Abstract
Objective
To test the hypothesis that brainstem hypoxic–ischemic injury on magnetic resonance imaging (MRI) would be independently associated with short-term outcomes in cooled asphyxiated infants.
Methods
A total of 90 consecutively cooled asphyxiated infants who survived to have brain MRI were reviewed. A neuroradiologist who was masked to outcomes evaluated MRI images for brainstem involvement. Outcomes were mortality and length of stay.
Results
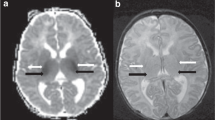
Brainstem lesions were present on post-cooling brain MRI in 20 of the 90 infants (22%). Overall, four infants died before discharge, and all four had brainstem involvement. The infants with brainstem involvement had longer hospital stay (29 days, IQR 20–47 versus 16 days, IQR 10–26; P = 0.0001), compared to infants without brainstem lesions (n = 70); and upon multivariate analysis, brainstem involvement remained independently associated with prolonged hospital stay (β = 12.4, P = 0.001).
Conclusion
This study demonstrates the importance of recognizing brainstem injury for the prediction of short-term outcomes in cooled asphyxiated infants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Leech RW, Alvord EC Jr. Anoxic ischemic encephalopathy in the human neonatal period: the significance of brain stem involvement. Arch Neurol. 1977;34:109–13.
Roland EH, Hill A, Norman MG, Flodmark O, MacNab AJ. Selective brainstem injury in an asphyxiated newborn. Ann Neurol. 1988;23:89–92.
Quattrocchi CC, Fariello G, Longo D. Brainstem tegmental lesions in neonates with hypoxic- ischemic encephalopathy: magnetic resonance diagnosis and clinical outcome. World J Radiol. 2016;8:117–23.
Logitharajah P, Rutherford MA, Cowan FM. Hypoxic-ischemic encephalopathy in preterm infants: antecedent factors, brain imaging, and outcome. Pediatr Res. 2009;66:222–9.
Martinez-Biarge M, Diez-Sebastian J, Kapellou O, Gindner D, Allsop JM, Rutherford MA, et al. Predicting motor outcome and death in term hypoxicischemic encephalopathy. Neurology. 2011;76:2055–61.
Martinez-Biarge M, Diez-Sebastian J, Wusthoff CJ, Lawrence S, Aloysius A, Rutherford MA, et al. Feeding and communication impairments in infants with central grey matter lesions following perinatal hypoxic-ischaemic injury. Eur J Paediatr Neurol. 2012;16:688–96.
Barkovich AJ, Westmark K, Partridge C, Sola A, Ferriero DM. Perinatal asphyxia: MR findings in the first 10 days. AJNR Am J Neuroradiol. 1995;16:427–38.
Myers RE. Two patterns of perinatal brain damage and their conditions of occurrence. Am J Obstet Gynecol. 1972;112:246–76.
Shankaran S, Laptook AR, Ehrenkranz RA, Tyson JE, McDonald SA, Donovan EF, et al. National Institute of Child Health and Human Development Neonatal Research Network. Whole-body hypothermia for neonates with hypoxic-ischemic encephalopathy. N Engl J Med. 2005;353:1574–84.
Ambalavanan N, Carlo WA, Shankaran S, Bann CM, Emrich SL, Higgins RD, et al. National Institute of Child Health and Human Development Neonatal Research Network. Predicting outcomes of neonates diagnosed with hypoxemic–ischemic encephalopathy. Pediatrics. 2006;118:2084–93.
Barkovich AJ, Hajnal BL, Vigneron D, Sola A, Patridge JC, Allen F, et al. Prediction of neuromotor outcome in perinatal asphyxia: evaluation of MR scoring systems. Am J Neuroradiol. 1998;19:143–9.
Sarkar S, Donn SM, Bapuraj JR, Bhagat I, Barks JD. Distribution and severity of hypoxic–ischemic lesions on brain MRI following therapeutic cooling: selective head versus whole body cooling. Arch Dis Child Fetal Neonatol Ed. 2012;97:F335–9.
Sarkar S, Bhagat I, Bapuraj JR, Dechert RE, Donn SM. Does clinical status 1 week after therapeutic hypothermia predict brain MRI abnormalities? J Perinatol. 2013;33:538–42.
Gupta S, Bapuraj JR, Carlson G, Trumpower E, Dechert RE, Sarkar S. Predicting the need for home gavage or g-tube feeds in asphyxiated neonates treated with therapeutic hypothermia. J Perinatol. 2018;38:728–33.
Rutherford MA. The asphyxiated term infant. In: Rutherford M, editor. MRI of the neonatal brain. London: Saunders; 2002. p. 99–128.
Cowan F, Rutherford M, Groenendaal F, Eken P, Mercuri E, Bydder GM, et al. Origin and timing of brain lesions in term infants with neonatal encephalopathy. Lancet. 2003;361:736–42.
Miller SP, Ramaswamy V, Michelson D, Barkovich AJ, Holshouser B, Wycliffe N, et al. Patterns of brain injury in term neonatal encephalopathy. J Pediatr. 2005;146:453–60.
Shankaran S, McDonald SA, Laptook AR, Hintz SR, Barnes PD, Das A, et al. Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Neonatal magnetic resonance imaging pattern of brain injury as a biomarker of childhood outcomes following a trial of hypothermia for neonatal hypoxic-ischemic encephalopathy. J Pediatr. 2015;167:987–93.
Arca-Dı azG, Re TJ, Drottar M, Fortuno CR, De Macedo-Rodrigues K, Im K, et al. Can cerebellar and brainstem apparent diffusion coefficient (ADC) values predict neuromotor outcome in term neonates with hypoxic-ischemic encephalopathy (HIE) treatedwith hypothermia? PLoS ONE. 2017;12:e0178510.
Quattrocchi CC, Longo D, Delfino LN, Cilio MR, Piersigilli F, Capua MD, et al. Dorsal brain stem syndrome: MR imaging location of brain stem tegmental lesions in neonates with oral motor dysfunction. AJNR Am J Neuroradiol. 2010;31:1438–42.
Rutherford M, Ramenghi LA, Edwards AD, Brocklehurst P, Halliday H, Levene M, et al. Assessment of brain tissue injury after moderate hypothermia in neonates with hypoxic–ischaemic encephalopathy: a nested subset of a randomised control trial. Lancet Neurol. 2010;9:39–45.
Cheong JL, Coleman L, Hunt RW, Lee KJ, Doyle LW, Inder TE, et al. Prognostic utility of magnetic resonance imaging in neonatal hypoxic‒ischemic encephalopathy: substudy of a randomized trial. Arch Pediatr Adolesc Med. 2012;166:634–40.
Shankaran S, Barnes PD, Hintz SR, Laptook AR, Zaterka-Baxter KM, McDonald SA, et al. Brain injury following trial of hypothermia for neonatal hypoxic‒ischaemic encephalopathy. Arch Dis Child Fetal Neonatal Ed. 2012;97:F398–404.
Gulati IK, Shubert TR, Sitaram S, Wei L, Jadcherla SR. Effects of birth asphyxia on the modulation of pharyngeal provocation-induced adaptive reflexes. Am J Physiol Gastrointest Liver Physiol. 2015;309:G662–9.
Massaro AN, Murthy K, Zaniletti I, Cook N, DiGeronimo R, Dizon M, et al. Short-term outcomes after perinatal hypoxic ischemic encephalopathy: a report from the Children’s Hospitals Neonatal Consortium HIE focus group. J Perinatol. 2015;35:290–6.
Takashima S, Becker LE. Relationship between abnormal respiratory control and perinatal brainstem and cerebellar infarctions. Pediatr Neurol. 1989;5:211–4.
Sarnat HB. Watershed infarcts in the fetal and neonatal brainstem. An aetiology of central hypoventilation, dysphagia, Mo¨bius syndrome and micrognathia. Eur J Pediatr Neurol. 2004;8:71–87.
Saito K. Reflections on the brainstem dysfunction in neurologically disabled children. Brain Dev. 2009;31:529–36.
Author information
Authors and Affiliations
Contributions
Subrata Sarkar, MD, developed the study concept, design, and analysis. He also critically revised and approved the final paper. Siddhartha Sarkar collected the data and provided critical input into the analysis of the data. He wrote the first draft of the paper and received input from the authors below as part of the paper revision. Other co-investigators (Suneeti Gupta, MD; Jayapalli Bapuraj, MD; and Ronald E. Dechert, RRT) provided critical input into the design and analysis of the study. They also critically revised and approved the final paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sarkar, S.S., Gupta, S., Bapuraj, J.R. et al. Brainstem hypoxic–ischemic lesions on MRI in infants treated with therapeutic cooling: effects on the length of stay and mortality. J Perinatol 41, 512–518 (2021). https://doi.org/10.1038/s41372-020-00873-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-020-00873-y