Abstract
Repeated cycles of intoxication and withdrawal enhance the negative reinforcing properties of alcohol and lead to neuroadaptations that underlie withdrawal symptoms driving alcohol dependence. Pharmacotherapies that target these neuroadaptations may help break the cycle of dependence. The sigma-1 receptor (σ1R) subtype has attracted interest as a possible modulator of the rewarding and reinforcing effects of alcohol. However, whether the sigma-2 receptor, recently cloned and identified as transmembrane protein 97 (σ2R/TMEM97), plays a role in alcohol-related behaviors is currently unknown. Using a Caenorhabditis elegans model, we identified two novel, selective σ2R/Tmem97 modulators that reduce alcohol withdrawal behavior via an ortholog of σ2R/TMEM97. We then show that one of these compounds blunted withdrawal-induced excessive alcohol drinking in a well-established rodent model of alcohol dependence. These discoveries provide the first evidence that σ2R/TMEM97 is involved in alcohol withdrawal behaviors and that this receptor is a potential new target for treating alcohol use disorder.
Similar content being viewed by others
Introduction
Alcohol use disorder (AUD) has a lifetime prevalence as high as 29% in the US [1]. Unfortunately, drugs approved by the FDA to treat alcohol addiction have a high failure rate [2,3,4]. Accordingly, uncovering novel biochemical pathways underlying the disease is critical to breaking the cycle of dependence, is critical to breaking the cycle of dependence [5]. Drugs that mitigate symptoms of withdrawal may represent a promising approach because their severity is considered a major driving force for relapse [6].
Pharmacological studies have suggested that sigma receptors (σRs) may represent useful targets to counter substance abuse [7,8,9,10]. Originally mistaken as members of the opioid receptor family and as high affinity phencyclidine binding sites, two σR subtypes have been identified based on their ligand binding profiles, and more recently their genetic identity has been reported [11, 12]. The sigma-1 receptor (σ1R) was cloned over a decade ago, but the sigma-2 receptor was only recently cloned and identified as transmembrane protein 97 (σ2R/TMEM97) [11, 13]. Given that certain σ2R/Tmem97 ligands (receptor affinity defined in rat PC12 cells) appear to modulate the reinforcing, behavioral, and toxic effects of cocaine [14,15,16,17,18], we sought to test if known and novel σ2R/Tmem97 ligands might modulate alcohol-related behaviors and whether this action is actually mediated by σ2R/Tmem97.
Toward discovering novel compounds for biological screening, we developed a general platform for preparing diverse collections of substituted heterocyclic scaffolds, and we discovered a set of analogs of norbenzomorphans 1 and methanobenzazocines 2 (Fig. 1) that exhibit high affinity and selectivity for σ2R/Tmem97 [19,20,21,22]. Several of these σ2R/Tmem97 modulators show therapeutic effects in animal models of central nervous system (CNS) disorders [23, 24]. For example, the piperazine derivative SAS-0132 improves cognitive performance in a transgenic animal model of Alzheimer’s disease (AD) (Fig. 1a) [24], whereas the biaryls UKH-1114 and DKR-1051 reduce mechanical hypersensitivity in an animal model of neuropathic pain [23]. In a different study, DKR-1051 induced a rapid Ca2+ transient when applied to SKH-neuroblastoma cells that was blocked by SAS-0132 [24]. The clinical efficacy of the σ2R/TMEM97 binding molecules CT-1812 and MIN-101, which are drug candidates for AD and schizophrenia, respectively, indicate that targeting σ2R/TMEM97 may be a promising approach for developing therapeutics for certain unmet medical needs in neuroscience [25, 26].
In order to identify novel compounds that affect alcohol-related behaviors, we screened a small collection of analogs of 1 and 2 that are potent and selective σ2R/Tmem97 binding ligands [19, 20, 22]) in a model of alcohol withdrawal in the nematode Caenorhabditis elegans [27, 28]. The subtype-selective σ2R/Tmem97 ligands 3 and JVW-1034 improved behavioral impairments in worms withdrawn from chronic exposure to ethanol. The activity of these compounds was dependent upon both the worm TMEM97 ortholog that was identified during this study as well as a potential signaling partner of σ2R/Tmem97, the progesterone receptor membrane component 1 (PGRMC1). Furthermore, validating the predictive power of C. elegans, we discovered that treating ethanol-dependent rats with JVW-1034 during the ethanol withdrawal period significantly reduced voluntary ethanol intake, without affecting the low ethanol intake of non-dependent animals. The successful identification of a σ2R/Tmem97 ligand that is efficacious in a rodent model of alcohol dependence is, to our knowledge, the first evidence that σ2R/Tmem97 modulates the behavioral effects of chronic ethanol exposure and suggests that selective σ2R/Tmem97 modulators may be further developed into drug leads for treating AUD by a new mechanism of action.
Materials and methods
C. elegans ethanol exposure paradigm and compound screen
The ethanol withdrawal paradigm was modified from Mitchell et al. (2010) as described previously [28]. Briefly, L4-stage worms raised on the same plates were divided between ethanol-infused (+ethanol) and standard control (−ethanol) seeded plates. The plates were 6-cm diameter Petri dishes filled with 12-mL NGM-agar and seeded with OP50 bacteria. Ethanol plates (400 mM) were prepared by adding 280 µL of 200-proof ethanol (Sigma Aldrich) beneath the agar of the standard seeded plates and allowing the ethanol to soak into the agar. The plates were sealed with Parafilm and worms were exposed for ~24 h. C. elegans only absorbs a fraction of the high external concentration of ethanol when treated on standard plates [28, 29]. The internal concentration achieved by these methods, ~50 mM [28], is physiologically relevant to alcohol dependence in humans [30,31,32,33].
The ethanol-treated worms were withdrawn on seeded plates for 1 h returning the internal ethanol concentration of the worms to near baseline [28]. For drug-treatment experiments, these withdrawal plates contained either vehicle (DMSO) alone or compound dissolved in DMSO. The DMSO concentration was less than 0.2%. The compounds were screened at 100–200 µM due to poor absorption of pharmacological agents through the worm cuticle [34] and the limited 1-h treatment, which is far shorter than typical application durations in C. elegans [35, 36].
Following this 1-h withdrawal period, diacetyl races were performed as described previously [28]. The area under the curve (AUC) was calculated for the fraction of worms at goal versus time for each race. Unless otherwise noted, group means for AUC (in Table S2) were compared by one-way ANOVA with Holm-Sidak post hoc comparisons. In order to compare the magnitude of impairment during withdrawal between strains, the performance of withdrawn animals was normalized to the performance of the naive animals run in tandem. These normAUC values (in Table S3) were compared by one-way ANOVA with Holm-Sidak post hoc comparisons.
Internal ethanol measurements were estimated using previous methods [28]. The internal ethanol concentration was compared between WT worms treated with vehicle or JVW-1034 and between WT and PGRMC1/vem-1-overexpressing worms. Group means were compared with Student’s t-tests.
Rat ethanol exposure paradigm and compound testing
The test chambers used for operant oral ethanol self-administration (Med Associates, Inc., St. Albans, VT) were located in sound-attenuating, ventilated cubicles. Syringe pumps (Med Associates, St. Albans, VT) dispensed the solution into one of two stainless steel drinking cups mounted 2 cm above the grid floor in the middle of one side panel. Two retractable levers were located 3.2 cm to either side of the drinking cups. Fluid delivery and recording of operant responses were controlled by microcomputers. Each lever press resulted in delivery of 0.1 mL of either 10% w/v ethanol or water. Lever presses had no scheduled consequences for 2 s after the activation of the pumps to avoid double-lever hits.
Rats (N = 17) were allowed to learn to press a lever to obtain a 10% w/v ethanol solution in 1–3 overnight sessions (16 h) of operant self-administration with standard food available ad libitum. Thereafter, sessions lasted 30 min and were run daily, as previously reported [37,38,39]. After stabilization of performance, rats were divided into two groups, matched for body weight and ethanol intake. One group received chronic, intermittent exposure to ethanol vapor (CIE), and the other group was exposed to room air (“Controls”). CIE rats were made dependent by chronic, intermittent exposure to alcohol vapors; they were housed within sealed, clear plastic chambers (La Jolla Alcohol Research, Inc, CA) into which ethanol vapor was intermittently introduced, as described previously [38]. The chambers were connected to a timer that turned the ethanol vapor on (8:00 pm) and off (10:00 am), for 14 h of daily ethanol exposure. Tail blood was sampled at vapor offset for blood alcohol level (BAL) determination; plasma was assayed for alcohol content by injection into an oxygen-rate alcohol analyzer (Analox Instruments, Lunenburg, MA), and target BALs were 150–200 mg/dL. This paradigm induces dependence and increases operant ethanol self-administration during withdrawal [40, 41]. Control rats were kept under similar conditions without ethanol vapor exposure.
After 6 weeks of ethanol vapor exposure, rats returned to the operant ethanol self-administration sessions twice weekly, 6–10 h after vapor offset. At this timepoint, used previously by our as well as other laboratories, BALs have been shown to be negligible [38, 42, 43]. Once the ethanol responding stabilized, the effects of the σ2R/Tmem97 binding ligand JVW-1034 were tested. JVW-1034 was dissolved in (2-hydroxypropyl)-β-cyclodextrin in saline (20% w/v) and administered intraperitoneally (i.p.) 30 min before the test session, using a Latin square, within-subject design (0, 1, 3, 10 mg/kg). CIE rats were returned to the vapor chambers after each self-administration session. Drug treatments were separated by at least one treatment-free operant session to ensure ethanol responding had returned to baseline levels.
Self-administration data were analyzed using a two-way mixed design ANOVA, with Group as between-subject factor and Dose as within-subject factor. Pairwise post hoc comparisons were made using the Student Newman-Keuls test. Significance was set at p < 0.05. The software/graphic packages used were Statistica 7.0 and SigmaPlot 11.0.
Results
We screened our σ2R/Tmem97 binding molecules (see Fig. 1, S3 for their ability to restore behavioral function after withdrawal from chronic ethanol exposure in C. elegans. Withdrawal severity in C. elegans was assessed by behavioral performance in a chemotaxis race to the attractant diacetyl (Fig. 2a,b). Withdrawal-induced impairments in performance are reduced by low-dose ethanol [28], a fundamental trait of alcohol withdrawal in higher animals and humans [44,45,46]. Using this assay, we tested whether five σ2R/Tmem97 ligands of the biaryl series (Fig. 1; UKH-1114, DKR-1051,5–7) and three σ2R/Tmem97 ligands in the aryl chloride series (Figs. 1 and 2d; 3, JVW-1034, and 4) could reduce withdrawal-induced behavioral impairments. Vehicle-treated withdrawn worms showed significantly impaired performance in the diacetyl race compared to naive worms (p < 0.001; N = 61–77). Vehicle-treatment alone did not alter the performance of naive worms, nor did it exacerbate withdrawal-induced impairments in ethanol-treated worms (see Supplemental Table 2). The biaryl series exhibited no notable effects. In contrast, two of the three aryl chlorides, JVW-1034 and 3, significantly improved the performance of withdrawn worms (p < 0.001; N = 11–77). Notably, JVW-1034 improved performance to within the range of naive worms (Fig. 2d). Although low-dose ethanol can improve the performance of withdrawn worms, JVW-1034 did not restore behavioral function by limiting ethanol clearance. Worms treated with JVW-1034 or vehicle during withdrawal both showed negligible levels of internal ethanol before behavioral testing (vehicle-treated, 0.03 ± 0.43 mM; JVW-1034-treated, −0.26 ± 0.37 mM; t-test, n.s.; N = 5–6).
Select σ2R/Tmem97 ligands improve behavioral deficits during ethanol withdrawal in C. elegans. a Worms were exposed to ethanol for 24 h prior to withdrawal (red) or kept naive (black). Analogs of UKH-1114 (c) or 3 (d) were administered during a 1-hour withdrawal period to determine the effect on behavior. b During the behavioral assay, worms migrated towards a goal containing the volatile attractant diacetyl. c, d Withdrawal-induced behavioral impairment was substantially improved by treatment with JVW-1034 and 3. Group means for AUC were compared by one-way ANOVA with Holm-Sidak post hoc comparison to the withdrawn control (vehicle-treated) group (***p < 0.001)
We then compared the behavioral effects of 3 and JVW-1034 with two commercially-available σR ligands, siramesine and opipramol, both of which have a history of clinical use [47, 48] (Fig. 3). Opipramol did not alter behavioral performance in withdrawn worms (Fig. 3d; n.s., N = 11–77), whereas siramesine improved performance in worms similar to levels for JVW-1034 and 3 (Fig. 3d; p < 0.001; N = 9–77). While JVW-1034 and 3 appear to restore naive-like behavior, withdrawn worms treated with siramesine tended to move even faster than naive worms (Fig. 3e; p = 0.07; N = 9–61). Correspondingly, siramesine sped up performance of alcohol-naive worms, while neither JVW-1034 or 3 altered the behavior of alcohol-naive worms (Fig. 3e; JVW-1034, 3: n.s.; siramesine: p < 0.05; N = 7–43). Thus, in contrast to siramesine, JVW-1034 and 3 more specifically regulated alcohol-modified behavior.
JVW-1034 and 3 improve behavioral deficits during ethanol withdrawal without altering the behavior of worms naive to ethanol. Structure and σR affinities for novel ligands (a) and two commercially available σ2R/Tmem97 ligands, siramesine (b) and opipramol (c). Unlike the commercially available ligands, treatment with selective σ2R/Tmem97 ligands 3 and JVW-1034 improved behavioral performance during ethanol withdrawal (d), but did not affect performance of naive control worms (e). Group means for AUC compared by one-way ANOVA with Holm-Sidak post hoc comparison to vehicle-treated withdrawn (d) or naive (e) worms (***p < 0.001, *p < 0.05)
Compounds 3 and JVW-1034 exhibit similar high affinity and moderate within-family selectivity for σ2R/Tmem97 (Fig. 1), but they do not share off-target binding profiles for 45 other CNS proteins tested (Supplemental Table 1). This suggests that σ2R/Tmem97 binding, rather than an off-target interaction, is driving activity. We then further probed the specificity of action of these compounds in C. elegans. Though σ2R has been identified in mammals as Tmem97 [11], an ortholog of σ2R/TMEM97 had not yet been identified in C. elegans. Thus, we first identified a potential candidate, Y38H6C.16, which has 30% sequence similarity to σ2R/TMEM97, a similar predicted secondary structure, and at least partial conservation of the aspartate residues presumed to be important for ligand binding [11] (Figure S4a,b). The σ2R/TMEM97-like protein Y38H6C.16 appeared to not be required for alcohol withdrawal in C. elegans because a Y38H6C.16 null strain (allele gk910298) showed normal alcohol withdrawal (Fig. 4c; p < 0.05, N = 15). However, neither treatment with JVW-1034 nor 3 improved withdrawal behavior for the Y38H6C.16(null) mutant (Fig. 4c; n.s., N = 13–15). Importantly, reintroduction of either endogenous Y38H6C.16 or the human TMEM97 gene under the endogenous promoter and 3’ UTR rescued JVW-1034 efficacy (Fig. 4d,e; p < 0.01, N = 17–18; p < 0.05, N = 16, respectively). Collectively, these findings provide functional evidence that Y38H6C.16 is an ortholog of σ2R/TMEM97 and that it is required for the observed activity of JVW-1034 and 3.
Behavioral improvement in ethanol withdrawn worms requires human or worm orthologs of σ2R/TMEM97. a A strain carrying a null mutation in the worm ortholog of σ2R/TMEM97 showed normal withdrawal behavior (naive, black vs. withdrawn, red). However, treatment with JVW-1034 or 3 failed to improve the withdrawal-induced behavioral deficit. b, c Transgenic expression of worm or human σ2R/TMEM97 rescued the ability of JVW-1034 to reduce behavioral impairment during withdrawal from ethanol exposure. Group means for AUC compared by one-way ANOVA with Holm-Sidak post hoc comparison to vehicle-treated withdrawn worms (**p < 0.01, ***p < 0.001)
There is accumulating evidence of a functional relationship between σ2R/Tmem97 and the PGRMC1. Compounds that bind to σ2R/Tmem97 can affect PGRMC1-dependent mechanisms in cultured rat neurons, human brain tissue, and in worms [24, 49]. PGRMC1 is highly conserved from yeast through humans [50], including in worms, which express the PGRMC1 ortholog, VEM-1 [51]. To determine whether JVW-1034 improves ethanol withdrawal through a PGRMC1-mediated pathway, we tested the effects of JVW-1034 on strains carrying the independent vem-1 null alleles, gk220 and ok1058. Both PGRMC1/vem-1 strains showed normal withdrawal-induced behavioral impairment (Figure S5a,b; naive vs. vehicle-treated withdrawn worms: p < 0.001, N = 22–46). Notably, this impairment was not improved by treatment with JVW-1034 during withdrawal (Figure S5a,b; n.s., N = 17–46). Transformation of the vem-1(ok1058) strain with genomic vem-1 significantly reduced withdrawal impairments relative to WT (Figure S5b,c vs. Figure 3c; Supp. Table 3; post hoc comparisons of NormAUC p < 0.001, N = 8–24). However, only one transformant strain showed significantly impaired behavior for withdrawn worms relative to naive worms (Figure S5c,d; n.s., N = 8 and p < 0.001, N = 22, respectively). In this strain, treatment with JVW-1034 significantly improved behavioral performance in withdrawn animals (Figure S5d, p < 0.05, N = 22). Thus, reduction of withdrawal symptoms in C. elegans by JVW-1034 requires PGRMC1/VEM-1.
To further explore the in vivo function of JVW-1034, we considered how overexpression of PGRMC1/VEM-1 affected withdrawal. Extrachromosomal expression of genes leads to multi-copy expression in the range of ~2–60 copies [52]. Because extrachromosomal expression of worm PGRMC1(+) on the null background reduced withdrawal impairment relative to WT, it appears that a high copy number of PGRMC1/vem-1 reduced withdrawal symptoms. To ensure that the strains had more than two copies of PGRMC1/vem-1, the two independent extrachromosomal arrays were crossed from the PGRMC1/vem-1 null background to a WT background. The subsequent strains showed less impairment after withdrawal from chronic ethanol exposure than WT (Figure S5e,f vs. Figure 2c; Supp. Table 3; post hoc comparisons of NormAUC, p < 0.001, N = 9–24). Thus, like treatment with JVW-1034, multi-copy expression of PGRMC1 reduced behavioral impairments after withdrawal. Multi-copy overexpression of PGRMC1 did not limit withdrawal behavior by altering the steady state levels or clearance of ethanol. The internal ethanol concentration for WT and PGRMC1/VEM-1-overexpressing worms was similar after a 24-h treatment with ethanol (WT, 39.7 ± 3.34; pvem-1::vem-1(+)#1, 38.2 ± 1.77; t-test, n.s.; N = 5) and again after a 1-h withdrawal period (WT, −0.88 ± 0.26; pvem-1::vem-1(+)#1, −0.98 ± 0.39; t-test, n.s.; N = 5–6). Based upon the results of the receptor binding assays and the behavioral genetic studies, we presume that the effects of JVW-1034 arise from binding to the σ2R/TMEM97 ortholog in worms, thereby activating a PGRMC1/VEM-1-dependent mechanism and leading to the observed reduction in withdrawal impairments.
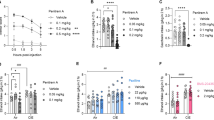
We predicted that compounds that improve withdrawal behavior in C. elegans would modulate alcohol behaviors in higher order animals. To test this hypothesis, we assessed the effects of JVW-1034 on withdrawal-induced excessive ethanol drinking in rats. Rats treated with a chronic-intermittent ethanol (CIE) paradigm showed excessive ethanol self-administration compared to control rats beginning from the third session, as shown in Fig. 5a. The i.p. administration of JVW-1034 dose-dependently blocked this excessive ethanol self-administration in CIE rats, but not in Controls (Fig. 5b, Group × Dose: F(3,45) = 3.05, p < 0.05; Dose: F(3,45) = 2.92, p < 0.05; Group: F(1,15) = 63.40, p < 0.001). Post hoc analysis revealed that the middle and highest doses of JVW-1034 both significantly reduced ethanol intake compared to the vehicle condition, with the 10 mg/kg dose producing a 32.7% decrease. As shown in Fig. 5c, lever pressing for water was unaffected by JVW-1034 treatment in either group (Group × Dose: F(3,45) = 2.42, n.s; Dose: F(3,45) = 0.20, n.s.; Group: F(1,15) = 0.05, n.s.), ruling out non-specific behavioral effects of the drug.
JVW-1034 reduces voluntary ethanol self-administration in ethanol-dependent rats. a Escalation of ethanol intake in rats exposed to chronic intermittent ethanol (CIE) vs. control (Ctrl.). CIE rats were exposed to 6 weeks of intermittent ethanol vapor (14 h/day) in which no operant sessions were given, correspondent to the dotted line. Then operant sessions resumed and were given twice weekly, 6–10 h into withdrawal from vapor. Ctrl. rats were exposed to air only. BL stands for baseline (intake before CIE exposure). #p < 0.05, ##p < 0.01, ###p < 0.001 vs. Ctrl. b When administered 30 min prior to testing, JVW-1034 caused a dose-dependent reduction in voluntary ethanol self-administration in CIE, but not Ctrl. animals. *p < 0.05, ***p < 0.01, vs. Vehicle. c Treatment with JVW-1034 did not alter water intake in either Ctrl. or CIE animals
Discussion
σ1R ligands have been recognized as modulators of the rewarding and reinforcing effects of alcohol and other drugs of abuse. However, the effect of σ2R/Tmem97-subtype ligands on alcohol-related behaviors was so far unknown. Here, we used a C. elegans platform to screen for novel high affinity σ2R/Tmem97 ligands that reduced an alcohol withdrawal behavior. We discovered that JVW-1034 and 3 specifically reduced alcohol withdrawal symptoms via σ2R/TMEM97 in C. elegans. Compounds JVW-1034 and 3 each reduce behavioral deficits during withdrawal from alcohol exposure in a manner dependent on the human or worm orthologs of σ2R/TMEM97, but did not alter the behavior of worms unexposed to alcohol. JVW-1034 was then tested in a mammalian model and found to block excessive alcohol self-administration dose-dependently in chronic intermittent alcohol-treated rats, but not in controls. These findings suggest that σ2R/Tmem97-mediated pathways modulate neuroadaptation to chronic alcohol exposure and that σ2R/Tmem97 can be targeted to lessen alcohol consumption in alcohol-dependent subjects.
Our results suggest there is a conserved σ2R/TMEM97-mediated pathway that also requires PGRMC1 for modulating neuroadaptation to alcohol because JVW-1034 and 3 required both the worm orthologs of σ2R/TMEM97 and PGRMC1 to alleviate withdrawal symptoms in C. elegans. Compound activity in the strain lacking the σ2R/TMEM97 ortholog was rescued by reintroduction of either the worm or human σ2R/TMEM97 gene, indicating that σ2R/TMEM97-signaling is conserved. PGRMC1 function and signaling pathways are also well conserved in C. elegans [51, 53]). These findings provide further support for a working hypothesis that there is a functional relationship between σ2R/Tmem97 and PGRMC1 [24, 49, 54, 55]. For example, previous reports have shown that σ2R/Tmem97 ligands modulate PGRMC1-dependent mechanisms to abate neurodegeneration and toxicity from either amyloid-β or extra amyloid precursor protein [24, 49].
Although the molecular underpinnings of alcohol-related behaviors are not fully understood in any organism, many mammalian genes and molecular pathways are conserved in C. elegans [56, 57], and C. elegans has been used successfully to identify or confirm gene targets for modifying alcohol behaviors [58,59,60]. Here we show that pharmacological alleviation of a worm alcohol-withdrawal behavior predicts a reduction in voluntary ethanol consumption in alcohol-dependent rats. The successful identification of a σ2R/Tmem97 modulator that is efficacious in a rodent model of alcohol dependence underscores the utility of C. elegans for chemical screens and target validation. Moreover, the conserved σ2R/TMEM97-dependent and the PGRMC-dependent pathways described in worms likely mediates the effects of σ2R/Tmem97 ligands in higher animal models of alcohol dependence. Future work can harness the power of C. elegans genetics to determine whether genes known to modulate alcohol withdrawal behaviors in worms and mammals [27, 28, 58] are part of the pathway through which σ2R/Tmem97 modulators act.
Our findings suggest that targeting σ2R/Tmem97 with JVW-1034 reduces the rewarding properties of alcohol consumption in rats. The transition to alcohol dependence can be modeled by exposing rodents to chronic, intermittent alcohol vapors. Prolonged and intermittent periods of high blood alcohol levels that commonly occur in human alcoholics trigger long-term dysregulations of brain neurochemical systems and lead to excessive voluntary drinking and the appearance of a negative emotional state characterized by anxiety-like behavior during withdrawal [61, 62]. Using this model with high face validity, we show that JVW-1034 significantly reduces excessive alcohol drinking selectively in alcohol-dependent rats, without affecting alcohol drinking in non-dependent rats or water drinking in either group of animals. These results indicate that JVW-1034 regulates one or more of the neuroadaptations caused by chronic alcohol exposure that promote and sustain the addictive behavior. Though we cannot completely rule out polypharmacological effects of JVW-1034 in these rodent experiments, our findings that JVW-1034 binds with high affinity to rat σ2R/Tmem97 and that JVW-1034 activity depends on σ2R/TMEM97 function in C. elegans strongly suggest that σ2R/TMEM97 is mediating the effects of the drug. However, future experiments involving the use of more selective compounds, perhaps coupled with genetic manipulation of σ2R/TMEM97, will be needed to definitively probe the role of σ2R/TMEM97 in voluntary ethanol consumption in rodents.
The selective reduction in alcohol drinking in alcohol-dependent rats with JVW-1034 treatment is likely due to activity in the CNS, even though these experiments involving systemic administration of the drug cannot completely rule out the contribution of peripheral effects. σ2R/Tmem97 and PGRMC1 are both expressed widely in the mammalian nervous system, including many structures of the mesolimbic reward pathway, the amygdala and frontocortical regions, which have been shown to contribute to both the positive and the negative reinforcing effects of alcohol [63,64,65,66]. Interestingly, some σ2R/Tmem97 ligands can modulate dopamine release in reward pathways [14], and glutamatergic transmission is affected by other σR ligands [16, 67]. How σ2R/Tmem97 influences neurotransmission is not yet known, but one possibility is by regulating intracellular calcium levels [68, 69]. PGRMC1 also influences intracellular calcium [70] and is detected in synaptosomes [71], potentially regulating calcium levels at the synapse.
Compared to other σR ligands that have been used in humans, JVW-1034 and 3 appear to be uniquely advantageous for modulating alcohol-related behaviors. In our C. elegans platform, opipramol was ineffective, due to either the wrong activity at the target or off-target activities, perhaps resulting from its high σ1R affinity [13]. Siramesine, which has high specificity for σ2R/Tmem97 relative to σ1R, reduced behavioral impairment during withdrawal. However, unlike JVW-1034 or 3, siramesine altered the behavior of alcohol-naive controls. Thus, the activity of JVW-1034 and 3 appears to be more selective to chronic alcohol-induced alterations of neural and molecular pathways.
AUD is a major medical problem, so it is critical to identify new pharmacological targets to break the cycle of dependence. The present findings are thus highly significant because they are the first to show that alcohol withdrawal-associated behaviors are mitigated by regulating a σ2R/TMEM97-mediated pathway not previously associated with alcohol abuse. Moreover, these findings suggest that small molecule modulators of σ2R/TMEM97 could be developed into novel drug candidates that might provide more effective treatments for the millions of people suffering from AUD.
References
Grant BF, Goldstein RB, Saha TD, Chou SP, Jung J, Zhang H, et al. Epidemiology of DSM-5 alcohol use disorder: results from the national epidemiologic survey on alcohol and related conditions III. JAMA Psychiatry. 2015;72:757–66.
Anton RF, O’Malley SS, Ciraulo DA, Cisler RA, Couper D, Donovan DM, et al. Combined pharmacotherapies and behavioral interventions for alcohol dependence: the COMBINE study: a randomized controlled trial. JAMA. 2006;295:2003–17.
Mann K, Lehert P, Morgan MY. The efficacy of acamprosate in the maintenance of abstinence in alcohol-dependent individuals: results of a meta-analysis. Alcohol Clin Exp Res. 2004;28:51–63.
Meyers RJ, Smith JE, Lash DN. The community reinforcement approach. Recent Dev Alcohol. 2003;16:183–95.
Becker HC, Mulholland PJ. Neurochemical mechanisms of alcohol withdrawal. Handb Clin Neurol. 2014;125:133–56.
Winward JL, Bekman NM, Hanson KL, Lejuez CW, Brown SA. Changes in emotional reactivity and distress tolerance among heavy drinking adolescents during sustained abstinence. Alcohol Clin Exp Res. 2014;38:1761–9.
Hiranita T, Soto PL, Tanda G, Katz JL. Reinforcing effects of sigma-receptor agonists in rats trained to self-administer cocaine. J Pharmacol Exp Ther. 2010;332:515–24.
Martin-Fardon R, Maurice T, Aujla H, Bowen WD, Weiss F. Differential effects of sigma1 receptor blockade on self-administration and conditioned reinstatement motivated by cocaine vs natural reward. Neuropsychopharmacology. 2007;32:1967–73.
Maurice T, Martin-Fardon R, Romieu P, Matsumoto RR. Sigma 1 receptor antagonists represent a new strategy against cocaine addiction and toxicity. Neurosci Biobehav Rev. 2002;26:499–527.
Nguyen EC, McCracken KA, Liu Y, Pouw B, Matsumoto RR. Involvement of sigma receptors in the acute actions of methamphetamine: receptor binding and behavioral studies. Neuropharmacology. 2005;49:638–45.
Alon A, Schmidt HR, Wood MD, Sahn JJ, Martin SF, Kruse AC. Identification of the gene that codes for the σ2 receptor. Proc Natl Acad Sci USA. 2017;114:7160–5.
Matsumoto RR. σ Receptors: Historical Perspective and Background. In: Su T-P, Matsumoto RR, Bowen WD, editors. Sigma receptors: Chemistry, cell biology and clinical implications. Boston, MA: Springer US; 2007. pp. 1–23.
Hanner M, Moebius FF, Flandorfer A, Knaus HG, Striessnig J, Kempner E, et al. Purification, molecular cloning, and expression of the mammalian sigma1-binding site. Proc Natl Acad Sci USA. 1996;93:8072–7.
Garces-Ramirez L, Green JL, Hiranita T, Kopajtic TA, Mereu M, Thomas AM, et al. Sigma receptor agonists: receptor binding and effects on mesolimbic dopamine neurotransmission assessed by microdialysis. Biol Psychiatry. 2011;69:208–17.
Kaushal N, Robson MJ, Vinnakota H, Narayanan S, Avery BA, McCurdy CR, et al. Synthesis and pharmacological evaluation of 6-acetyl-3-(4-(4-(4-fluorophenyl)piperazin-1-yl)butyl)benzo[d]oxazol-2(3H)-one (SN79), a cocaine antagonist, in rodents. AAPS J. 2011;13:336–46.
Klawonn AM, Nilsson A, Radberg CF, Lindstrom SH, Ericson M, Granseth B, et al. The Sigma-2 Receptor Selective Agonist Siramesine (Lu 28-179) decreases cocaine-reinforced pavlovian learning and alters glutamatergic and dopaminergic input to the striatum. Front Pharmacol. 2017;8:714.
Matsumoto RR, Pouw B, Mack AL, Daniels A, Coop A. Effects of UMB24 and (+/-)-SM 21, putative sigma2-preferring antagonists, on behavioral toxic and stimulant effects of cocaine in mice. Pharmacol Biochem Behav. 2007;86:86–91.
Mesangeau C, Narayanan S, Green AM, Shaikh J, Kaushal N, Viard E, et al. Conversion of a highly selective sigma-1 receptor-ligand to sigma-2 receptor preferring ligands with anticocaine activity. J Med Chem. 2008;51:1482–6.
Sahn JJ, Granger BA, Martin SF. Evolution of a strategy for preparing bioactive small molecules by sequential multicomponent assembly processes, cyclizations, and diversification. Org Biomol Chem. 2014;12:7659–72.
Sahn JJ, Hodges TR, Chan JZ, Martin SF. Norbenzomorphan framework as a novel scaffold for generating Sigma 2 Receptor/PGRMC1 subtype-selective ligands. Chem Med Chem. 2016;11:556–61.
Sahn JJ, Hodges TR, Chan JZ, Martin SF. Norbenzomorphan scaffold: chemical tool for modulating Sigma Receptor-subtype selectivity. ACS Med Chem Lett. 2017a;8:455–60.
Sahn JJ, Martin SF. Expedient synthesis of norbenzomorphan library via multicomponent assembly process coupled with ring-closing reactions. ACS Comb Sci. 2012;14:496–502.
Sahn JJ, Mejia GL, Ray PR, Martin SF, Price TJ. Sigma 2 receptor/Tmem97 agonists produce long lasting antineuropathic pain effects in mice. ACS Chem Neurosci. 2017b;8:1801–11.
Yi B, Sahn JJ, Ardestani PM, Evans AK, Scott L, Chan JZ, et al. Small molecule modulator of sigma 2 receptor is neuroprotective and reduces cognitive deficits and neuro-inflammation in experimental models of Alzheimer’s disease. J Neurochem. 2017;140:561–75.
Davidson M, Saoud J, Staner C, Noel N, Luthringer E, Werner S, et al. Efficacy and Safety of MIN-101: a 12-week randomized, double-blind, placebo-controlled trial of a new drug in development for the treatment of negative symptoms in schizophrenia. Am J Psych. 2017;174:1195–202.
Schneider LS, Grundman M, DeKosky S, Morgan R, Guttendorf R, Higgin M, et al. (2017). The Anti-Aβ Oligomer Drug CT1812 for Alzheimer’s: phase 1b/2a safety trial outcomes. 10th Clinical Trials on Alzheimer’s Disease Meeting, Boston, MA.
Mitchell P, Mould R, Dillon J, Glautier S, Andrianakis I, James C, et al. A differential role for neuropeptides in acute and chronic adaptive responses to alcohol: behavioural and genetic analysis in Caenorhabditis elegans. PLoS ONE. 2010;5:e10422.
Scott LL, Davis SJ, Yen RC, Ordemann GJ, Nordquist SK, Bannai D, et al. (2017). Behavioral deficits following withdrawal from chronic ethanol are influenced by SLO channel function in Caenorhabditis elegans. Genetics. 2017;206:1445–58
Alaimo JT, Davis SJ, Song SS, Burnette CR, Grotewiel M, Shelton KL, et al. Ethanol metabolism and osmolarity modify behavioral responses to ethanol in C. elegans. Alcohol Clin Exp Res. 2012;36:1840–50.
Lindblad B, Olsson R. Unusually high levels of blood alcohol? JAMA. 1976;236:1600–2.
Teplin LA, Abram KM, Michaels SK. Blood alcohol level among emergency room patients: a multivariate analysis. J Stud Alcohol. 1989;50:441–7.
Urso T, Gavaler JS, Van Thiel DH. Blood ethanol levels in sober alcohol users seen in an emergency room. Life Sci. 1981;28:1053–6.
Cartlidge D, Redmond AD. Alcohol and conscious level. Biomed Pharmacother. 1990;44:205–8.
Rand JB, Johnson CD. Genetic pharmacology: interactions between drugs and gene products in Caenorhabditis elegans. Methods Cell Biol. 1995;48:187–204.
Crisford A, Murray C, O’Connor V, Edwards RJ, Kruger N, Welz C, et al. Selective toxicity of the anthelmintic emodepside revealed by heterologous expression of human KCNMA1 in Caenorhabditis elegans. Mol Pharmacol. 2011;79:1031–43.
Kwok TC, Ricker N, Fraser R, Chan AW, Burns A, Stanley EF, et al. A small-molecule screen in C. elegans yields a new calcium channel antagonist. Nature. 2006;441:91–5.
Sabino V, Cottone P, Blasio A, Iyer MR, Steardo L, Rice KC, et al. Activation of sigma-receptors induces binge-like drinking in Sardinian alcohol-preferring rats. Neuropsychopharmacology. 2011;36:1207–18.
Sabino V, Cottone P, Zhao Y, Iyer MR, Steardo L Jr., Steardo L, et al. The sigma-receptor antagonist BD-1063 decreases ethanol intake and reinforcement in animal models of excessive drinking. Neuropsychopharmacology. 2009;34:1482–93.
Sabino V, Narayan AR, Zeric T, Steardo L, Cottone P. mTOR activation is required for the anti-alcohol effect of ketamine, but not memantine, in alcohol-preferring rats. Behav Brain Res. 2013;247:9–16.
Funk CK, Zorrilla EP, Lee MJ, Rice KC, Koob GF. Corticotropin-releasing factor 1 antagonists selectively reduce ethanol self-administration in ethanol-dependent rats. Biol Psychiatry. 2007;61:78–86.
O’Dell LE, Roberts AJ, Smith RT, Koob GF. Enhanced alcohol self-administration after intermittent versus continuous alcohol vapor exposure. Alcohol Clin Exp Res. 2004;28:1676–82.
Gilpin NW, Smith AD, Cole M, Weiss F, Koob GF, Richardson HN. Operant behavior and alcohol levels in blood and brain of alcohol-dependent rats. Alcohol Clin Exp Res. 2009;33:2113–23.
Rimondini R, Arlinde C, Sommer W, Heilig M. Long-lasting increase in voluntary ethanol consumption and transcriptional regulation in the rat brain after intermittent exposure to alcohol. FASEB J. 2002;16:27–35.
Heinz A, Schafer M, Higley JD, Krystal JH, Goldman D. Neurobiological correlates of the disposition and maintenance of alcoholism. Pharmacopsychiatry. 2003;36:S255–8.
Koob GF. Alcohol use disorders: tracts, twins, and trajectories. Am J Psychiatry. 2015;172:499–501.
Koob GF, Le Moal M. Plasticity of reward neurocircuitry and the ‘dark side’ of drug addiction. Nat Neurosci. 2005;8:1442–4.
Moller HJ, Volz HP, Reimann IW, Stoll KD. Opipramol for the treatment of generalized anxiety disorder: a placebo-controlled trial including an alprazolam-treated group. J Clin Psychopharmacol. 2001;21:59–65.
Volz HP, Stoll KD. Clinical trials with sigma ligands. Pharmacopsychiatry. 2004;37:S214–20.
Izzo NJ, Xu J, Zeng C, Kirk MJ, Mozzoni K, Silky C, et al. Alzheimer’s therapeutics targeting amyloid beta 1-42 oligomers II: Sigma-2/PGRMC1 receptors mediate Abeta 42 oligomer binding and synaptotoxicity. PLoS ONE. 2014;9:e111899.
Rohe HJ, Ahmed IS, Twist KE, Craven RJ. PGRMC1 (progesterone receptor membrane component 1): a targetable protein with multiple functions in steroid signaling, P450 activation and drug binding. Pharmacol Ther. 2009;121:14–9.
Runko E, Kaprielian Z. Caenorhabditis elegans VEM-1, a novel membrane protein, regulates the guidance of ventral nerve cord-associated axons. J Neurosci. 2004;24:9015–26.
Stinchcomb DT, Shaw JE, Carr SH, Hirsh D. Extrachromosomal DNA transformation of Caenorhabditis elegans. Mol Cell Biol. 1985;5:3484–96.
Cahill MA. Progesterone receptor membrane component 1: an integrative review. J Steroid Biochem Mol Biol. 2007;105:16–36.
Xu J, Zeng C, Chu W, Pan F, Rothfuss JM, Zhang F. et al. Identification of the PGRMC1 protein complex as the putative sigma-2 receptor binding site. Nat Commun. 2011;2:380
Zeng C, Garg N, Mach RH. The PGRMC1 Protein Level Correlates with the Binding Activity of a Sigma-2 Fluorescent Probe (SW120) in Rat Brain Cells. Mol Imaging Biol. 2016;18:172–9.
Kuwabara P, O’Neil N. The use of functional genomics in C. elegans for studying human development and disease. J Inherit Metab Dis. 2001;24:127–38.
The C. elegans Sequencing Consortium. Genome sequence of the nematode C. elegans: a platform for investigating biology. Science. 1998;282:2012–8.
Grotewiel M, Bettinger JC. Drosophila and Caenorhabditis elegans as discovery platforms for genes involved in human alcohol use disorder. Alcohol Clin Exp Res. 2015;39:1292–311.
Bettinger JC, Davies AG. The role of the BK channel in ethanol response behaviors: evidence from model organism and human studies. Front Physiol. 2014;5:346.
Vengeliene V, Bilbao A, Molander A, Spanagel R. Neuropharmacology of alcohol addiction. Br J Pharmacol. 2008;154:299–315.
Heilig M, Koob GF. A key role for corticotropin-releasing factor in alcohol dependence. Trends Neurosci. 2007;30:399–406.
Roberts AJ, Heyser CJ, Cole M, Griffin P, Koob GF. Excessive ethanol drinking following a history of dependence: animal model of allostasis. Neuropsychopharmacology. 2000;22:581–94.
Intlekofer KA, Petersen SL. Distribution of mRNAs encoding classical progestin receptor, progesterone membrane components 1 and 2, serpine mRNA binding protein 1, and progestin and ADIPOQ receptor family members 7 and 8 in rat forebrain. Neuroscience. 2011;172:55–65.
Lovinger DM, Crabbe JC. Laboratory models of alcoholism: treatment target identification and insight into mechanisms. Nat Neurosci. 2005;8:1471–80.
Ron D, Jurd R. The “ups and downs” of signaling cascades in addiction. Sci STKE. 2005;2005:re14.
Bouchard P, Quirion R. [3H]1,3-di(2-tolyl)guanidine and [3H](+)pentazocine binding sites in the rat brain: Autoradiographic visualization of the putative sigma1 and sigma2 receptor subtypes. Neuroscience. 1997;76:467–77.
Sabino V, Hicks C, Cottone P. Sigma receptors and substance use disorders. Adv Exp Med Biol. 2017;964:177–99.
Vilner BJ, Bowen WD. Modulation of cellular calcium by Sigma-2 receptors: release from intracellular stores in human SK-N-SH neuroblastoma cells. J Pharmacol Exp Ther. 2000;292:900.
Cassano G, Gasparre G, Contino M, Niso M, Berardi F, Perrone R, et al. The sigma-2 receptor agonist PB28 inhibits calcium release from the endoplasmic reticulum of SK-N-SH neuroblastoma cells. Cell Calcium. 2006;40:23–8.
Lai J-N, Wang OY-H, Lin VH-C, Liao C-F, Tarng D-C, Chien EJ. The non-genomic rapid acidification in peripheral T cells by progesterone depends on intracellular calcium increase and not on Na+/H+-exchange inhibition. Steroids. 2012;77:1017–24.
Munton RP, Tweedie-Cullen R, Livingstone-Zatchej M, Weinandy F, Waidelich M, Longo D, et al. Qualitative and quantitative analyses of protein phosphorylation in naive and stimulated mouse synaptosomal preparations. Mol Cell Proteom. 2007;6:283–93.
Acknowledgements
We thank the CGC for certain worm strains and the UNC Psychoactive Drug Screening Program (Bryan Roth and XP Huang) for receptor binding assays. This work was supported by The Robert A. Welch Foundation (F-0652) (SFM), the Dell Medical School’s Texas Health Catalyst program (SFM and JJS), donations from Tom Calhoon and Hornraiser (JTP), and NIAAA grants AA024439 (VS) and AA020992 (LS and JTP). Drs. Martin, Sahn, Hodges, Scott and Pierce and Mr. Wood report being co-inventors on pending patent applications related to work described in this article.
Author contributions
Conception and design: LLS, JJS, SFM, JTP, and VS. Design and synthesis of σ2R/Tmem97 ligands: JJS. Re-synthesis of JVW-1034: JJS, MDW, and TRH. Collection and analysis of data: LLS, JJS, AF, RCY, PNS, TS, BAP, KMF, and AS. Interpretation of data: LLS, JJS, AF, VS, JTP, and SFM. SFM, JTP, VS, AF, JJS, and LLS wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Drs. Martin and Sahn also report being co-founders of NuvoNuro, LLC. Drs. Sabino and Ferragud as well as Yen, Satarasinghe, Shi, Prakash, Friese, and Shen report no biomedical financial interests or potential competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Scott, L.L., Sahn, J.J., Ferragud, A. et al. Small molecule modulators of σ2R/Tmem97 reduce alcohol withdrawal-induced behaviors. Neuropsychopharmacol 43, 1867–1875 (2018). https://doi.org/10.1038/s41386-018-0067-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-018-0067-z
This article is cited by
-
σ2R/TMEM97 in retinal ganglion cell degeneration
Scientific Reports (2022)
-
Structures of the σ2 receptor enable docking for bioactive ligand discovery
Nature (2021)
-
Transcriptional analysis of the response of C. elegans to ethanol exposure
Scientific Reports (2021)
-
Sigma-1 receptor ligand PD144418 and sigma-2 receptor ligand YUN-252 attenuate the stimulant effects of methamphetamine in mice
Psychopharmacology (2019)