Abstract
We determined the risk of late morbidity and mortality after autologous blood or marrow transplantation (BMT) for lymphoma performed before age 40. The cohort included autologous BMT recipients who had survived ≥2 years after transplantation (N = 583 [HL = 59.9%; NHL = 40.1%]) and a comparison cohort (N = 1070). Participants self-reported sociodemographics and chronic health conditions. A severity score (grade 3 [severe], 4 [life threatening] or 5 [fatal]) was assigned to the conditions using CTCAE v5.0. Logistic regression estimated the odds of grade 3–4 conditions in survivors vs. comparison subjects. Proportional subdistribution hazards models identified predictors of grade 3–5 conditions among BMT recipients. Median age at BMT was 30.0 years (range: 2.0–40.0) and median follow-up was 9.8 years (2.0–32.1). Survivors were at a 3-fold higher adjusted odds for grade 3–4 conditions (95% CI = 2.3–4.1) vs. comparison subjects. Factors associated with grade 3–5 conditions among BMT recipients included age at BMT (>30 years: adjusted hazard ratio [aHR] = 2.31; 95% CI = 1.27–4.19; reference: ≤21 years), pre-BMT radiation (aHR = 1.52; 95% CI = 1.13–2.03; reference: non-irradiated), and year of BMT (≥2000: aHR = 0.54; 95% CI = 0.34–0.85; reference: <1990). The 25 years cumulative incidence of relapse-related and non-relapse-related mortality was 18.2% and 25.9%, respectively. The high risk for late morbidity and mortality after autologous BMT for lymphoma performed at age <40 calls for long-term anticipatory risk-based follow-up.
Similar content being viewed by others
Introduction
High-dose chemotherapy ± radiation with autologous blood or marrow transplantation (BMT) rescue is an integral part of consolidation and/or salvage therapy for children, adolescents and young adults with Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL) [1, 2]. While conventionally treated cancer patients are at risk for late morbidity and mortality [3,4,5,6,7], the high-intensity conditioning increases the risk further [8]. However, there is a paucity of information regarding the long-term burden of morbidity and cause-specific late mortality after autologous BMT for lymphoma performed in children, adolescents and young adults. The few studies that have examined this question are limited by small samples or relatively short follow-up [9,10,11]. We addressed these limitations by leveraging the Blood or Marrow Transplant Survivor Study (BMTSS) to determine the magnitude of risk of chronic health conditions as well as cause-specific late mortality in patients undergoing autologous BMT for lymphoma before age 40. We also sought to determine sub-populations at highest risk for morbidity to set the stage for future targeted interventions.
Methods
Study design and participants
BMTSS is a cohort study examining long-term outcomes in individuals who received BMT between 1974 and 2014 at the University of Minnesota, City of Hope, or University of Alabama at Birmingham and survived 2 or more years after BMT, regardless of disease status at cohort entry or vital status after cohort entry. A comparison cohort of siblings of all the BMT survivors participating in the BMTSS was also enrolled. However, only 16% of the BMT survivors were related to the siblings included in this analysis. Survivors and comparison subjects (or parents of participants <18 years) completed a 255-item BMTSS survey covering the following areas: diagnosis by a healthcare provider of chronic health conditions (including age at diagnosis), healthcare utilization and sociodemographic characteristics (race/ethnicity, sex, education, household income and insurance). The reliability and validity of the BMTSS survey was tested, and responses regarding chronic health conditions demonstrated a high level of agreement with the medical records [12]. Chronic health conditions were graded using the Common Terminology Criteria for Adverse Events (CTCAE), v5.0, distinguishing grades 1 through 5 based on the severity of each event (grade 1, mild; grade 2, moderate; grade 3, severe; grade 4, life-threatening/disabling; grade 5, death from chronic health conditions) [13]. For participants with more than one chronic health condition, the condition with a maximum grade was used. The same scoring system was applied to responses from the comparison cohort. A detailed description of the questions asked in the BMTSS survey, the corresponding chronic health condition categories created from the responses and the scoring of these conditions are presented in Supplementary Table 1. Information on primary diagnosis, age at BMT, stem cell source (bone marrow; peripheral blood stem cells [PBSCs]), risk of relapse at transplantation (high or standard risk), and exposure to total body irradiation (TBI) and chemotherapeutic agents used for conditioning, as well as pre-BMT therapeutic exposures was obtained on BMT recipients from the institutional transplant databases and/or medical records. Patients who underwent BMT in first or second complete remission were considered standard risk of relapse at transplantation; all other patients were considered at high risk. To be included in the current report, BMTSS participants with HL or NHL had to have received a single or tandem (two transplantations within 3 months) autologous BMT before age 40.
National Death Index (NDI) Plus [14] provided data regarding the date and cause of death through December 31, 2020. Additional information from medical records and Accurint databases [15] were used to extend the vital status information through May 31, 2022. For the deceased patients, we used the cause of death information from the NDI Plus; a CTCAE grade 5 was assigned to chronic health conditions stated as the cause of death in the NDI Plus files for those who died of a chronic health condition. Cause of death was classified as external (suicide, homicide or accident), relapse-related (RRM: primary cause of death matching the pre-transplant diagnosis) and non-relapse-related (NRM). The University of Alabama at Birmingham institutional review board served as the single institutional review board of record. Informed consent was obtained in accordance with the Declaration of Helsinki.
Statistical analyses
BMT survivors (alive at study participation) vs. comparison cohort
Standard parametric and nonparametric techniques compared the demographic and clinical characteristics and the prevalence of specific chronic health conditions between BMT survivors and the comparison cohort. Cumulative incidence of chronic health conditions as a function of attained age compared the burden of morbidity between BMT recipients and comparison subjects. Logistic regression was used for estimating the odds of grade 3–4 conditions in BMT survivors vs. the comparison cohort, adjusting for age at study participation, sex, race/ethnicity, education, household income and health insurance.
BMT cohort (alive and deceased)
For the BMT cohort, the cumulative incidence of chronic health conditions was calculated as a function of time from BMT, using death from other causes as competing risk. When analyzing predictors of grade 3–5 conditions in BMT recipients, proportional subdistribution hazards models (Fine-Gray) were used, treating death from primary disease or external causes as competing risk. The following variables were examined: sex, race/ethnicity, age at BMT, year of BMT, primary disease, source of stem cells, pre-BMT therapeutic exposures, TBI, chemotherapeutic agents used for conditioning, treating institution, education, household income and health insurance. BMT recipients with missing pre-BMT treatment data were treated as a separate group. For the multivariable model, we performed stepwise backward variable selection, retaining variables with p values < 0.1 in the final models. Mediation analysis was done by adding individual risk factors to the model examining the association between transplant era (<1990; 1990–1999; ≥2000) and risk of grade 3–5 conditions, censored at 7 years post-BMT, to examine if the hazard ratios changed. Censoring at 7 years post-BMT was chosen to ensure comparable follow-up across all eras. Overall survival was calculated using the Kaplan–Meier method, conditional on surviving 2 years post-BMT. The cumulative incidence of RRM used NRM and external causes as competing risk. The cumulative incidence of NRM used RRM and external causes as competing risk.
Data were analyzed using SAS version 9.4 (SAS Institute, Cary, NC). All statistical tests were two-sided, and p < 0.05 was considered statistically significant.
Results
Of the 862 BMT recipients eligible for study participation, 223 (25.9%) had died after surviving at least 2 years (Supplementary Fig. 1). Of the 639 alive eligible BMT survivors, 122 were lost to follow-up (19.1%). Of the 517 patients successfully contacted, 157 patients refused participation (30.4%), yielding 360 participants who completed the BMTSS questionnaire (participation rate: 69.6%). The cohort used in this report consisted of 583 individuals (360 alive and 223 deceased) who had received autologous BMT for HL or NHL before 2014 at age <40 years, and had survived ≥2 years. Females (46.7% vs. 31.8%, p = 0.001), non-Hispanic White individuals (72.7% vs. 62.4%, p = 0.007) and those transplanted before year 2000 (56.1% vs. 24.2%, p < 0.001), were more likely to participate (Supplementary Table 2).
Study cohort
The characteristics of the BMT recipients overall and by primary disease are shown in Table 1. In this cohort of 583 patients, 349 (59.9%) carried a primary diagnosis of HL and 234 (40.1%) of NHL. The median age at transplantation was 30.0 years (range: 2.0–40.0); median length of follow-up was 9.8 years (2.0–32.1); median age at study participation was 41.0 years (13.7–67.1); 311 were male (53.3%), 424 were non-Hispanic White (72.9%), and 255 had undergone BMT in the year 2000 or later (43.7%). PBSCs were used in 493 individuals (84.6%), and 367 were considered at high risk of relapse at BMT (63.0%). TBI was used for conditioning in 222 individuals (38.1%) (median dose: 12.0 Gy, range 5.0–13.2 Gy). Of the 438 (75.1%) individuals with available data on pre-BMT therapeutic exposures, 166 (37.9%) had received pre-BMT radiation with a median radiation dose of 32.5 Gy (range 10.0–60.0 Gy). The comparison cohort consisted of 1070 individuals. A comparison of the demographic characteristics between comparison subjects and BMT survivors alive at study participation is shown in Supplementary Table 3.
Severe/life-threatening chronic health conditions in autologous BMT survivors (alive at study participation) vs. comparison subjects
The prevalence of grade 3–4 chronic health conditions was significantly greater in survivors compared to comparison subjects (any condition: 39.3% vs. 26.8%, p < 0.0001; multiple conditions: 14.7% vs. 7.9%, p < 0.0001). By age 45, the cumulative incidence of a grade 3–4 chronic health conditions was 41.8% (95% CI; 35.8%–47.6%) among BMT survivors vs. 14.3% (95% CI; 12.1%–16.7%) among comparison subjects (p < 0.0001) (Fig. 1). The adjusted odds for grade 3–4 chronic health conditions was three-fold higher in BMT survivors compared to the comparison cohort (aOR = 3.0, 95% CI = 2.3–4.1) (Supplementary Table 4).
Severe/life-threatening/fatal chronic health conditions among autologous BMT recipients
Of the 583 BMT recipients in the cohort, 408 (70.0%) developed ≥1 grade 1–5 chronic health conditions (HL: N = 234 [67.1%]; NHL: n = 174 [74.4%]). Furthermore, 241 BMT recipients (41.3%) developed one or more grade 3–5 conditions (HL: N = 148 [42.4%]; NHL: N = 93 [39.7%]). The 20 years cumulative incidence of any grade 3–5 condition for the entire cohort was 47.1% (95% CI = 42.0–52.1%); it was 49.9% (95% CI = 43.2%–56.3%) for HL patients and 42.6% (95% CI = 34.7%–50.2%) for NHL patients (Fig. 2).
As shown in Table 2, the risk of a grade 3–5 condition in the entire cohort was higher among those transplanted at age >30 years (HR = 2.31; 95% CI = 1.27–4.19; reference: ≤21 years), and those receiving pre-BMT radiotherapy (HR = 1.52; 95% CI = 1.13–2.03; reference: non-irradiated). Patients treated in the most recent era (2000 or later) had a lower risk for a grade 3–5 condition compared to those transplanted before 1990 (HR = 0.54; 95% CI = 0.34–0.85). When restricting the follow-up to 7 years post-BMT, the lower risk among those transplanted in year 2000 or later remained significant (HR = 0.44; 95% CI = 0.26–0.74) (Supplementary Fig. 2). Among patients with HL, the risk of a grade 3–5 condition was higher among those transplanted at age >30 years (HR = 2.16; 95% CI = 1.12–4.18; reference: ≤21 years) (Table 2). Among patients with NHL, Black patients had a higher risk of a grade 3–5 condition (HR = 3.79; 95% CI = 1.88–7.67), as did those receiving pre-BMT radiotherapy (HR = 1.77; 95% CI = 1.13–2.76; reference: non-irradiated). Finally, patients with NHL transplanted ≥2000 had a HR of 0.34 (95% CI = 0.18–0.64) of a grade 3–5 condition compared to those transplanted before 1990. When restricting the follow-up to 7 years post-BMT, the lower risk among those transplanted in year 2000 or later remained significant among NHL patients (HR = 0.33; 95% CI = 0.14–0.75). In the mediation analysis, none of the measured risk factors explained the decrease in risk seen among those transplanted in the most recent era (Supplementary Table 5).
The most prevalent grade 3–5 chronic health conditions included subsequent malignant neoplasms (11.6%), and cardiovascular disease (10.5%) (Supplementary Table 6). Specific subsequent malignant neoplasms included leukemia/MDS (23.9%), skin cancer (17.9%), breast cancer (11.9%), sarcoma (7.5%), gastro-intestinal cancer (6.0%), lung cancer (6.0%), other cancer types (16.4%) and unknown (10.4%). The prevalence of grade 3–5 cardiovascular disease among Black BMT recipients was 18.4% and among non-Hispanic white 9.7%. The 10 years cumulative incidence of grade 3–5 cardiovascular disease was 6.1% (95% CI = 4.2–8.5%) and of grade 3–5 subsequent malignant neoplasms was 5.6% (95% CI = 3.8–7.8%) (Supplementary Table 7).
Late mortality in autologous BMT recipients
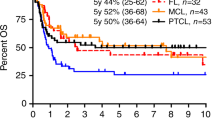
Conditional on surviving the first 2 years after BMT, the 25 years all-cause mortality was 62.6% (95% CI = 55.5–68.8%) for HL patients and 42.3% (95% CI = 33.7–50.6%) for NHL patients (Fig. 3). The adjusted hazards of all-cause mortality were higher in individuals with HL (HR = 1.75, 95% CI = 1.20–2.55; reference: NHL), among Black BMT recipients (HR = 2.36, 95% CI = 1.42–3.90; reference: non-Hispanic White), and in those with a lower income ($50,000–$74,999: HR = 3.07, 95% CI = 1.39–6.79; <$50,000: HR = 5.12, 95% CI = 2.55–10.28; reference: ≥$75,000) (Table 3).
Causes of death were available for 82% of the deceased BMT cohort, and included primary disease (35%), infection (22%), subsequent malignant neoplasm (16%), cardiovascular disease (13%), pulmonary disease (4%), homicide/suicide/accident (4%), and other causes (7%). As shown in Fig. 4, relapse-related mortality (RRM) plateaued after 20 years from BMT, whereas the non-relapse-related mortality (NRM) continued to climb, crossing RRM at 20 years from BMT. The 25 years cumulative incidence of RRM was 18.2% (95% CI 14.3–20.8%), while that of NRM was 25.9% (95% CI 21.3–30.6%).
Individuals with HL were at increased risk for RRM (HR = 2.11, 95% CI = 1.31–3.42; reference: NHL) (Table 4). Other factors associated with RRM included high risk of relapse at BMT (HR = 2.21, 95% CI = 1.17–4.18; reference: standard risk), and year of transplantation (≥2000: HR = 0.37, 95% CI = 0.20–0.67; reference: <1990). Factors associated with NRM included race/ethnicity (Black individuals: HR = 3.72, 95% CI = 1.68–8.24; Hispanic individuals: HR = 1.95, 95% CI = 1.05–3.60; reference: non-Hispanic White individuals), pre-BMT radiation (HR = 2.30, 95% CI = 1.37–3.86; reference: non-irradiated) and year of transplantation (≥2000: HR = 0.30, 95% CI = 0.13–0.69; reference: <1990). When restricting the follow-up to 7 years post-BMT, treatment era was no longer significantly associated with RRM nor NRM (results not shown). The specific causes of death are presented in Supplementary Table 8; death due to cardiovascular disease was overrepresented among Black patients (17.9%) vs. non-Hispanic White patients (10.2%) or Hispanic patients (9.1%).
Discussion
Conditional on surviving the first 2 years after autologous BMT for lymphoma performed in childhood, adolescence or young adulthood, the BMT survivors were at a three-fold higher odds of experiencing a severe or life-threatening chronic health condition compared to a comparison cohort. At 20 years from BMT, almost half of the BMT recipients had developed a severe, life threatening, or fatal chronic health condition. Individuals between 30 and 39 years of age at BMT, those transplanted before the year 2000, recipients of pre-BMT radiation and those with HL were at a higher risk of a severe, life-threatening, or fatal condition. In this cohort, the 25 years all-cause mortality exceeded 60% for individuals with HL patients and 40% for those with NHL. NRM continued to climb with time since BMT, crossing RRM at 20 years post-BMT.
Previous studies have reported a high burden of long-term morbidity borne by survivors of autologous BMT for lymphoma [16,17,18]. However, these studies focused either exclusively on adults or on cohorts that consisted largely of adults. The current study shows that the BMT recipients were at a three-fold higher risk of a grade 3–4 chronic health condition when compared with a comparison cohort. Within the BMT survivor cohort, important findings included a 20 years cumulative incidence of a grade 3–5 condition of 50% among individuals with HL, and 43% among those with NHL. Notably, the cumulative incidence continued to increase throughout follow-up. Risk factors for grade 3–5 conditions included transplantation at age >30 years, and pre-BMT radiotherapy. In adult recipients of autologous BMT, older age has been identified as a risk factor for late effects [17]. It is well-known that chest-directed radiotherapy increases the risk for chronic health conditions, especially subsequent malignant neoplasms and cardiovascular and pulmonary disease [19, 20], and that TBI increases the risk for a wide range of late effects [21, 22]. In the current study, pre-BMT radiotherapy was a significant predictor of a grade 3–5 chronic health condition, while TBI was not. This was likely due to the higher dose of radiation used in the pre-BMT radiation vs. TBI (median dose: 32.5 vs. 12.0 Gy). Patients transplanted in year 2000 or later had a lower risk for grade 3–5 conditions. Notably, none of the examined risk factors explained this decline in risk.
A CIBMTR report of 2-year survivors of autologous BMT for classical HL or diffuse large B-cell lymphoma (DLBCL) between 1990 and 2008, included a sub-analysis of 606 patients transplanted between 15 and 39 years of age, showing a 5 years overall survival of 91% for classical HL and 97% for DLBCL patients [9]. These 5 years survival rates, conditional on surviving 2 years, are higher than those of the present cohort (HL: 75% and NHL: 88%). The disparity may be influenced by differences in inclusion, with the current report including all NHL and HL sub-types, as well as patients transplanted before 1990.
In the CIBMTR report, the NRM rate 5 years post-BMT was 7% and 2% among patients with classical HL and DLBCL, respectively, which is comparable to that of the present study (5.4%) [9]. However, in our report, with extended follow-up, we show a continuous increase in NRM with time from BMT, with a cumulative incidence of 26% at 25 years post-BMT. In contrast to NRM, the cumulative incidence of RRM plateaued after 20 years post-BMT. The most common causes of death in our cohort were primary disease, infection, subsequent malignant neoplasms and cardiovascular disease. The findings of the current study warrant that HL and NHL patients treated with autologous BMT receive life-long follow-up using a risk-stratified approach as recommended by the Children’s Oncology Group guidelines [23].
In the current report, individuals with HL, Black individuals and those with a lower income had an increased risk of all-cause mortality. We believe that the higher risk of all-cause mortality in individuals with HL compared to those with NHL could have stemmed from the higher risk of relapse-related mortality as well as a higher prevalence of chronic health conditions due, in part, to the higher proportion of HL patients receiving pre-BMT radiation (46.8%) when compared with NHL patients (25.8%), at higher median dose (35.5 Gy vs. 30.0 Gy). For NRM, there was no observed difference in risk by type of lymphoma, while pre-BMT radiotherapy was found to be a significant predictor. The finding of low income as a predictor of all-cause mortality is in line with that of Hong et al., who reported an association between lower socioeconomic status and worse outcome after autologous BMT for lymphoma in adults [24]. In contrast to the study by Hong et al., race/ethnicity was also a predictor for all-cause mortality in the present study, with Black patients being at increased risk. A notably high share of deaths among Black patients was due to cardiovascular disease, which is in line with that of black individuals in the general population in the United States [25]. Inequality regarding access to follow-up care may contribute to the higher late mortality risk seen in vulnerable sub-populations [26]. In contrast to the CIBMTR report [9], TBI did not significantly increase the risk of RRM or NRM in the present study. The differences in inclusion, with the CIBMTR report not being restricted to the sub-group of adolescents and young adults, could influence the differences in the findings between the two studies. However, in the current study, pre-BMT radiation was associated with an increase in risk for NRM.
The results from our study need to be interpreted in the context of its limitations. These include the lack of detailed information on sub-types of HL and NHL. An additional potential limitation is the use of self-reported data, potentially influenced by study participants’ access to care, awareness among healthcare providers of the risk of late effects, and communication of these outcomes to their patients. The retrospective design of the study may further increase recall bias of self-reported chronic health conditions, including the age of diagnosis. However, examining severe/life-threatening/fatal chronic health conditions likely mitigated this issue, since we were focusing on clinically overt complications. Further, our survey demonstrated a high level of sensitivity and specificity against medical record reviews [12]. The mortality results in the current report are based on causes of death registered on death certificates, where some degree of misclassification is inherent. Nonetheless, use of NDI provides the most comprehensive evaluation of vital status. Finally, while we observed a decline in late morbidity among patients transplanted in the more recent years, we were unable to identify the underlying cause(s), likely due to some unmeasured changes in transplant practice.
These limitations notwithstanding, the current investigation describes the burden of chronic health conditions and late mortality experience of a cohort of patients undergoing autologous BMT for lymphoma in children, adolescents and young adults, the largest of its kind to date, followed for a median of almost 10 years. Novel findings include a three-fold increase in risk for serious or life-threatening conditions compared to a comparison cohort and a steadily increasing cumulative incidence of serious, life-threatening or fatal conditions with time from BMT. Relapse was the leading cause of death; however, while RRM plateaus with time, NRM becomes the major cause of death after 20 years from transplantation. Lastly, Black and Hispanic individuals and those with lower socioeconomic status were most vulnerable. Findings of the present study provide evidence for long-term anticipatory follow-up, with focus on vulnerable sub-populations.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Daw S, Hasenclever D, Mascarin M, Fernandez-Teijeiro A, Balwierz W, Beishuizen A, et al. Risk and response adapted treatment guidelines for managing first relapsed and refractory classical Hodgkin lymphoma in children and young people. Recommendations from the EuroNet Pediatric Hodgkin Lymphoma Group. Hemasphere. 2020;4:e329.
Harris RE, Termuhlen AM, Smith LM, Lynch J, Henry MM, Perkins SL, et al. Autologous peripheral blood stem cell transplantation in children with refractory or relapsed lymphoma: results of Children’s Oncology Group study A5962. Biol Blood Marrow Transplant. 2011;17:249–58.
Bluhm EC, Ronckers C, Hayashi RJ, Neglia JP, Mertens AC, Stovall M, et al. Cause-specific mortality and second cancer incidence after non-Hodgkin lymphoma: a report from the Childhood Cancer Survivor Study. Blood. 2008;111:4014–21.
Castellino SM, Geiger AM, Mertens AC, Leisenring WM, Tooze JA, Goodman P, et al. Morbidity and mortality in long-term survivors of Hodgkin lymphoma: a report from the Childhood Cancer Survivor Study. Blood. 2011;117:1806–16.
Schaapveld M, Aleman BM, van Eggermond AM, Janus CP, Krol AD, van der Maazen RW, et al. Second cancer risk up to 40 years after treatment for Hodgkin’s lymphoma. N Engl J Med. 2015;373:2499–511.
van Nimwegen FA, Schaapveld M, Janus CP, Krol AD, Petersen EJ, Raemaekers JM, et al. Cardiovascular disease after Hodgkin lymphoma treatment: 40-year disease risk. JAMA Intern Med. 2015;175:1007–17.
de Vries S, Schaapveld M, Janus CPM, Daniels LA, Petersen EJ, van der Maazen RWM, et al. Long-term cause-specific mortality in Hodgkin lymphoma patients. J Natl Cancer Inst. 2021;113:760–9.
Armenian SH, Sun CL, Kawashima T, Arora M, Leisenring W, Sklar CA, et al. Long-term health-related outcomes in survivors of childhood cancer treated with HSCT versus conventional therapy: a report from the Bone Marrow Transplant Survivor Study (BMTSS) and Childhood Cancer Survivor Study (CCSS). Blood. 2011;118:1413–20.
Myers RM, Hill BT, Shaw BE, Kim S, Millard HR, Battiwalla M, et al. Long-term outcomes among 2-year survivors of autologous hematopoietic cell transplantation for Hodgkin and diffuse large b-cell lymphoma. Cancer. 2018;124:816–25.
Majhail NS, Weisdorf DJ, Defor TE, Miller JS, McGlave PB, Slungaard A, et al. Long-term results of autologous stem cell transplantation for primary refractory or relapsed Hodgkin’s lymphoma. Biol Blood Marrow Transplant. 2006;12:1065–72.
Talleur AC, Flerlage JE, Shook DR, Chilsen AM, Hudson MM, Cheng C, et al. Autologous hematopoietic cell transplantation for the treatment of relapsed/refractory pediatric, adolescent, and young adult Hodgkin lymphoma: a single institutional experience. Bone Marrow Transplant. 2020;55:1357–66.
Louie AD, Robison LL, Bogue M, Hyde S, Forman SJ, Bhatia S. Validation of self-reported complications by bone marrow transplantation survivors. Bone Marrow Transplant. 2000;25:1191–6.
Common Terminology Criteria for Adverse Events (CTCAE) v5.0. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm#ctc_50 Accessed Jan 11 2023.
National Death Index. 2022. https://www.cdc.gov/nchs/ndi/index.htm Accessed Jan 11 2023.
Accurint databases. http://www.accurint.com Accessed Jan 11 2023.
Majhail NS, Ness KK, Burns LJ, Sun CL, Carter A, Francisco L, et al. Late effects in survivors of Hodgkin and non-Hodgkin lymphoma treated with autologous hematopoietic cell transplantation: a report from the bone marrow transplant survivor study. Biol Blood Marrow Transplant. 2007;13:1153–9.
Smeland K, Holte H, Fagerli UM, Bersvendsen H, Hjermstad MJ, Loge JH, et al. Total late effect burden in long-term lymphoma survivors after high-dose therapy with autologous stem-cell transplant and its effect on health-related quality of life. Haematologica. 2022;107:2698–707.
Georges GE, Bar M, Onstad L, Yi JC, Shadman M, Flowers ME, et al. Survivorship after autologous hematopoietic cell transplantation for lymphoma and multiple myeloma: late effects and quality of life. Biol Blood Marrow Transplant. 2020;26:407–12.
Holmqvist AS, Chen Y, Berano Teh J, Sun C, Birch JM, van den Bos C, et al. Risk of solid subsequent malignant neoplasms after childhood Hodgkin lymphoma-Identification of high-risk populations to guide surveillance: a report from the Late Effects Study Group. Cancer. 2019;125:1373–83.
Bates JE, Howell RM, Liu Q, Yasui Y, Mulrooney DA, Dhakal S, et al. Therapy-related cardiac risk in childhood cancer survivors: an analysis of the childhood cancer survivor Study. J Clin Oncol. 2019;37:1090–101.
Friedman DN, Hilden P, Moskowitz CS, Suzuki M, Boulad F, Kernan NA, et al. Cardiovascular risk factors in survivors of childhood hematopoietic cell transplantation treated with total body irradiation: a longitudinal analysis. Biol Blood Marrow Transplant. 2017;23:475–82.
Friedman DL, Rovo A, Leisenring W, Locasciulli A, Flowers ME, Tichelli A, et al. Increased risk of breast cancer among survivors of allogeneic hematopoietic cell transplantation: a report from the FHCRC and the EBMT-Late Effect Working Party. Blood. 2008;111:939–44.
Children’s Oncology Group guidelines. http://survivorshipguidelines.org Accessed Jan 11 2023.
Hong S, Rybicki L, Abounader DM, Bolwell BJ, Dean R, Gerds AT, et al. Association of socioeconomic status with autologous hematopoietic cell transplantation outcomes for lymphoma. Bone Marrow Transplant. 2016;51:1191–6.
National Center for Health Statistics. Racial and ethnic disparities in heart disease. 2019. www.cdc.gov/nchs/hus/spotlight/HeartDiseaseSpotlight_2019_0404.pdf.
Rokitka DA, Curtin C, Heffler JE, Zevon MA, Attwood K, Mahoney MC. Patterns of loss to follow-up care among childhood cancer survivors. J Adolesc Young Adult Oncol. 2017;6:67–73.
Acknowledgements
This study was supported in parts by grants from the National Cancer Institute U01CA213140 (SB), R01CA078938 (SB), the Leukemia Lymphoma Society (SB) and the Swedish Childhood Cancer Fund (ASH).
Author information
Authors and Affiliations
Contributions
SB, DW, SHA and ASH contributed to study conception and design. SB, LH, JW, ESR, NB, AB, HST, LFF and SHA collected and assembled the data. SB, QM, WL, LH, ASH and FLW analyzed and interpreted the data. ASH and SB drafted the manuscript. All authors critically reviewed the manuscript for important intellectual content and approved the final manuscript. SB, LH, WL, ESR, LFF and NB provided administrative, technical, or material support. SB, DW and SHA supervised the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Holmqvist, A.S., Meng, Q., Dai, C. et al. Late morbidity and mortality after autologous blood or marrow transplantation for lymphoma in children, adolescents and young adults—a BMTSS report. Leukemia 38, 601–609 (2024). https://doi.org/10.1038/s41375-024-02144-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-024-02144-7