to be multifactorial in their onset with affected PFC development. Intellectual disability (ID) is an umbrella term and is a comorbidity of many of the described NDDs and will therefore not be discussed separately.
Autism spectrum disorder (ASD) is an example heterogeneous NDD characterized by impaired communication and social interaction accompanied with repetitive behavior and stereotyped interests. It has been described that the PFC of individuals with ASD show structural and functional changes, specifically in the ACC, oFC and lPFC [367, 368]. The number of neurons (but also their size), specifically the chandelier cells, basket cells and other parvalbumin-expressing interneurons, is decreased in the PFC [369,370,371]. There are also indications that serotonergic signaling is affected during PFC development [372]. In the first couple of years of life there is a prefrontal hyperconnectivity in children with ASD followed by a hypoconnectivity resulting in a ‘disconnection’ with other cortical areas involved in higher-order associative processing [373,374,375,376]. The short- and long-range prefrontal axons, particularly those from the ACC, are affected in their guidance to subcortical targets and may underlie the network disruption characteristic for ASD [377, 378]. A new and interesting finding is that there appears to be a major change in the levels of various metabolites in the PFC of autistic individuals [379].
Attention deficit/hyperactivity disorder (ADHD) is a NDD characterized by signs of inattention, impulsivity, and hyperactivity [380, 381]. Control processes mediated by the PFC are hampered. Imaging studies have shown the PFC to be thinner in ADHD individuals, thereby hampering proper maturation and prefrontal connectivity leading to attentional dysfunction [382, 383]. Loss of catecholaminergic innervation underlies the most important ADHD symptoms [381].
Epilepsy, specifically childhood frontal lobe epilepsy (FLE) has various clinical outcomes but most often resulting in multi-cognitive symptoms [384,385,386,387]. Elevated prefrontal oscillations and hippocampal-prefrontal theta coherence could be observed after FLE [388]. And eventually the FLE seizures can cause structural and functional damage of the prefrontal areas including altered short-term plasticity [389, 390]. Important is also that the anti-epileptic drugs, given at young age or during pregnancy, can have major neurodevelopmental implications as well. These drugs act upon the major neurotranmitter and second messenger systems including ion channels, thereby affecting neurodevelopmental events [391,392,393].
Future research directions
The multidisciplinary nature of the field of developmental neurobiology has made enormous progress in recent years. The combination of classical (immuno)histological techniques with physiological, behavioral and high power molecular approaches such as large-scale genome-wide (single-cell) transcriptome and epigenome profiling studies have brought us an enormous amount of insight and resolution into the development and evolution of developing brain areas, specifically the PFC and their role in the onset of NDDs [21, 394]. Advances in molecular labeling and imaging techniques have added to this understanding [4, 395]. But maybe the most exciting field is the rapid emergence of stem cell approaches such as the generation of brain organoids that have led to some tremendous breakthroughs. A dazzling number of studies in gyrencephalic species have led to scientific breakthroughs and the description of novel types of cortical progenitors, including the basal and outer RGCs, both of which have been linked to cortical expansion and folding. Early features of corticogenesis can be recapitulated reliably; however the later stages in development still need to be optimized.
It has become clear that most neurotransmitter systems play neurotrophic roles during neurodevelopment as well. More holistic studies into the extrasynaptic neurotrophic functions of neurotransmitters during prefrontal development might also provide more understanding of their potential roles in the etiology of NDDs and eventually will enable us to design critical developmental windows in which we may be able to intervene. In the future, more longitudinal as well as interspecies studies will be needed to corroborate our understanding of prefrontal development.
Abnormal PFC development may lead to a variety of behavioral and cognitive problems inherent to psychiatric disorders including NDDs. In order to create tailored interventions targeted to the specific genetic syndromes, there is a strong need for research into the specific developmental and behavioral aspects accompanying these syndromes. A better understanding of the underlying neurodevelopmental and biological mechanisms will open doors to investigate the possibility of therapeutic (early/preventive) interventions and subsequent improvement of care.
References
Fuster JM. Prefrontal neurons in networks of executive memory. Brain Res Bull. 2000a;52:331–6.
Fuster JM. Memory networks in the prefrontal cortex. Prog brain Res. 2000b;122:309–16.
Goldman-Rakic PS. Circuitry of the primate prefrontal cortex and the regulation of behavior by representational memory. Handbook of Physiology, The Nervous System, Higher Functions of the Brain. In: Plum F, editor. Handbook of physiology, the nervous system, higher functions of the brain. Bethesda: American Physiological Society; 1987. pp. 373–417.
Li H, Yan G, Luo W, Liu T, Wang Y, Liu R, et al. Mapping fetal brain development based on automated segmentation and 4D brain atlasing. Brain Struct Funct. 2021;226:1961–72.
Flak JN, Solomon MB, Jankord R, Krause EG, Herman JP. Identification of chronic stress-activated regions reveals a potential recruited circuit in rat brain. Eur J Neurosci. 2012;36:2547–55.
Izquierdo A, Wellman CL, Holmes A. Brief uncontrollable stress causes dendritic retraction in infralimbic cortex and resistance to fear extinction in mice. J Neurosci. 2006;26:5733–8.
Moench KM, Maroun M, Kavushansky A, Wellman C. Alterations in neuronal morphology in infralimbic cortex predict resistance to fear extinction following acute stress. Neurobiol Stress. 2016;3:23–33.
Dias-Ferreira E, Sousa JC, Melo I, Morgado P, Mesquita AR, Cerqueira JJ, et al. Chronic stress causes frontostriatal reorganization and affects decision-making. Science 2009;325:621–5.
Shansky RM, Hamo C, Hof PR, McEwen BS, Morrison JH. Stress-induced dendritic remodeling in the prefrontal cortex is circuit specific. Cereb Cortex. 2009;19:2479–84.
Radley JJ, Rocher AB, Miller M, Janssen WG, Liston C, Hof PR, et al. Repeated stress induces dendritic spine loss in the rat medial prefrontal cortex. Cereb Cortex. 2006;16:313–20.
McKlveen JM, Morano RL, Fitzgerald M, Zoubovsky S, Cassella SN, Scheimann JR, et al. Chronic stress increases prefrontal inhibition: a mechanism for stress-induced prefrontal dysfunction. Biol Psychiatry. 2016;80:754–64.
Petanjek Z, Judas M, Simic G, Rasin MR, Uylings HB, Rakic P, et al. Extraordinary neoteny of synaptic spines in the human prefrontal cortex. Proc Natl Acad Sci USA. 2011;108:13281–6.
Donahue CJ, Glasser MF, Preuss TM, Rilling JK, Van, Essen DC. Quantitative assessment of prefrontal cortex in humans relative to nonhuman primates. Proc Natl Acad Sci USA. 2018;115:E5183–E92.
Povinelli DJ, Preuss TM. Theory of mind: evolutionary history of a cognitive specialization. Trends Neurosci. 1995;18:418–24.
Carlen M. What constitutes the prefrontal cortex? Science 2017;358:478–82.
Uylings HB, Groenewegen HJ, Kolb B. Do rats have a prefrontal cortex? Behav Brain Res. 2003;146:3–17.
Preuss TM. Do rats have prefrontal cortex? The rose-woolsey-akert program reconsidered. J Cogn Neurosci. 1995;7:1–24.
Wise SP. Forward frontal fields: phylogeny and fundamental function. Trends Neurosci. 2008;31:599–608.
Warren JM. Evolution, behavior and the prefrontal cortex. Acta Neurobiol Exp. 1972;32:581–93.
Llinares-Benadero C, Borrell V. Deconstructing cortical folding: genetic, cellular and mechanical determinants. Nat Rev Neurosci. 2019;20:161–76.
Johnson MB, Kawasawa YI, Mason CE, Krsnik Z, Coppola G, Bogdanovic D, et al. Functional and evolutionary insights into human brain development through global transcriptome analysis. Neuron. 2009;62:494–509.
Pletikos M, Sousa AM, Sedmak G, Meyer KA, Zhu Y, Cheng F, et al. Temporal specification and bilaterality of human neocortical topographic gene expression. Neuron. 2014;81:321–32.
Stiles J, Jernigan TL. The basics of brain development. Neuropsychol Rev. 2010;20:327–48.
Rakic P, Suner I, Williams RW. A novel cytoarchitectonic area induced experimentally within the primate visual cortex. Proc Natl Acad Sci USA. 1991;88:2083–7.
Rubenstein JL, Rakic P. Genetic control of cortical development. Cereb Cortex. 1999;9:521–3.
Elsen GE, Hodge RD, Bedogni F, Daza RA, Nelson BR, Shiba N, et al. The protomap is propagated to cortical plate neurons through an Eomes-dependent intermediate map. Proc Natl Acad Sci USA. 2013;110:4081–6.
Hevner RF, Haydar TF. The (not necessarily) convoluted role of basal radial glia in cortical neurogenesis. Cereb Cortex. 2012;22:465–8.
Miyashita-Lin EM, Hevner R, Wassarman KM, Martinez S, Rubenstein JL. Early neocortical regionalization in the absence of thalamic innervation. Science. 1999;285:906–9.
O’Leary DD, Stocker, AM, Zembrycki A. Area patterning of the mammalian cortex. In: Rubenstein JL, Rakic P editors. Patterning and Cell Type Specification in the Developing CNS and Pns. San Diego: Elsevier. vol. 1, pp. 61–85.
Cholfin JA, Rubenstein JL. Frontal cortex subdivision patterning is coordinately regulated by Fgf8, Fgf17, and Emx2. J Comp Neurol. 2008;509:144–55.
Borrell V, Reillo I. Emerging roles of neural stem cells in cerebral cortex development and evolution. Dev Neurobiol. 2012;72:955–71.
Cholfin JA, Rubenstein JL. Patterning of frontal cortex subdivisions by Fgf17. Proc Natl Acad Sci USA. 2007;104:7652–7.
Rakic P. Specification of cerebral cortical areas. Science. 1988;241:170–6.
Geschwind DH, Rakic P. Cortical evolution: judge the brain by its cover. Neuron. 2013;80:633–47.
O’Leary DD. Do cortical areas emerge from a protocortex? Trends Neurosci. 1989;12:400–6.
Creutzfeldt OD. Generality of the functional structure of the neocortex. Die Naturwissenschaften. 1977;64:507–17.
Bystron I, Blakemore C, Rakic P. Development of the human cerebral cortex: Boulder Committee revisited. Nat Rev Neurosci. 2008;9:110–22.
Stancik EK, Navarro-Quiroga I, Sellke R, Haydar TF. Heterogeneity in ventricular zone neural precursors contributes to neuronal fate diversity in the postnatal neocortex. J Neurosci. 2010;30:7028–36.
Hilgetag CC, Barbas H. Developmental mechanics of the primate cerebral cortex. Anat Embryol. 2005;210:411–7.
Chenn A, Walsh CA. Regulation of cerebral cortical size by control of cell cycle exit in neural precursors. Science. 2002;297:365–9.
Kuida K, Haydar TF, Kuan CY, Gu Y, Taya C, Karasuyama H, et al. Reduced apoptosis and cytochrome c-mediated caspase activation in mice lacking caspase 9. Cell. 1998;94:325–37.
Huang C, Liu T, Wang Q, Hou W, Zhou C, Song Z, et al. Loss of PP2A disrupts the retention of radial glial progenitors in the telencephalic niche to impair the generation for late-born neurons during cortical developmentdagger. Cereb Cortex. 2020;30:4183–96.
Grasby KL, Jahanshad N, Painter JN, Colodro-Conde L, Bralten J, Hibar DP, et al. The genetic architecture of the human cerebral cortex. Science. 2020;367:6484.
Samuelsen GB, Larsen KB, Bogdanovic N, Laursen H, Graem N, Larsen JF, et al. The changing number of cells in the human fetal forebrain and its subdivisions: a stereological analysis. Cereb Cortex. 2003;13:115–22.
Oberst P, Fievre S, Baumann N, Concetti C, Bartolini G, Jabaudon D. Temporal plasticity of apical progenitors in the developing mouse neocortex. Nature. 2019;573:370–4.
Telley L, Agirman G, Prados J, Amberg N, Fievre S, Oberst P, et al. Temporal patterning of apical progenitors and their daughter neurons in the developing neocortex. Science. 2019;364:6440.
Rice D, Barone S Jr. Critical periods of vulnerability for the developing nervous system: evidence from humans and animal models. Environ Health Perspect. 2000;108:511–33.
Rakic P. Neurons in rhesus monkey visual cortex: systematic relation between time of origin and eventual disposition. Science. 1974;183:425–7.
Bourgeois JP, Goldman-Rakic PS, Rakic P. Synaptogenesis in the prefrontal cortex of rhesus monkeys. Cereb Cortex. 1994;4:78–96.
Noctor SC, Flint AC, Weissman TA, Dammerman RS, Kriegstein AR. Neurons derived from radial glial cells establish radial units in neocortex. Nature. 2001;409:714–20.
Miyata T, Kawaguchi A, Okano H, Ogawa M. Asymmetric inheritance of radial glial fibers by cortical neurons. Neuron. 2001;31:727–41.
Gogtay N, Giedd JN, Lusk L, Hayashi KM, Greenstein D, Vaituzis AC, et al. Dynamic mapping of human cortical development during childhood through early adulthood. Proc Natl Acad Sci USA. 2004;101:8174–9.
Ge WP, Miyawaki A, Gage FH, Jan YN, Jan LY. Local generation of glia is a major astrocyte source in postnatal cortex. Nature. 2012;484:376–80.
Kessaris N, Fogarty M, Iannarelli P, Grist M, Wegner M, Richardson WD. Competing waves of oligodendrocytes in the forebrain and postnatal elimination of an embryonic lineage. Nat Neurosci. 2006;9:173–9.
Muffat J, Li Y, Yuan B, Mitalipova M, Omer A, Corcoran S, et al. Efficient derivation of microglia-like cells from human pluripotent stem cells. Nat Med. 2016;22:1358–67.
Rakic P. Mode of cell migration to the superficial layers of fetal monkey neocortex. J Comp Neurol. 1972;145:61–83.
Sidman RL, Rakic P. Neuronal migration, with special reference to developing human brain: a review. Brain Res. 1973;62:1–35.
Paredes MF, James D, Gil-Perotin S, Kim H, Cotter JA, Ng C, et al. Sidman RLRP. Development of the human central nervous system. Haymaker WARD, editor. Sprinfield, IL: Thomas; 1982.
Nobrega-Pereira S, Marin O. Transcriptional control of neuronal migration in the developing mouse brain. Cereb Cortex. 2009;19:i107–13.
Paredes MF, James D, Gil-Perotin S, Kim H, Cotter JA, Ng C, et al. Extensive migration of young neurons into the infant human frontal lobe. Science. 2016;354:6308.
Shiba N, Daza RA, Shaffer LG, Barkovich AJ, Dobyns WB, Hevner RF. Neuropathology of brain and spinal malformations in a case of monosomy 1p36. Acta Neuropathol Commun. 2013;1:45.
Rakic P, Stensas LJ, Sayre E, Sidman RL. Computer-aided three-dimensional reconstruction and quantitative analysis of cells from serial electron microscopic montages of foetal monkey brain. Nature. 1974;250:31–4.
Schmechel DE, Rakic P. A Golgi study of radial glial cells in developing monkey telencephalon: morphogenesis and transformation into astrocytes. Anat Embryol. 1979;156:115–52.
Rakic P. Radial versus tangential migration of neuronal clones in the developing cerebral cortex. Proc Natl Acad Sci USA. 1995;92:11323–7.
Ayala R, Shu T, Tsai LH. Trekking across the brain: the journey of neuronal migration. Cell. 2007;128:29–43.
Lim L, Pakan JMP, Selten MM, Marques-Smith A, Llorca A, Bae SE, et al. Optimization of interneuron function by direct coupling of cell migration and axonal targeting. Nat Neurosci. 2018;21:920–31.
Mi D, Li Z, Lim L, Li M, Moissidis M, Yang Y, et al. Early emergence of cortical interneuron diversity in the mouse embryo. Science. 2018;360:81–5.
Kosik KS, Nowakowski T. Evolution of new miRNAs and cerebro-cortical development. Annu Rev Neurosci. 2018;41:119–37.
Pollen AA, Nowakowski TJ, Chen J, Retallack H, Sandoval-Espinosa C, Nicholas CR, et al. Molecular identity of human outer radial glia during cortical development. Cell. 2015;163:55–67.
Nowakowski TJ, Bhaduri A, Pollen AA, Alvarado B, Mostajo-Radji MA, Di Lullo E, et al. Spatiotemporal gene expression trajectories reveal developmental hierarchies of the human cortex. Science. 2017;358:1318–23.
Molnar Z, Clowry GJ, Sestan N, Alzu’bi A, Bakken T, Hevner RF, et al. New insights into the development of the human cerebral cortex. J Anat. 2019;235:432–51.
Molnar Z, Hoerder-Suabedissen A. Regional scattering of primate subplate. Proc Natl Acad Sci USA. 2016;113:9676–8.
Molnar Z, Kaas JH, de Carlos JA, Hevner RF, Lein E, Nemec P. Evolution and development of the mammalian cerebral cortex. Brain Behav Evol. 2014;83:126–39.
Molnar Z, Luhmann HJ, Kanold PO. Transient cortical circuits match spontaneous and sensory-driven activity during development. Science. 2020;370:6514.
Molnar Z, Pollen A. How unique is the human neocortex? Development. 2014;141:11–6.
Hansen DV, Lui JH, Parker PR, Kriegstein AR. Neurogenic radial glia in the outer subventricular zone of human neocortex. Nature. 2010;464:554–61.
Rash BG, Duque A, Morozov YM, Arellano JI, Micali N, Rakic P. Gliogenesis in the outer subventricular zone promotes enlargement and gyrification of the primate cerebrum. Proc Natl Acad Sci USA. 2019;116:7089–94.
Sherwood CC, Stimpson CD, Raghanti MA, Wildman DE, Uddin M, Grossman LI, et al. Evolution of increased glia-neuron ratios in the human frontal cortex. Proc Natl Acad Sci USA. 2006;103:13606–11.
Dombrowski SM, Hilgetag CC, Barbas H. Quantitative architecture distinguishes prefrontal cortical systems in the rhesus monkey. Cereb Cortex. 2001;11:975–88.
Hilgetag CC, Barbas H. Are there ten times more glia than neurons in the brain? Brain Struct Funct. 2009;213:365–6.
von Bartheld CS, Bahney J, Herculano-Houzel S. The search for true numbers of neurons and glial cells in the human brain: a review of 150 years of cell counting. J Comp Neurol. 2016;524:3865–95.
Kroon T, van Hugte E, van Linge L, Mansvelder HD, Meredith RM. Early postnatal development of pyramidal neurons across layers of the mouse medial prefrontal cortex. Sci Rep. 2019;9:5037.
Petanjek Z, Judas M, Kostovic I, Uylings HB. Lifespan alterations of basal dendritic trees of pyramidal neurons in the human prefrontal cortex: a layer-specific pattern. Cereb Cortex. 2008;18:915–29.
Mrzljak L, Uylings HB, Kostovic I, van Eden CG. Prenatal development of neurons in the human prefrontal cortex. II. A quantitative Golgi study. J Comp Neurol. 1992;316:485–96.
Yang JM, Zhang J, Yu YQ, Duan S, Li XM. Postnatal development of 2 microcircuits involving fast-spiking interneurons in the mouse prefrontal cortex. Cereb Cortex. 2014;24:98–109.
Cooper MA, Koleske AJ. Ablation of ErbB4 from excitatory neurons leads to reduced dendritic spine density in mouse prefrontal cortex. J Comp Neurol. 2014;522:3351–62.
Huttenlocher PR. Morphometric study of human cerebral cortex development. Neuropsychologia. 1990;28:517–27.
Kiryushko D, Berezin V, Bock E. Regulators of neurite outgrowth: role of cell adhesion molecules. Ann NY Acad Sci. 2004;1014:140–54.
Iafrati J, Orejarena MJ, Lassalle O, Bouamrane L, Gonzalez-Campo C, Chavis P. Reelin, an extracellular matrix protein linked to early onset psychiatric diseases, drives postnatal development of the prefrontal cortex via GluN2B-NMDARs and the mTOR pathway. Mol Psychiatry. 2014;19:417–26.
Saab BJ, Mansuy IM. Neuroepigenetics of memory formation and impairment: the role of microRNAs. Neuropharmacology. 2014;80:61–9.
Schratt G. microRNAs at the synapse. Nat Rev Neurosci. 2009;10:842–9.
Rakic P, Bourgeois JP, Eckenhoff MF, Zecevic N, Goldman-Rakic PS. Concurrent overproduction of synapses in diverse regions of the primate cerebral cortex. Science. 1986;232:232–5.
Huttenlocher PR, Dabholkar AS. Regional differences in synaptogenesis in human cerebral cortex. J Comp Neurol. 1997;387:167–78.
Lenroot RK, Giedd JN. Brain development in children and adolescents: insights from anatomical magnetic resonance imaging. Neurosci Biobehav Rev. 2006;30:718–29.
LaMantia AS, Rakic P. Axon overproduction and elimination in the corpus callosum of the developing rhesus monkey. J Neurosci. 1990;10:2156–75.
LaMantia AS, Rakic P. Axon overproduction and elimination in the anterior commissure of the developing rhesus monkey. J Comp Neurol. 1994;340:328–36.
Rakic P, Riley KP. Overproduction and elimination of retinal axons in the fetal rhesus monkey. Science 1983;219:1441–4.
Meinecke DL, Rakic P. Expression of GABA and GABAA receptors by neurons of the subplate zone in developing primate occipital cortex: evidence for transient local circuits. J Comp Neurol. 1992;317:91–101.
Schwartz ML, Meinecke DL. Early expression of GABA-containing neurons in the prefrontal and visual cortices of rhesus monkeys. Cereb Cortex. 1992;2:16–37.
Tessier CR, Broadie K. Activity-dependent modulation of neural circuit synaptic connectivity. Front Mol Neurosci. 2009;2:8.
Keshavan MS, Anderson S, Pettegrew JW. Is schizophrenia due to excessive synaptic pruning in the prefrontal cortex? The Feinberg hypothesis revisited. J Psychiatr Res. 1994;28:239–65.
Mrzljak L, Uylings HB, Van Eden CG, Judas M. Neuronal development in human prefrontal cortex in prenatal and postnatal stages. Prog Brain Res. 1990;85:185–222.
Pattwell SS, Liston C, Jing D, Ninan I, Yang RR, Witztum J, et al. Dynamic changes in neural circuitry during adolescence are associated with persistent attenuation of fear memories. Nat Commun. 2016;7:11475.
Lidow MS, Goldman-Rakic PS, Gallager DW, Rakic P. Distribution of dopaminergic receptors in the primate cerebral cortex: quantitative autoradiographic analysis using [3H]raclopride, [3H]spiperone and [3H]SCH23390. Neuroscience 1991;40:657–71.
Lidow MS, Goldman-Rakic PS, Gallager DW, Rakic P. Quantitative autoradiographic mapping of serotonin 5-HT1 and 5-HT2 receptors and uptake sites in the neocortex of the rhesus monkey. J Comp Neurol. 1989;280:27–42.
Giedd JN, Blumenthal J, Jeffries NO, Castellanos FX, Liu H, Zijdenbos A, et al. Brain development during childhood and adolescence: a longitudinal MRI study. Nat Neurosci. 1999;2:861–3.
Barnea-Goraly N, Menon V, Eckert M, Tamm L, Bammer R, Karchemskiy A, et al. White matter development during childhood and adolescence: a cross-sectional diffusion tensor imaging study. Cereb Cortex. 2005;15:1848–54.
Beul SF, Barbas H, Hilgetag CC. A predictive structural model of the primate connectome. Sci Rep. 2017;7:43176.
Garcia-Cabezas MA, Zikopoulos B, Barbas H. The Structural Model: a theory linking connections, plasticity, pathology, development and evolution of the cerebral cortex. Brain Struct Funct. 2019;224:985–1008.
Barbas H, Garcia-Cabezas MA. How the prefrontal executive got its stripes. Curr Opin Neurobiol. 2016;40:125–34.
Cavada C, Company T, Tejedor J, Cruz-Rizzolo RJ, Reinoso-Suarez F. The anatomical connections of the macaque monkey orbitofrontal cortex. A review. Cereb Cortex. 2000;10:220–42.
Rapan L, Froudist-Walsh S, Niu M, Xu T, Funck T, Zilles K, et al. Multimodal 3D atlas of the macaque monkey motor and premotor cortex. NeuroImage. 2021;226:117574.
Verney C, Lebrand C, Gaspar P. Changing distribution of monoaminergic markers in the developing human cerebral cortex with special emphasis on the serotonin transporter. Anat Rec. 2002;267:87–93.
Bonnin A, Levitt P. Fetal, maternal, and placental sources of serotonin and new implications for developmental programming of the brain. Neuroscience. 2011;197:1–7.
Muzerelle A, Scotto-Lomassese S, Bernard JF, Soiza-Reilly M, Gaspar P. Conditional anterograde tracing reveals distinct targeting of individual serotonin cell groups (B5-B9) to the forebrain and brainstem. Brain Struct Funct. 2016;221:535–61.
Bang SJ, Jensen P, Dymecki SM, Commons KG. Projections and interconnections of genetically defined serotonin neurons in mice. Eur J Neurosci. 2012;35:85–96.
Teng T, Gaillard A, Muzerelle A, Gaspar P. EphrinA5 signaling is required for the distinctive targeting of raphe serotonin neurons in the forebrain. eNeuro. 2017;4:0327–16.
Bonnin A, Goeden N, Chen K, Wilson ML, King J, Shih JC, et al. A transient placental source of serotonin for the fetal forebrain. Nature. 2011;472:347–50.
Gaspar P, Cases O, Maroteaux L. The developmental role of serotonin: news from mouse molecular genetics. Nat Rev Neurosci. 2003;4:1002–12.
Buznikov GA, Lambert HW, Lauder JM. Serotonin and serotonin-like substances as regulators of early embryogenesis and morphogenesis. Cell Tissue Res. 2001;305:177–86.
Booij L, Tremblay RE, Szyf M, Benkelfat C. Genetic and early environmental influences on the serotonin system: consequences for brain development and risk for psychopathology. J Psychiatry Neurosci. 2015;40:5–18.
Janusonis S, Gluncic V, Rakic P. Early serotonergic projections to Cajal-Retzius cells: relevance for cortical development. J Neurosci. 2004;24:1652–9.
Garcia LP, Witteveen JS, Middelman A, van Hulten JA, Martens GJM, Homberg JR, et al. Perturbed developmental serotonin signaling affects prefrontal catecholaminergic innervation and cortical integrity. Mol Neurobiol. 2019;56:1405–20.
Radnikow G, Feldmeyer D, Lubke J. Axonal projection, input and output synapses, and synaptic physiology of Cajal-Retzius cells in the developing rat neocortex. J Neurosci. 2002;22:6908–19.
Hanswijk SI, Spoelder M, Shan L, Verheij MMM, Muilwijk OG, Li W, et al. Gestational factors throughout fetal neurodevelopment: the serotonin link. Int J Mol Sci. 2020;21:5850.
Homberg JR, Kolk SM, Schubert D. Editorial perspective of the Research Topic “Deciphering serotonin’s role in neurodevelopment”. Front Cell Neurosci. 2013;7:212.
Kinast K, Peeters D, Kolk SM, Schubert D, Homberg JR. Genetic and pharmacological manipulations of the serotonergic system in early life: neurodevelopmental underpinnings of autism-related behavior. Front Cell Neurosci. 2013;7:72.
Witteveen JS, Middelman A, van Hulten JA, Martens GJ, Homberg JR, Kolk SM. Lack of serotonin reuptake during brain development alters rostral raphe-prefrontal network formation. Front Cell Neurosci. 2013;7:143.
Trowbridge S, Narboux-Neme N, Gaspar P. Genetic models of serotonin (5-HT) depletion: what do they tell us about the developmental role of 5-HT? Anat Rec. 2011;294:1615–23.
Verney C, Berger B, Baulac M, Helle KB, Alvarez C. Dopamine-beta-hydroxylase-like immunoreactivity in the fetal cerebral cortex of the rat: Noradrenergic ascending pathways and terminal fields. Int J Developmental Neurosci. 1984;2:491–503.
Levitt P, Moore RY. Development of the noradrenergic innervation of neocortex. Brain Res. 1979;162:243–59.
Zecevic N, Verney C. Development of the catecholamine neurons in human embryos and fetuses, with special emphasis on the innervation of the cerebral cortex. J Comp Neurol. 1995;351:509–35.
Chandler DJ, Gao WJ, Waterhouse BD. Heterogeneous organization of the locus coeruleus projections to prefrontal and motor cortices. Proc Natl Acad Sci USA. 2014;111:6816–21.
Uematsu A, Tan BZ, Ycu EA, Cuevas JS, Koivumaa J, Junyent F, et al. Modular organization of the brainstem noradrenaline system coordinates opposing learning states. Nat Neurosci. 2017;20:1602–11.
Cerpa JC, Marchand AR, Coutureau E. Distinct regional patterns in noradrenergic innervation of the rat prefrontal cortex. J Chem Neuroanat. 2019;96:102–9.
Lauder JM, Bloom FE. Ontogeny of monoamine neurons in the locus coeruleus, Raphe nuclei and substantia nigra of the rat. I. Cell differentiation. J Comp Neurol. 1974;155:469–81.
Duncan CP, Seidler FJ, Lappi SE, Slotkin TA. Dual control of DNA synthesis by alpha- and beta-adrenergic mechanisms in normoxic and hypoxic neonatal rat brain. Brain Res Dev Brain Res. 1990;55:29–33.
Felten DL, Hallman H, Jonsson G. Evidence for a neurotropic role of noradrenaline neurons in the postnatal development of rat cerebral cortex. J Neurocytol. 1982;11:119–35.
Blue ME, Parnavelas JG. The effect of neonatal 6-hydroxydopamine treatment on synaptogenesis in the visual cortex of the rat. J Comp Neurol. 1982;205:199–205.
Parnavelas JG, Blue ME. The role of the noradrenergic system on the formation of synapses in the visual cortex of the rat. Brain Res. 1982;255:140–4.
Winzer-Serhan UH, Leslie FM. Expression of alpha2A adrenoceptors during rat neocortical development. J Neurobiol. 1999;38:259–69.
Naqui SZ, Harris BS, Thomaidou D, Parnavelas JG. The noradrenergic system influences the fate of Cajal-Retzius cells in the developing cerebral cortex. Brain Res Dev Brain Res. 1999;113:75–82.
Bear MF, Singer W. Modulation of visual cortical plasticity by acetylcholine and noradrenaline. Nature. 1986;320:172–6.
Moron JA, Brockington A, Wise RA, Rocha BA, Hope BT. Dopamine uptake through the norepinephrine transporter in brain regions with low levels of the dopamine transporter: evidence from knock-out mouse lines. J Neurosci. 2002;22:389–95.
Dwyer JB, Cardenas A, Franke RM, Chen Y, Bai Y, Belluzzi JD, et al. Prenatal nicotine sex-dependently alters adolescent dopamine system development. Transl Psychiatry. 2019;9:304.
Mirmiran M, Dijcks FA, Bos NP, Gorter JA, Van, der Werf D. Cortical neuron sensitivity to neurotransmitters following neonatal noradrenaline depletion. Int J Dev Neurosci. 1990;8:217–21.
Bortel A, Nowak P, Brus R. Neonatal DSP-4 treatment modifies GABAergic neurotransmission in the prefrontal cortex of adult rats. Neurotox Res. 2008;13:247–52.
Datta D, Yang ST, Galvin VC, Solder J, Luo F, Morozov YM, et al. Noradrenergic alpha1-adrenoceptor actions in the primate dorsolateral prefrontal cortex. J Neurosci. 2019;39:2722–34.
Oda S, Tsuneoka Y, Yoshida S, Adachi-Akahane S, Ito M, Kuroda M, et al. Immunolocalization of muscarinic M1 receptor in the rat medial prefrontal cortex. J Comp Neurol. 2018;526:1329–50.
Vijayraghavan S, Major AJ, Everling S. Muscarinic M1 receptor overstimulation disrupts working memory activity for rules in primate prefrontal cortex. Neuron. 2018;98:1256–68. e4
Galvin VC, Yang ST, Paspalas CD, Yang Y, Jin LE, Datta D, et al. Muscarinic M1 receptors modulate working memory performance and activity via KCNQ potassium channels in the primate prefrontal cortex. Neuron. 2020;106:649–61. e4
Celada P, Puig MV, Casanovas JM, Guillazo G, Artigas F. Control of dorsal raphe serotonergic neurons by the medial prefrontal cortex: Involvement of serotonin-1A, GABA(A), and glutamate receptors. J Neurosci. 2001;21:9917–29.
Saboory E, Ghasemi M, Mehranfard N. Norepinephrine, neurodevelopment and behavior. Neurochemistry Int. 2020;135:104706.
Berridge CW, Waterhouse BD. The locus coeruleus-noradrenergic system: modulation of behavioral state and state-dependent cognitive processes. Brain Res Brain Res Rev. 2003;42:33–84.
Lammel S, Hetzel A, Hackel O, Jones I, Liss B, Roeper J. Unique properties of mesoprefrontal neurons within a dual mesocorticolimbic dopamine system. Neuron. 2008;57:760–73.
Kolk SM, Gunput RA, Tran TS, van den Heuvel DM, Prasad AA, Hellemons AJ, et al. Semaphorin 3F is a bifunctional guidance cue for dopaminergic axons and controls their fasciculation, channeling, rostral growth, and intracortical targeting. J Neurosci. 2009;29:12542–57.
Kalsbeek A, De Bruin JP, Feenstra MG, Uylings HB. Age-dependent effects of lesioning the mesocortical dopamine system upon prefrontal cortex morphometry and PFC-related behaviors. Prog Brain Res. 1990;85:257–82. discussion 82-3
Kalsbeek A, Voorn P, Buijs RM, Pool CW, Uylings HB. Development of the dopaminergic innervation in the prefrontal cortex of the rat. J Comp Neurol. 1988;269:58–72.
Manitt C, Eng C, Pokinko M, Ryan RT, Torres-Berrio A, Lopez JP, et al. dcc orchestrates the development of the prefrontal cortex during adolescence and is altered in psychiatric patients. Transl Psychiatry. 2013;3:e338.
Reynolds LM, Pokinko M, Torres-Berrio A, Cuesta S, Lambert LC, Del Cid Pellitero E. et al. DCC receptors drive prefrontal cortex maturation by determining dopamine axon targeting in adolescence. Biol psychiatry. 2018;83:181–92.
Cuesta S, Nouel D, Reynolds LM, Morgunova A, Torres-Berrio A, White A, et al. Dopamine axon targeting in the nucleus accumbens in adolescence requires netrin-1. Front Cell Dev Biol. 2020;8:487.
Manitt C, Mimee A, Eng C, Pokinko M, Stroh T, Cooper HM, et al. The netrin receptor DCC is required in the pubertal organization of mesocortical dopamine circuitry. J Neurosci. 2011;31:8381–94.
Benes FM, Taylor JB, Cunningham MC. Convergence and plasticity of monoaminergic systems in the medial prefrontal cortex during the postnatal period: implications for the development of psychopathology. Cereb Cortex. 2000;10:1014–27.
Naneix F, Marchand AR, Di Scala G, Pape JR, Coutureau E. Parallel maturation of goal-directed behavior and dopaminergic systems during adolescence. J Neurosci. 2012;32:16223–32.
Rosenberg DR, Lewis DA. Postnatal maturation of the dopaminergic innervation of monkey prefrontal and motor cortices: a tyrosine hydroxylase immunohistochemical analysis. J Comp Neurol. 1995;358:383–400.
Lambe EK, Krimer LS, Goldman-Rakic PS. Differential postnatal development of catecholamine and serotonin inputs to identified neurons in prefrontal cortex of rhesus monkey. J Neurosci. 2000;20:8780–7.
Weickert CS, Webster MJ, Gondipalli P, Rothmond D, Fatula RJ, Herman MM, et al. Postnatal alterations in dopaminergic markers in the human prefrontal cortex. Neuroscience. 2007;144:1109–19.
Rothmond DA, Weickert CS, Webster MJ. Developmental changes in human dopamine neurotransmission: cortical receptors and terminators. BMC Neurosci. 2012;13:18.
Arnsten AF, Wang M, Paspalas CD. Dopamine’s actions in primate prefrontal cortex: challenges for treating cognitive disorders. Pharmacol Rev. 2015;67:681–96.
Yamaguchi T, Wang HL, Li X, Ng TH, Morales M. Mesocorticolimbic glutamatergic pathway. J Neurosci. 2011;31:8476–90.
Gorelova N, Mulholland PJ, Chandler LJ, Seamans JK. The glutamatergic component of the mesocortical pathway emanating from different subregions of the ventral midbrain. Cereb Cortex. 2012;22:327–36.
Kabanova A, Pabst M, Lorkowski M, Braganza O, Boehlen A, Nikbakht N, et al. Function and developmental origin of a mesocortical inhibitory circuit. Nat Neurosci. 2015;18:872–82.
Lavin A, Nogueira L, Lapish CC, Wightman RM, Phillips PE, Seamans JK. Mesocortical dopamine neurons operate in distinct temporal domains using multimodal signaling. J Neurosci. 2005;25:5013–23.
Yamasaki M, Takeuchi T. Locus coeruleus and dopamine-dependent memory consolidation. Neural Plasticity. 2017;2017:8602690.
Engelhard B, Finkelstein J, Cox J, Fleming W, Jang HJ, Ornelas S, et al. Specialized coding of sensory, motor and cognitive variables in VTA dopamine neurons. Nature. 2019;570:509–13.
Leung HC, Gore JC, Goldman-Rakic PS. Differential anterior prefrontal activation during the recognition stage of a spatial working memory task. Cereb Cortex. 2005;15:1742–9.
Noudoost B, Moore T. The role of neuromodulators in selective attention. Trends Cogn Sci. 2011;15:585–91.
Brozoski TJ, Brown RM, Rosvold HE, Goldman PS. Cognitive deficit caused by regional depletion of dopamine in prefrontal cortex of rhesus monkey. Science. 1979;205:929–32.
Money KM, Stanwood GD. Developmental origins of brain disorders: roles for dopamine. Front Cell Neurosci. 2013;7:260.
Kriegstein AR, Noctor SC. Patterns of neuronal migration in the embryonic cortex. Trends Neurosci. 2004;27:392–9.
Krimer LS, Goldman-Rakic PS. Prefrontal microcircuits: membrane properties and excitatory input of local, medium, and wide arbor interneurons. J Neurosci. 2001;21:3788–96.
Anderson SA, Eisenstat DD, Shi L, Rubenstein JL. Interneuron migration from basal forebrain to neocortex: dependence on Dlx genes. Science. 1997;278:474–6.
Ben-Ari Y. Excitatory actions of gaba during development: the nature of the nurture. Nat Rev Neurosci. 2002;3:728–39.
LoTurco JJ, Owens DF, Heath MJ, Davis MB, Kriegstein AR. GABA and glutamate depolarize cortical progenitor cells and inhibit DNA synthesis. Neuron. 1995;15:1287–98.
Wang DD, Kriegstein AR. Defining the role of GABA in cortical development. J Physiol. 2009;587:1873–9.
Owens DF, Boyce LH, Davis MB, Kriegstein AR. Excitatory GABA responses in embryonic and neonatal cortical slices demonstrated by gramicidin perforated-patch recordings and calcium imaging. J Neurosci. 1996;16:6414–23.
Ohtani N, Goto T, Waeber C, Bhide PG. Dopamine modulates cell cycle in the lateral ganglionic eminence. J Neurosci. 2003;23:2840–50.
Crandall JE, McCarthy DM, Araki KY, Sims JR, Ren JQ, Bhide PG. Dopamine receptor activation modulates GABA neuron migration from the basal forebrain to the cerebral cortex. J Neurosci. 2007;27:3813–22.
Kilb W. Development of the GABAergic system from birth to adolescence. Neuroscientist. 2012;18:613–30.
Romo-Parra H, Trevino M, Heinemann U, Gutierrez R. GABA actions in hippocampal area CA3 during postnatal development: differential shift from depolarizing to hyperpolarizing in somatic and dendritic compartments. J Neurophysiol. 2008;99:1523–34.
Erickson SL, Lewis DA. Postnatal development of parvalbumin- and GABA transporter-immunoreactive axon terminals in monkey prefrontal cortex. J Comp Neurol. 2002;448:186–202.
Cruz DA, Eggan SM, Lewis DA. Postnatal development of pre- and postsynaptic GABA markers at chandelier cell connections with pyramidal neurons in monkey prefrontal cortex. J Comp Neurol. 2003;465:385–400.
Radonjic NV, Ayoub AE, Memi F, Yu X, Maroof A, Jakovcevski I, et al. Diversity of cortical interneurons in primates: the role of the dorsal proliferative niche. Cell Rep. 2014;9:2139–51.
Fertuzinhos S, Krsnik Z, Kawasawa YI, Rasin MR, Kwan KY, Chen JG, et al. Selective depletion of molecularly defined cortical interneurons in human holoprosencephaly with severe striatal hypoplasia. Cereb Cortex. 2009;19:2196–207.
Verwer RW, Van Vulpen EH, Van Uum JF. Postnatal development of amygdaloid projections to the prefrontal cortex in the rat studied with retrograde and anterograde tracers. J Comp Neurol. 1996;376:75–96.
Bacon SJ, Headlam AJ, Gabbott PL, Smith AD. Amygdala input to medial prefrontal cortex (mPFC) in the rat: a light and electron microscope study. Brain Res. 1996;720:211–9.
Carr DB, Sesack SR. Hippocampal afferents to the rat prefrontal cortex: synaptic targets and relation to dopamine terminals. J Comp Neurol. 1996;369:1–15.
Hoover WB, Vertes RP. Anatomical analysis of afferent projections to the medial prefrontal cortex in the rat. Brain Struct Funct. 2007;212:149–79.
Wang J, John Y, Barbas H. Pathways for contextual memory: the primate hippocampal pathway to anterior cingulate cortex. Cerebral Cortex. 2021;31:1807–26.
Ghashghaei HT, Hilgetag CC, Barbas H. Sequence of information processing for emotions based on the anatomic dialogue between prefrontal cortex and amygdala. NeuroImage. 2007;34:905–23.
Perry BAL, Lomi E, Mitchell AS. Thalamocortical interactions in cognition and disease: the mediodorsal and anterior thalamic nuclei. Neurosci Biobehav Rev. 2021;130:162–77.
Homman-Ludiye J, Bourne JA. The medial pulvinar: function, origin and association with neurodevelopmental disorders. J Anat. 2019;235:507–20.
Shibata M, Pattabiraman K, Lorente-Galdos B, Andrijevic D, Xing X, Sousa AMM, et al. Regulation of prefrontal patterning, connectivity and synaptogenesis by retinoic acid. bioRxiv. 2019; https://www.biorxiv.org/content/10.1101/2019.12.31.891036v1.
Sesack SR, Pickel VM. Prefrontal cortical efferents in the rat synapse on unlabeled neuronal targets of catecholamine terminals in the nucleus accumbens septi and on dopamine neurons in the ventral tegmental area. J Comp Neurol. 1992;320:145–60.
Christie MJ, James LB, Beart PM. An excitant amino acid projection from the medial prefrontal cortex to the anterior part of nucleus accumbens in the rat. J Neurochem. 1985;45:477–82.
Gao M, Liu CL, Yang S, Jin GZ, Bunney BS, Shi WX. Functional coupling between the prefrontal cortex and dopamine neurons in the ventral tegmental area. J Neurosci. 2007;27:5414–21.
Warden MR, Selimbeyoglu A, Mirzabekov JJ, Lo M, Thompson KR, Kim SY, et al. A prefrontal cortex-brainstem neuronal projection that controls response to behavioural challenge. Nature. 2012;492:428–32.
Geddes SD, Assadzada S, Lemelin D, Sokolovski A, Bergeron R, Haj-Dahmane S, et al. Target-specific modulation of the descending prefrontal cortex inputs to the dorsal raphe nucleus by cannabinoids. Proc Natl Acad Sci USA. 2016;113:5429–34.
Pollak Dorocic I, Furth D, Xuan Y, Johansson Y, Pozzi L, Silberberg G, et al. A whole-brain atlas of inputs to serotonergic neurons of the dorsal and median raphe nuclei. Neuron. 2014;83:663–78.
Janiesch PC, Kruger HS, Poschel B, Hanganu-Opatz IL. Cholinergic control in developing prefrontal-hippocampal networks. J Neurosci. 2011;31:17955–70.
Dupont E, Hanganu IL, Kilb W, Hirsch S, Luhmann HJ. Rapid developmental switch in the mechanisms driving early cortical columnar networks. Nature. 2006;439:79–83.
Matthews DA, Nadler JV, Lynch GS, Cotman CW. Development of cholinergic innervation in the hippocampal formation of the rat. I. Histochemical demonstration of acetylcholinesterase activity. Dev Biol. 1974;36:130–41.
Hanganu IL, Staiger JF, Ben-Ari Y, Khazipov R. Cholinergic modulation of spindle bursts in the neonatal rat visual cortex in vivo. J Neurosci. 2007;27:5694–705.
Hellstrom-Lindahl E, Gorbounova O, Seiger A, Mousavi M, Nordberg A. Regional distribution of nicotinic receptors during prenatal development of human brain and spinal cord. Brain Res Dev Brain Res. 1998;108:147–60.
Atluri P, Fleck MW, Shen Q, Mah SJ, Stadfelt D, Barnes W, et al. Functional nicotinic acetylcholine receptor expression in stem and progenitor cells of the early embryonic mouse cerebral cortex. Dev Biol. 2001;240:143–56.
Ma W, Maric D, Li BS, Hu Q, Andreadis JD, Grant GM, et al. Acetylcholine stimulates cortical precursor cell proliferation in vitro via muscarinic receptor activation and MAP kinase phosphorylation. Eur J Neurosci. 2000;12:1227–40.
Zheng JQ, Felder M, Connor JA, Poo MM. Turning of nerve growth cones induced by neurotransmitters. Nature. 1994;368:140–4.
Kostovic I, Goldman-Rakic PS. Transient cholinesterase staining in the mediodorsal nucleus of the thalamus and its connections in the developing human and monkey brain. J Comp Neurol. 1983;219:431–47.
Kostovic I, Rakic P. Development of prestriate visual projections in the monkey and human fetal cerebrum revealed by transient cholinesterase staining. J Neurosci. 1984;4:25–42.
Mechawar N, Descarries L. The cholinergic innervation develops early and rapidly in the rat cerebral cortex: a quantitative immunocytochemical study. Neuroscience. 2001;108:555–67.
Mesulam MM, Hersh LB, Mash DC, Geula C. Differential cholinergic innervation within functional subdivisions of the human cerebral cortex: a choline acetyltransferase study. J Comp Neurol. 1992;318:316–28.
Kostovic I, Skavic J, Strinovic D. Acetylcholinesterase in the human frontal associative cortex during the period of cognitive development: early laminar shifts and late innervation of pyramidal neurons. Neurosci Lett. 1988;90:107–12.
Lewis DA. Distribution of choline acetyltransferase-immunoreactive axons in monkey frontal cortex. Neuroscience. 1991;40:363–74.
Konsolaki E, Skaliora I. Premature aging phenotype in mice lacking high-affinity nicotinic receptors: region-specific changes in layer V pyramidal cell morphology. Cereb Cortex. 2015;25:2138–48.
Ballesteros-Yanez I, Benavides-Piccione R, Bourgeois JP, Changeux JP, DeFelipe J. Alterations of cortical pyramidal neurons in mice lacking high-affinity nicotinic receptors. Proc Natl Acad Sci USA. 2010;107:11567–72.
Bailey CD, Alves NC, Nashmi R, De Biasi M, Lambe EK. Nicotinic alpha5 subunits drive developmental changes in the activation and morphology of prefrontal cortex layer VI neurons. Biol Psychiatry. 2012;71:120–8.
Tseng KY, O’Donnell P. Dopamine-glutamate interactions controlling prefrontal cortical pyramidal cell excitability involve multiple signaling mechanisms. J Neurosci. 2004;24:5131–9.
Tseng KY, O’Donnell P. Post-pubertal emergence of prefrontal cortical up states induced by D1-NMDA co-activation. Cereb Cortex. 2005;15:49–57.
Ruediger T, Bolz J. Neurotransmitters and the development of neuronal circuits. Adv Exp Med Biol. 2007;621:104–15.
Hensler JG, Artigas F, Bortolozzi A, Daws LC, De Deurwaerdere P, Milan L, et al. Catecholamine/Serotonin interactions: systems thinking for brain function and disease. Adv Pharmacol. 2013;68:167–97.
Galvin VC, Arnsten AFT, Wang M. Evolution in neuromodulation-the differential roles of acetylcholine in higher order association vs. primary visual cortices. Front Neural Circuits. 2018;12:67.
Di Giovanni G, Di Matteo V, Esposito E. Serotonin-dopamine interaction: experimental evidence and therapeutic relevance. Preface. Prog Brain Res. 2008;172:ix.
Di Giovanni G, Di Matteo V, Pierucci M, Esposito E. Serotonin-dopamine interaction: electrophysiological evidence. Prog Brain Res. 2008;172:45–71.
Esposito E, Di Matteo V, Di Giovanni G. Serotonin-dopamine interaction: an overview. Prog Brain Res. 2008;172:3–6.
D’Amato RJ, Blue ME, Largent BL, Lynch DR, Ledbetter DJ, Molliver ME, et al. Ontogeny of the serotonergic projection to rat neocortex: transient expression of a dense innervation to primary sensory areas. Proc Natl Acad Sci USA. 1987;84:4322–6.
Zhou FC, Sari Y, Zhang JK. Expression of serotonin transporter protein in developing rat brain. Brain Res Dev Brain Res. 2000;119:33–45.
Narboux-Neme N, Pavone LM, Avallone L, Zhuang X, Gaspar P. Serotonin transporter transgenic (SERTcre) mouse line reveals developmental targets of serotonin specific reuptake inhibitors (SSRIs). Neuropharmacology. 2008;55:994–1005.
Altamura C, Dell’Acqua ML, Moessner R, Murphy DL, Lesch KP, Persico AM. Altered neocortical cell density and layer thickness in serotonin transporter knockout mice: a quantitation study. Cereb Cortex. 2007;17:1394–401.
Tribollet E, Bertrand D, Marguerat A, Raggenbass M. Comparative distribution of nicotinic receptor subtypes during development, adulthood and aging: an autoradiographic study in the rat brain. Neuroscience. 2004;124:405–20.
Gaspar P, Bloch B, Le, Moine C. D1 and D2 receptor gene expression in the rat frontal cortex: cellular localization in different classes of efferent neurons. Eur J Neurosci. 1995;7:1050–63.
Rosenfeld CS. The placenta-brain-axis. J Neurosci Res. 2021;99:271–83.
Vitalis T, Parnavelas JG. The role of serotonin in early cortical development. Dev Neurosci. 2003;25:245–56.
Fuster JM. Frontal lobe and cognitive development. J Neurocytol. 2002;31:373–85.
Sowell ER, Thompson PM, Holmes CJ, Jernigan TL, Toga AW. In vivo evidence for post-adolescent brain maturation in frontal and striatal regions. Nat Neurosci. 1999;2:859–61.
Spear LP. The adolescent brain and age-related behavioral manifestations. Neurosci Biobehav Rev. 2000;24:417–63.
Jacobs B, Schall M, Prather M, Kapler E, Driscoll L, Baca S, et al. Regional dendritic and spine variation in human cerebral cortex: a quantitative golgi study. Cereb Cortex. 2001;11:558–71.
Travis K, Ford K, Jacobs B. Regional dendritic variation in neonatal human cortex: a quantitative Golgi study. Dev Neurosci. 2005;27:277–87.
Oga T, Elston GN, Fujita I. Postnatal dendritic growth and spinogenesis of layer-V pyramidal cells differ between visual, inferotemporal, and prefrontal cortex of the macaque monkey. Front Neurosci. 2017;11:118.
Elston GN, Benavides-Piccione R, Elston A, Manger PR, Defelipe J. Pyramidal cells in prefrontal cortex of primates: marked differences in neuronal structure among species. Front Neuroanat. 2011;5:2.
Tsujimoto S. The prefrontal cortex: functional neural development during early childhood. Neuroscientist. 2008;14:345–58.
Lin Q, Wei W, Coelho CM, Li X, Baker-Andresen D, Dudley K, et al. The brain-specific microRNA miR-128b regulates the formation of fear-extinction memory. Nat Neurosci. 2011;14:1115–7.
Darcq E, Warnault V, Phamluong K, Besserer GM, Liu F, Ron D. MicroRNA-30a-5p in the prefrontal cortex controls the transition from moderate to excessive alcohol consumption. Mol Psychiatry. 2015;20:1261.
Caballero A, Granberg R, Tseng KY. Mechanisms contributing to prefrontal cortex maturation during adolescence. Neurosci Biobehav Rev. 2016;70:4–12.
Flensborg-Madsen T, Mortensen EL. Language development and intelligence in midlife. Br J Dev Psychol. 2019;37:269–83.
Demetriou A, Spanoudis G. From cognitive development to intelligence: translating developmental mental milestones into intellect. J Intell. 2017;5:30.
Moghaddam B. Stress activation of glutamate neurotransmission in the prefrontal cortex: implications for dopamine-associated psychiatric disorders. Biol Psychiatry. 2002;51:775–87.
Popoli M, Yan Z, McEwen BS, Sanacora G. The stressed synapse: the impact of stress and glucocorticoids on glutamate transmission. Nat Rev Neurosci. 2011;13:22–37.
Duman RS, Aghajanian GK. Synaptic dysfunction in depression: potential therapeutic targets. Science. 2012;338:68–72.
Thompson SM, Kallarackal AJ, Kvarta MD, Van Dyke AM, LeGates TA, Cai X. An excitatory synapse hypothesis of depression. Trends Neurosci. 2015;38:279–94.
Kang HJ, Voleti B, Hajszan T, Rajkowska G, Stockmeier CA, Licznerski P, et al. Decreased expression of synapse-related genes and loss of synapses in major depressive disorder. Nat Med. 2012;18:1413–7.
McEwen BS, Morrison JH. The brain on stress: vulnerability and plasticity of the prefrontal cortex over the life course. Neuron. 2013;79:16–29.
Morin EL, Howell BR, Feczko E, Earl E, Pincus M, Reding K, et al. Developmental outcomes of early adverse care on amygdala functional connectivity in nonhuman primates. Dev Psychopathol. 2020;32:1579–96.
Niemi MEK, Martin HC, Rice DL, Gallone G, Gordon S, Kelemen M, et al. Common genetic variants contribute to risk of rare severe neurodevelopmental disorders. Nature. 2018;562:268–71.
Pieretti M, Zhang FP, Fu YH, Warren ST, Oostra BA, Caskey CT, et al. Absence of expression of the FMR-1 gene in fragile X syndrome. Cell. 1991;66:817–22.
Verheij C, Bakker CE, de Graaff E, Keulemans J, Willemsen R, Verkerk AJ, et al. Characterization and localization of the FMR-1 gene product associated with fragile X syndrome. Nature. 1993;363:722–4.
Siegel JJ, Chitwood RA, Ding JM, Payne C, Taylor W, Gray R, et al. Prefrontal cortex dysfunction in fragile X mice depends on the continued absence of fragile X mental retardation protein in the adult brain. J Neurosci. 2017;37:7305–17.
Howell CJ, Sceniak MP, Lang M, Krakowiecki W, Abouelsoud FE, Lad SU, et al. Activation of the medial prefrontal cortex reverses cognitive and respiratory symptoms in a mouse model of rett syndrome. eNeuro. 0277-17. 2017:1–12.
Armstrong DD, Dunn K, Antalffy B. Decreased dendritic branching in frontal, motor and limbic cortex in Rett syndrome compared with trisomy 21. J Neuropathol Exp Neurol. 1998;57:1013–7.
Liu Z, Li X, Zhang JT, Cai YJ, Cheng TL, Cheng C, et al. Autism-like behaviours and germline transmission in transgenic monkeys overexpressing MeCP2. Nature. 2016;530:98–102.
Yu ZX, Wang DY, Xu XH. Gene editing to the rescue: reversal of social deficits associated with MECP2 duplication. Neurosci Bull. 2020;36:567–9.
Yu B, Yuan B, Dai JK, Cheng TL, Xia SN, He LJ, et al. Reversal of social recognition deficit in adult mice with MECP2 duplication via normalization of MeCP2 in the medial prefrontal cortex. Neurosci Bull. 2020;36:570–84.
Alarcon JM, Malleret G, Touzani K, Vronskaya S, Ishii S, Kandel ER, et al. Chromatin acetylation, memory, and LTP are impaired in CBP+/- mice: a model for the cognitive deficit in Rubinstein-Taybi syndrome and its amelioration. Neuron. 2004;42:947–59.
van Dongen LCM, Wingbermuhle E, van der Veld WM, Vermeulen K, Bos-Roubos AG, Ockeloen CW, et al. Exploring the behavioral and cognitive phenotype of KBG syndrome. Genes Brain Behav. 2019;18:e12553.
van Dongen LCM, Wingbermuhle PAM, van der Veld WM, Stumpel C, Kleefstra T, Egger JIM. Exploring the cognitive phenotype of Kabuki (Niikawa-Kuroki) syndrome. J Intellect Disabil Res. 2019;63:498–506.
Schut EHS, Alonso A, Smits S, Khamassi M, Samanta A, Negwer M, et al. The Object Space Task reveals increased expression of cumulative memory in a mouse model of Kleefstra syndrome. Neurobiol Learn Mem. 2020;173:107265.
Egger JI, Zwanenburg RJ, van Ravenswaaij-Arts CM, Kleefstra T, Verhoeven WM. Neuropsychological phenotype and psychopathology in seven adult patients with Phelan-McDermid syndrome: implications for treatment strategy. Genes Brain Behav. 2016;15:395–404.
Sidorov MS, Judson MC, Kim H, Rougie M, Ferrer AI, Nikolova VD, et al. Enhanced operant extinction and prefrontal excitability in a mouse model of angelman syndrome. J Neurosci. 2018;38:2671–82.
Laje G, Bernert R, Morse R, Pao M, Smith AC. Pharmacological treatment of disruptive behavior in Smith-Magenis syndrome. Am J Med Genet Part C Semin Med Genet. 2010;154C:463–8.
Laje G, Morse R, Richter W, Ball J, Pao M, Smith AC. Autism spectrum features in Smith-Magenis syndrome. Am J Med Genet Part C Semin Med Genet. 2010;154C:456–62.
Proulx E, Young EJ, Osborne LR, Lambe EK. Enhanced prefrontal serotonin 5-HT(1A) currents in a mouse model of Williams-Beuren syndrome with low innate anxiety. J Neurodev Disord. 2010;2:99–108.
Witteveen JS, Willemsen MH, Dombroski TC, van Bakel NH, Nillesen WM, van Hulten JA, et al. Haploinsufficiency of MeCP2-interacting transcriptional co-repressor SIN3A causes mild intellectual disability by affecting the development of cortical integrity. Nat Genet. 2016;48:877–87.
Frega M, Linda K, Keller JM, Gumus-Akay G, Mossink B, van Rhijn JR, et al. Neuronal network dysfunction in a model for Kleefstra syndrome mediated by enhanced NMDAR signaling. Nat Commun. 2019;10:4928.
Boisgontier J, Tacchella JM, Lemaitre H, Lehman N, Saitovitch A, Gatinois V, et al. Anatomical and functional abnormalities on MRI in kabuki syndrome. NeuroImage Clin. 2019;21:101610.
Jacot-Descombes S, Keshav NU, Dickstein DL, Wicinski B, Janssen WGM, Hiester LL, et al. Altered synaptic ultrastructure in the prefrontal cortex of Shank3-deficient rats. Mol Autism. 2020;11:89.
Celen C, Chuang JC, Luo X, Nijem N, Walker AK, Chen F, et al. Arid1b haploinsufficient mice reveal neuropsychiatric phenotypes and reversible causes of growth impairment. eLife. 2017;6:e25730:1–22.
Rotaru DC, van Woerden GM, Wallaard I, Elgersma Y. Adult Ube3a gene reinstatement restores the electrophysiological deficits of prefrontal cortex layer 5 neurons in a mouse model of angelman syndrome. J Neurosci. 2018;38:8011–30.
Huang WH, Wang DC, Allen WE, Klope M, Hu H, Shamloo M, et al. Early adolescent Rai1 reactivation reverses transcriptional and social interaction deficits in a mouse model of Smith-Magenis syndrome. Proc Natl Acad Sci USA. 2018;115:10744–9.
Tamm L, Menon V, Reiss AL. Abnormal prefrontal cortex function during response inhibition in Turner syndrome: functional magnetic resonance imaging evidence. Biol Psychiatry. 2003;53:107–11.
Wilfert AB, Sulovari A, Turner TN, Coe BP, Eichler EE. Recurrent de novo mutations in neurodevelopmental disorders: properties and clinical implications. Genome Med. 2017;9:101.
Marin-Padilla M. Structural abnormalities of the cerebral cortex in human chromosomal aberrations: a Golgi study. Brain Res. 1972;44:625–9.
Jay V, Chan FW, Becker LE. Dendritic arborization in the human fetus and infant with the trisomy 18 syndrome. brain Res Developmental brain Res. 1990;54:291–4.
Marin-Padilla M. Pyramidal cell abnormalities in the motor cortex of a child with Down’s syndrome. A Golgi study. J Comp Neurol. 1976;167:63–81.
Contestabile A, Benfenati F, Gasparini L. Communication breaks-Down: from neurodevelopment defects to cognitive disabilities in Down syndrome. Prog Neurobiol. 2010;91:1–22.
Chakrabarti L, Galdzicki Z, Haydar TF. Defects in embryonic neurogenesis and initial synapse formation in the forebrain of the Ts65Dn mouse model of Down syndrome. J Neurosci: Off J Soc Neurosci. 2007;27:11483–95.
Cheng A, Haydar TF, Yarowsky PJ, Krueger BK. Concurrent generation of subplate and cortical plate neurons in developing trisomy 16 mouse cortex. Developmental Neurosci. 2004;26:255–65.
Chang P, Bush D, Schorge S, Good M, Canonica T, Shing N, et al. Altered hippocampal-prefrontal neural dynamics in mouse models of Down syndrome. Cell Rep. 2020;30:1152–63.e4.
Chailangkarn T, Trujillo CA, Freitas BC, Hrvoj-Mihic B, Herai RH, Yu DX, et al. A human neurodevelopmental model for Williams syndrome. Nature 2016;536:338–43.
Hrvoj-Mihic B, Hanson KL, Lew CH, Stefanacci L, Jacobs B, Bellugi U, et al. Basal dendritic morphology of cortical pyramidal neurons in Williams syndrome: prefrontal cortex and beyond. Front Neurosci. 2017;11:419.
Lew CH, Brown C, Bellugi U, Semendeferi K. Neuron density is decreased in the prefrontal cortex in Williams syndrome. Autism Res. 2017;10:99–112.
Aman LCS, Manning KE, Whittington JE, Holland AJ. Mechanistic insights into the genetics of affective psychosis from Prader-Willi syndrome. Lancet Psychiatry. 2018;5:370–8.
Manning KE, Tait R, Suckling J, Holland AJ. Grey matter volume and cortical structure in Prader-Willi syndrome compared to typically developing young adults. NeuroImage Clin. 2018;17:899–909.
Kates WR, Burnette CP, Bessette BA, Folley BS, Strunge L, Jabs EW, et al. Frontal and caudate alterations in velocardiofacial syndrome (deletion at chromosome 22q11.2). J Child Neurol. 2004;19:337–42.
Antshel KM, Fremont W, Kates WR. The neurocognitive phenotype in velo-cardio-facial syndrome: a developmental perspective. Dev Disabilities Res Rev. 2008;14:43–51.
Kates WR, Antshel K, Willhite R, Bessette BA, AbdulSabur N, Higgins AM. Gender-moderated dorsolateral prefrontal reductions in 22q11.2 Deletion Syndrome: implications for risk for schizophrenia. Child Neuropsychol. 2005;11:73–85.
Tripathi A, Spedding M, Schenker E, Didriksen M, Cressant A, Jay TM. Cognition- and circuit-based dysfunction in a mouse model of 22q11.2 microdeletion syndrome: effects of stress. Transl Psychiatry. 2020;10:41.
Gudbrandsen M, Daly E, Murphy CM, Blackmore CE, Rogdaki M, Mann C, et al. Brain morphometry in 22q11.2 deletion syndrome: an exploration of differences in cortical thickness, surface area, and their contribution to cortical volume. Sci Rep. 2020;10:18845.
Rogdaki M, Gudbrandsen M, McCutcheon RA, Blackmore CE, Brugger S, Ecker C, et al. Magnitude and heterogeneity of brain structural abnormalities in 22q11.2 deletion syndrome: a meta-analysis. Mol Psychiatry. 2020;25:1704–17.
Nyaradi A, Foster JK, Hickling S, Li J, Ambrosini GL, Jacques A, et al. Prospective associations between dietary patterns and cognitive performance during adolescence. J Child Psychol Psychiatry Allied Discip. 2014;55:1017–24.
Maayan L, Hoogendoorn C, Sweat V, Convit A. Disinhibited eating in obese adolescents is associated with orbitofrontal volume reductions and executive dysfunction. Obesity. 2011;19:1382–7.
Nyaradi A, Oddy WH, Hickling S, Li J, Foster JK. The relationship between nutrition in infancy and cognitive performance during adolescence. Front Nutr. 2015;2:2.
Rincel M, Lepinay AL, Delage P, Fioramonti J, Theodorou VS, Laye S, et al. Maternal high-fat diet prevents developmental programming by early-life stress. Transl Psychiatry. 2016;6:e966.
Bronson SL, Chan JC, Bale TL. Sex-specific neurodevelopmental programming by placental insulin receptors on stress reactivity and sensorimotor gating. Biol Psychiatry. 2017;82:127–38.
Nousen EK, Franco JG, Sullivan EL. Unraveling the mechanisms responsible for the comorbidity between metabolic syndrome and mental health disorders. Neuroendocrinology. 2013;98:254–66.
Rivell A, Mattson MP. Intergenerational Metabolic Syndrome and Neuronal Network Hyperexcitability in Autism. Trends Neurosci. 2019;42:709–26.
Coles CD, Kalberg W, Kable JA, Tabachnick B, May PA, Chambers CD. Characterizing alcohol-related neurodevelopmental disorder: prenatal alcohol exposure and the spectrum of outcomes. Alcohol Clin Exp Res. 2020;44:1245–60.
Kable JA, Coles CD, Mattson SN. Neurodevelopmental outcomes associated with prefrontal cortical deoxygenation in children with fetal alcohol spectrum disorders. Dev Neuropsychol. 2020;45:1–16.
Tang S, Xu S, Waddell J, Zhu W, Gullapalli RP, Mooney SM. Functional connectivity and metabolic alterations in medial prefrontal cortex in a rat model of fetal alcohol spectrum disorder: a resting-state functional magnetic resonance imaging and in vivo proton magnetic resonance spectroscopy study. Dev Neurosci. 2019;41:67–78.
Wang X, Cuzon Carlson VC, Studholme C, Newman N, Ford MM, Grant KA, et al. In utero MRI identifies consequences of early-gestation alcohol drinking on fetal brain development in rhesus macaques. Proc Natl Acad Sci USA. 2020;117:10035–44.
Skorput AG, Gupta VP, Yeh PW, Yeh HH. Persistent interneuronopathy in the prefrontal cortex of young adult offspring exposed to ethanol in utero. J Neurosci. 2015;35:10977–88.
Skorput AG, Yeh HH. Effects of ethanol exposure in utero on Cajal-Retzius cells in the developing cortex. Alcohol Clin Exp Res. 2015;39:853–62.
Skorput AG, Yeh HH. Chronic gestational exposure to ethanol leads to enduring aberrances in cortical form and function in the medial prefrontal cortex. Alcohol Clin Exp Res. 2016;40:1479–88.
Nygaard E, Slinning K, Moe V, Due-Tonnessen P, Fjell A, Walhovd KB. Neuroanatomical characteristics of youths with prenatal opioid and poly-drug exposure. Neurotoxicol Teratol. 2018;68:13–26.
Bhide PG. Dopamine, cocaine and the development of cerebral cortical cytoarchitecture: a review of current concepts. Semin Cell Dev Biol. 2009;20:395–402.
Kubrusly RC, Bhide PG. Cocaine exposure modulates dopamine and adenosine signaling in the fetal brain. Neuropharmacology. 2010;58:436–43.
Stanwood GD, Levitt P. Drug exposure early in life: functional repercussions of changing neuropharmacology during sensitive periods of brain development. Curr Opin Pharmacol. 2004;4:65–71.
Cisler JM, Elton A, Kennedy AP, Young J, Smitherman S, Andrew James G, et al. Altered functional connectivity of the insular cortex across prefrontal networks in cocaine addiction. Psychiatry Res. 2013;213:39–46.
Li Z, Santhanam P, Coles CD, Ellen Lynch M, Hamann S, Peltier S, et al. Prenatal cocaine exposure alters functional activation in the ventral prefrontal cortex and its structural connectivity with the amygdala. Psychiatry Res. 2013;213:47–55.
Grewen K, Burchinal M, Vachet C, Gouttard S, Gilmore JH, Lin W, et al. Prenatal cocaine effects on brain structure in early infancy. NeuroImage. 2014;101:114–23.
Salzwedel AP, Grewen KM, Vachet C, Gerig G, Lin W, Gao W. Prenatal drug exposure affects neonatal brain functional connectivity. J Neurosci. 2015;35:5860–9.
Zhao T, Li C, Wei W, Zhang H, Ma D, Song X, et al. Prenatal ketamine exposure causes abnormal development of prefrontal cortex in rat. Sci Rep. 2016;6:26865.
Thomason ME, Hect JL, Rauh VA, Trentacosta C, Wheelock MD, Eggebrecht AT, et al. Prenatal lead exposure impacts cross-hemispheric and long-range connectivity in the human fetal brain. NeuroImage. 2019;191:186–92.
Kougias DG, Sellinger EP, Willing J, Juraska JM. Perinatal exposure to an environmentally relevant mixture of phthalates results in a lower number of neurons and synapses in the medial prefrontal cortex and decreased cognitive flexibility in adult male and female rats. J Neurosci. 2018;38:6864–72.
Woodward NC, Haghani A, Johnson RG, Hsu TM, Saffari A, Sioutas C, et al. Prenatal and early life exposure to air pollution induced hippocampal vascular leakage and impaired neurogenesis in association with behavioral deficits. Transl Psychiatry. 2018;8:261.
Ejaredar M, Nyanza EC, Ten Eycke K, Dewey D. Phthalate exposure and childrens neurodevelopment: a systematic review. Environ Res. 2015;142:51–60.
van de Wijer L, Garcia LP, Hanswijk SI, Rando J, Middelman A, Ter Heine R, et al. Neurodevelopmental and behavioral consequences of perinatal exposure to the HIV drug efavirenz in a rodent model. Transl Psychiatry. 2019;9:84.
Garcia LP, Van de Wijer L, Hanswijk SI, Rando J, Witteveen JS, Middelman A, et al. Perinatal exposure of rats to the HIV drug efavirenz affects medial prefrontal cortex cytoarchitecture. Biochemical Pharmacol. 2020;178:114050.
Stegmann BJ, Carey JC. TORCH Infections. Toxoplasmosis, Other (syphilis, varicella-zoster, parvovirus B19), Rubella, Cytomegalovirus (CMV), and Herpes infections. Curr Women’s Health Rep. 2002;2:253–8.
Mlakar J, Korva M, Tul N, Popovic M, Poljsak-Prijatelj M, Mraz J, et al. Zika virus associated with microcephaly. N Engl J Med. 2016;374:951–8.
Rubin EJ, Greene MF, Baden LR. Zika virus and microcephaly. N Engl J Med. 2016;374:984–5.
Molnar Z, Kennedy S. Neurodevelopmental disorders: risks of Zika virus during the first trimester of pregnancy. Nat Rev Neurol. 2016;12:315–6.
Onorati M, Li Z, Liu F, Sousa AMM, Nakagawa N, Li M, et al. Zika virus disrupts phospho-TBK1 localization and mitosis in human neuroepithelial stem cells and radial glia. Cell Rep. 2016;16:2576–92.
Li C, Xu D, Ye Q, Hong S, Jiang Y, Liu X, et al. Zika virus disrupts neural progenitor development and leads to microcephaly in mice. Cell Stem Cell. 2016;19:120–6.
Wu KY, Zuo GL, Li XF, Ye Q, Deng YQ, Huang XY, et al. Vertical transmission of Zika virus targeting the radial glial cells affects cortex development of offspring mice. Cell Res. 2016;26:645–54.
Raper J, Kovacs-Balint Z, Mavigner M, Gumber S, Burke MW, Habib J, et al. Long-term alterations in brain and behavior after postnatal Zika virus infection in infant macaques. Nat Commun. 2020;11:2534.
Lu Y, Li X, Geng D, Mei N, Wu PY, Huang CC, et al. Cerebral micro-structural changes in COVID-19 patients - an MRI-based 3-month follow-up study. EClinicalMedicine. 2020;25:100484.
Egbert AR, Cankurtaran S, Karpiak S. Brain abnormalities in COVID-19 acute/subacute phase: a rapid systematic review. Brain Behav Immun. 2020;89:543–54.
Kas A, Soret M, Pyatigoskaya N, Habert MO, Hesters A, Le Guennec L, et al. The cerebral network of COVID-19-related encephalopathy: a longitudinal voxel-based 18F-FDG-PET study. Eur J Nucl Med Mol Imaging. 2021;1–15.
Lopez-Diaz A, Ayesa-Arriola R, Crespo-Facorro B, Ruiz-Veguilla M. COVID-19 infection during pregnancy and risk of neurodevelopmental disorders in offspring: time for collaborative research. Biol Psychiatry. 2021;89:e29–e30.
Abbott A. Neuroscience: the brain, interrupted. Nature. 2015;518:24–6.
Komitova M, Xenos D, Salmaso N, Tran KM, Brand T, Schwartz ML, et al. Hypoxia-induced developmental delays of inhibitory interneurons are reversed by environmental enrichment in the postnatal mouse forebrain. J Neurosci. 2013;33:13375–87.
Fagel DM, Ganat Y, Cheng E, Silbereis J, Ohkubo Y, Ment LR, et al. Fgfr1 is required for cortical regeneration and repair after perinatal hypoxia. J Neurosci. 2009;29:1202–11.
Delcour M, Russier M, Amin M, Baud O, Paban V, Barbe MF, et al. Impact of prenatal ischemia on behavior, cognitive abilities and neuroanatomy in adult rats with white matter damage. Behav Brain Res. 2012;232:233–44.
Lefevre J, Germanaud D, Dubois J, Rousseau F, de Macedo Santos I, Angleys H, et al. Are developmental trajectories of cortical folding comparable between cross-sectional datasets of fetuses and preterm newborns? Cereb Cortex. 2016;26:3023–35.
Ang ES Jr., Gluncic V, Duque A, Schafer ME, Rakic P. Prenatal exposure to ultrasound waves impacts neuronal migration in mice. Proc Natl Acad Sci USA. 2006;103:12903–10.
Selemon LD, Ceritoglu C, Ratnanather JT, Wang L, Harms MP, Aldridge K, et al. Distinct abnormalities of the primate prefrontal cortex caused by ionizing radiation in early or midgestation. J Comp Neurol. 2013;521:1040–53.
Kovalchuk A, Kolb B. Low dose radiation effects on the brain - from mechanisms and behavioral outcomes to mitigation strategies. Cell Cycle. 2017;16:1266–70.
Alves MB, Laureano DP, Dalle Molle R, Machado TD, Salvador APA, Miguel PM, et al. Intrauterine growth restriction increases impulsive behavior and is associated with altered dopamine transmission in both medial prefrontal and orbitofrontal cortex in female rats. Physiol Behav. 2019;204:336–46.
Alves MB, Dalle Molle R, Desai M, Ross MG, Silveira PP. Increased palatable food intake and response to food cues in intrauterine growth-restricted rats are related to tyrosine hydroxylase content in the orbitofrontal cortex and nucleus accumbens. Behav Brain Res. 2015;287:73–81.
Vlassaks E, Gavilanes AW, Vles JS, Deville S, Kramer BW, Strackx E, et al. The effects of fetal and perinatal asphyxia on neuronal cytokine levels and ceramide metabolism in adulthood. J Neuroimmunol. 2013;255:97–101.
Nalivaeva NN, Turner AJ, Zhuravin IA. Role of prenatal hypoxia in brain development, cognitive functions, and neurodegeneration. Front Neurosci. 2018;12:825.
Allin M, Henderson M, Suckling J, Nosarti C, Rushe T, Fearon P, et al. Effects of very low birthweight on brain structure in adulthood. Dev Med Child Neurol. 2004;46:46–53.
Bjuland KJ, Lohaugen GC, Martinussen M, Skranes J. Cortical thickness and cognition in very-low-birth-weight late teenagers. Early Hum Dev. 2013;89:371–80.
Skranes J, Lohaugen GC, Martinussen M, Haberg A, Brubakk AM, Dale AM. Cortical surface area and IQ in very-low-birth-weight (VLBW) young adults. Cortex A J Devoted Study Nerv Syst Behav. 2013;49:2264–71.
Nosarti C, Nam KW, Walshe M, Murray RM, Cuddy M, Rifkin L, et al. Preterm birth and structural brain alterations in early adulthood. NeuroImage Clin. 2014;6:180–91.
Tokariev A, Stjerna S, Lano A, Metsaranta M, Palva JM, Vanhatalo S. Preterm birth changes networks of newborn cortical activity. Cereb Cortex. 2019;29:814–26.
Leikos S, Tokariev A, Koolen N, Nevalainen P, Vanhatalo S. Cortical responses to tactile stimuli in preterm infants. Eur J Neurosci. 2020;51:1059–73.
Willsey AJ, Sanders SJ, Li M, Dong S, Tebbenkamp AT, Muhle RA, et al. Coexpression networks implicate human midfetal deep cortical projection neurons in the pathogenesis of autism. Cell. 2013;155:997–1007.
Zikopoulos B, Barbas H. Changes in prefrontal axons may disrupt the network in autism. J Neurosci. 2010;30:14595–609.
Ariza J, Rogers H, Hashemi E, Noctor SC, Martinez-Cerdeno V. The number of chandelier and basket cells are differentially decreased in prefrontal cortex in autism. Cereb Cortex. 2018;28:411–20.
Hashemi E, Ariza J, Rogers H, Noctor SC, Martinez-Cerdeno V. The number of parvalbumin-expressing interneurons is decreased in the prefrontal cortex in autism. Cereb Cortex. 2018;28:690.
Courchesne E, Mouton PR, Calhoun ME, Semendeferi K, Ahrens-Barbeau C, Hallet MJ, et al. Neuron number and size in prefrontal cortex of children with autism. JAMA. 2011;306:2001–10.
Yang CJ, Tan HP, Du YJ. The developmental disruptions of serotonin signaling may involved in autism during early brain development. Neuroscience. 2014;267:1–10.
Supekar K, Uddin LQ, Khouzam A, Phillips J, Gaillard WD, Kenworthy LE, et al. Brain hyperconnectivity in children with autism and its links to social deficits. Cell Rep. 2013;5:738–47.
Solso S, Xu R, Proudfoot J, Hagler DJ Jr, Campbell K, Venkatraman V, et al. Diffusion tensor imaging provides evidence of possible axonal overconnectivity in frontal lobes in autism spectrum disorder toddlers. Biol Psychiatry. 2016;79:676–84.
Courchesne E, Pierce K. Why the frontal cortex in autism might be talking only to itself: local over-connectivity but long-distance disconnection. Curr Opin Neurobiol. 2005;15:225–30.
Petanjek Z, Sedmak D, Dzaja D, Hladnik A, Rasin MR, Jovanov-Milosevic N. The protracted maturation of associative layer IIIC pyramidal neurons in the human prefrontal cortex during childhood: a major role in cognitive development and selective alteration in autism. Front Psychiatry. 2019;10:122.
Garcia-Cabezas MA, Barbas H, Zikopoulos B. Parallel development of chromatin patterns, neuron morphology, and connections: potential for disruption in autism. Front Neuroanat. 2018;12:70.
Zikopoulos B, Liu X, Tepe J, Trutzer I, John YJ, Barbas H. Opposite development of short- and long-range anterior cingulate pathways in autism. Acta Neuropathologica. 2018;136:759–78.
Kurochkin I, Khrameeva E, Tkachev A, Stepanova V, Vanyushkina A, Stekolshchikova E, et al. Metabolome signature of autism in the human prefrontal cortex. Commun Biol. 2019;2:234.
Ambrosino S, de Zeeuw P, Wierenga LM, van Dijk S, Durston S. What can cortical development in attention-deficit/hyperactivity disorder teach us about the early developmental mechanisms involved? Cereb Cortex. 2017;27:4624–34.
Arnsten AF. The emerging neurobiology of attention deficit hyperactivity disorder: the key role of the prefrontal association cortex. J Pediatrics. 2009;154:I–S43.
Shaw P, Lerch J, Greenstein D, Sharp W, Clasen L, Evans A, et al. Longitudinal mapping of cortical thickness and clinical outcome in children and adolescents with attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 2006;63:540–9.
Shaw P, Rabin C. New insights into attention-deficit/hyperactivity disorder using structural neuroimaging. Curr Psychiatry Rep. 2009;11:393–8.
Neyens LG, Aldenkamp AP, Meinardi HM. Prospective follow-up of intellectual development in children with a recent onset of epilepsy. Epilepsy Res. 1999;34:85–90.
Holmes GL. Cognitive impairment in epilepsy: the role of network abnormalities. Epileptic Disord. 2015;17:101–16.
Korman B, Krsek P, Duchowny M, Maton B, Pacheco-Jacome E, Rey G. Early seizure onset and dysplastic lesion extent independently disrupt cognitive networks. Neurology. 2013;81:745–51.
Hernan AE, Alexander A, Jenks KR, Barry J, Lenck-Santini PP, Isaeva E, et al. Focal epileptiform activity in the prefrontal cortex is associated with long-term attention and sociability deficits. Neurobiol Dis. 2014;63:25–34.
Kleen JK, Wu EX, Holmes GL, Scott RC, Lenck-Santini PP. Enhanced oscillatory activity in the hippocampal-prefrontal network is related to short-term memory function after early-life seizures. J Neurosci. 2011;31:15397–406.
Kleen JK, Sesque A, Wu EX, Miller FA, Hernan AE, Holmes GL, et al. Early-life seizures produce lasting alterations in the structure and function of the prefrontal cortex. Epilepsy Behav. 2011;22:214–9.
Hernan AE, Holmes GL, Isaev D, Scott RC, Isaeva E. Altered short-term plasticity in the prefrontal cortex after early life seizures. Neurobiol Dis. 2013;50:120–6.
Kaindl AM, Asimiadou S, Manthey D, Hagen MV, Turski L, Ikonomidou C. Antiepileptic drugs and the developing brain. Cell Mol life Sci. 2006;63:399–413.
Veroniki AA, Cogo E, Rios P, Straus SE, Finkelstein Y, Kealey R, et al. Comparative safety of anti-epileptic drugs during pregnancy: a systematic review and network meta-analysis of congenital malformations and prenatal outcomes. BMC Med. 2017;15:95.
Veroniki AA, Rios P, Cogo E, Straus SE, Finkelstein Y, Kealey R, et al. Comparative safety of antiepileptic drugs for neurological development in children exposed during pregnancy and breast feeding: a systematic review and network meta-analysis. BMJ Open. 2017;7:e017248.
Zhong S, Zhang S, Fan X, Wu Q, Yan L, Dong J, et al. A single-cell RNA-seq survey of the developmental landscape of the human prefrontal cortex. Nature. 2018;555:524–8.
Kovacs-Balint ZA, Payne C, Steele J, Li L, Styner M, Bachevalier J, et al. Structural development of cortical lobes during the first 6 months of life in infant macaques. Developmental Cogn Neurosci. 2021;48:100906.
Silver DL, Rakic P, Grove EA, Haydar TF, Hensch TK, Huttner WB, et al. “Evolution and Ontogenetic Development of Cortical Structures” In Singer W, Sejnowski TJ, Rakic P. editors. The Neocortex, pp. 61–93. Strüngmann Forum Reports, vol. 27, J. R. Lupp, series editor. Cambridge, MA: MIT Press. 2019.
Funding
This review was supported by a NWA-ORC grant (ProMiSe NWA.1160.18.320) of the Dutch organization of scientific research to SK, and National Institutes of Health (USA) NIDA grant DA023999 to (PR).
Author information
Authors and Affiliations
Contributions
Both authors (SMK, PR) contributed equally to the writing of this manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kolk, S.M., Rakic, P. Development of prefrontal cortex. Neuropsychopharmacol. 47, 41–57 (2022). https://doi.org/10.1038/s41386-021-01137-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-021-01137-9
This article is cited by
-
olf413 an octopamine biogenesis pathway gene is required for axon growth and pathfinding during embryonic nervous system development in Drosophila melanogaster
BMC Research Notes (2024)
-
The Principle of Cortical Development and Evolution
Neuroscience Bulletin (2024)
-
The Association Between Emotion Recognition and Internalizing Problems in Children and Adolescents: A Three-Level Meta-Analysis
Journal of Youth and Adolescence (2024)
-
Early life stress, literacy and dyslexia: an evolutionary perspective
Brain Structure and Function (2024)
-
Interindividual Variability in Mental Fatigue-Related Impairments in Endurance Performance: A Systematic Review and Multiple Meta-regression
Sports Medicine - Open (2023)