Abstract
Background
Biomass fuels are still in use for cooking by many households in resource poor countries such as Nepal and is a major source of household air pollution (HAP). Chronic exposure to HAP has been shown to be associated with shorter telomere length in adults.
Objectives
To measure the association between exposure related to household biomass fuel in infancy and leukocyte telomere length (LTL) at 18–23 months of age among 497 children from Bhaktapur, Nepal.
Methods
In a prospective cohort study design, we have collected information on household cooking fuel use and several clinical, anthropometric, demographic, and socioeconomic variables. We estimated the association between biomass fuel use and the relative LTL in multiple linear regression models.
Results
Most of the families (78%) reported liquified petroleum gas (LPG) as the primary cooking fuel, and 18.7% used biomass. The mean relative (SD) LTL was 1.03 (0.19). Children living in households using biomass fuel had on average 0.09 (95% CI: 0.05 to 0.13) units shorter LTL than children in households with no biomass fuel use. The observed association was unaltered after adjusting for relevant confounders. The association between LTL and biomass use was strongest among children from households with ≤2 rooms and without separate kitchen.
Significance
Exposure to biomass fuel use in early life might have consequences for longevity, and risk of chronic illnesses reflected in shortening of the telomeres. Our findings support the ongoing effort to reduce exposure to biomass fuel in low-resource settings.
Impact statements
Biomass for cooking is a leading source of household air pollution in low and middle-income countries, contributing to many chronic diseases and premature deaths. Chronic exposure to biomass fuel through oxidative stress and inflammation has been associated with a shortening of the telomeres, a “biological marker” of longevity. This prospective cohort study describes the association between household biomass fuel use and leukocyte telomere length among 497 toddlers. Leukocyte telomere length was significantly shorter among children living in households with biomass fuel than in children from homes where mainly LPG was used for cooking.
Clinical Trial registration
Clinicaltrials.gov: NCT02272842, registered October 21, 2014, Universal Trial Number: U1111-1161-5187 (September 8, 2014)
Similar content being viewed by others
Introduction
Biomass cooking fuel, mainly used in the form of firewood, coal, saw dust, animal dung or crop residues, is the main source of household air pollution (HAP) in many low- and middle-income countries (LMIC) [1]. The burning of biomass results in toxic pollutants such as particulate matter (PM), methane, black carbon, nitrous oxide, and carbon monoxide [2]. Globally more than 40% of households in LMICs are still dependent on biomass for cooking primarily due to poverty and the lack of alternative cleaner fuels [3].
Chronic exposure to HAP has been linked to several morbidities such as respiratory diseases (pneumonia, COPD, lung cancer, pulmonary tuberculosis), cardiovascular diseases (ischemic heart disease, stroke, hypertension), as well as cataracts [4, 5]. HAP was estimated to be responsible for 91.5 million DALY leading to 2.3 million deaths in 2019, which is 4% of all deaths [3]. Women and children are particularly vulnerable to HAP as they spend more time cooking indoors being directly exposed [6]. HAP during pregnancy has been associated with adverse pregnancy outcomes such as miscarriage, eclampsia, prematurity, and low birth weight [4], whereas exposure during early childhood has been associated with pneumonia [7], poor growth and impaired cognitive development [8, 9].
In Nepal, HAP, particularly from biomass fuel use, was listed as the second-largest contributor to the total disease burden [10]. This burden, however, varied substantially according to geographical locations and socio-economic status [11]. In 2015, the Government of Nepal initiated campaigns for cleaner energy for cooking and smokeless stoves to reduce the use of household biomass fuel from 75% to 30% within 2030 (SDG goal 7, target 7.1.2) [12].
Although the exact mechanisms on how HAP leads to chronic disease and premature death are unclear, recent evidence suggests that a possible pathway could be through shortening of the leukocyte telomere length (LTL) [13]. Telomeres are crucial to maintain chromosome stability and consist of repetitive DNA and protein complexes [14]. Telomere length serves as a “biological clock” and the shorter length is considered a propensity for an early onset of non-communicable diseases such as diabetes [15], obesity [16], COPD [17], and cancer [18]. LTL is naturally shortened with each cellular replication but may also be prematurely shortened due to various diseases [19] and environmental factors including smoking [20]. The process of telomere shortening may accelerate due to oxidative stress and inflammation [21], and both conditions are common during chronic exposure to HAP [22].
Two available systematic reviews and meta-analyses based on observational studies among adults confirmed a negative association between air pollution and telomere length [23, 24]. For HAP, a study among rural Chinese women showed that those using biomass fuel for cooking with a high exposure to PM2.5 and black carbon had shorter LTL than women with lower exposure [25]. Although LTL at birth has been shown to be associated with exposure to toxic pollutants during pregnancy [26], it remains unclear if the observed associations between biomass fuel use and telomere length can also be seen at young ages. In our study, we estimate the association between biomass use as a primary kitchen fuel assessed during infancy and LTL measured at 18–23 months of age in children from Bhaktapur, Nepal.
Methods
Study design and population
The study was conducted in Bhaktapur municipality which is predominantly a residence of the Newar ethnic group. The area also has an immigrant population from other parts of the country, many of whom work at carpet factories. Bhaktapur is one of four municipalities in the Bhaktapur district and has a population of approximately 79,000 with an annual growth rate of 3.3%. The municipality typically has clusters of old brick houses and is one of the most densely populated cities in Nepal (11,430 inhabitants/km2) [27]. Agriculture, small family businesses, services at the government or private organizations, and daily wage jobs are the main sources of income [28].
There are many brick kilns located at the outskirts of Bhaktapur municipality, which are functioning mainly during the winter and spring season (from October to May) and contribute to the poor ambient air quality due to the use of coal and firewood for burning bricks [29]. Additionally, relatively high CO2 emissions from vehicles are also common in Bhaktapur with an estimated emission load of 3310 tones/year [30]. Communal firing spots (ash kilns) are still in use in close proximity to residential houses in some areas of Bhaktapur where ceramic raw pots are stacked in layers of hay and then covered by ash for approximately 4 days to maintain the required temperature. All these factors may contribute to the high ambient air pollution in Bhaktapur, reflected in recordings of a mean 24 h PM2.5 concentration exceeding 15 μg/m3, which is the cut-off in the air quality index by the World Health Organization (WHO) [31].
Enrollment procedure and definitions
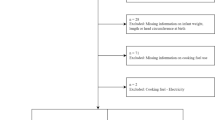
For the current analyses, we used data of children enrolled in a community-based, randomized, double-blind, placebo-controlled trial assessing the effect of daily vitamin B12 supplementation over a period of 12 months on neurodevelopment and growth [32]. Between April 2015 and February 2018, 600 children aged 6–11 months, mildly stunted (length-for-age Z-score of <−1) and available written consent from one of the parents were enrolled into the study and weekly followed up for one year. We targeted infants with length-for-age Z-score of <−1 SD because they constitute a high risk population [33] with regard to poor cognitive development, growth failure, and malnutrition. Screening of children for the eligibility to enroll in the study were done at different vaccination clinics and through a household survey. Infants who were eligible, were requested to visit at the study clinic at 6 months of age for re-assessment. Children were not enrolled if they had one of the following conditions: 1. Severe systemic illnesses requiring hospitalization, 2. Ongoing or planned multivitamin supplement intake that included vitamin B12, 3. Severe anemia (Hb < 7 g/dl), 4. Ongoing acute infection such as fever, cough or diarrhea that requires medical treatment, and 5. Severe malnutrition (weight-for-length Z-score < −3). The main results of the project have been reported elsewhere. In short, the intervention resulted in a substantially improved vitamin B12 status but did not improve growth or neurodevelopment neither at 18–23 months [34] nor 30–47 months of age [35].
Regular anthropometric measurements of children were carried out by trained field workers using digital weighing scales and infantometers (Seca, Germany). All measurements were taken twice, and the mean of these measurements were used in the analyses. For parental measurements, we used a stadiometer for height (Prestige, Hardik Medi Tech, India) and a portable electronic scale for weight (Salter, UK).
Baseline information covering socio-demographic, education and family characteristics were obtained from the caretakers at enrollment. We used a modified WAMI index to indicate socio-economic status, which stand for Water and sanitation, availability of Assets at the household and Maternal education as described by others [36]. The original WAMI index also includes household income, since we did not have information on income, we only included the remaining 3 indicators. The WAMI index ranges from 0–1 where a higher score indicates a better socio-economic condition.
We also collected information to describe the quality of indoor air in the households during enrollment. This included availability of a separate kitchen, smoking habits inside the house, and primary kitchen fuel use. Fuel use was defined as biomass if the family reported a current primary use of one of the following fuels for cooking: firewood, saw dust, cow dung, straw, or any crop residues.
Blood collection
One year after enrollment, when the children were 18–23 months of age, we collected ~3 ml blood into vials containing EDTA as anticoagulant (Eppendorf, Germany). The hemoglobin concentration was measured using HemoCue immediately after blood collection (HemoCue, Vedbæk, Denmark). Anemia at 6–11 months (enrollment) and 18–23 months (one year follow up period) of age was defined when hemoglobin concentration was <11 g/dl as per the WHO guidelines. The sample was centrifuged at 2000 to 2500 at room temperature for 10 minutes and after separation from the plasma stored at −70 degree Celsius until analyses during November 2020.
Leukocyte telomere length assay
The LTL analysis was done in the Research Laboratory at Harvard School of Public Health, Boston, USA (De Vivo lab). Five nanograms of genomic DNA were dried down in each well of a 384-well plate and resuspended in 10 µL of either the telomere or 36B4 PCR reaction mixture and stored in 4 °C up to 6 hours. The telomere reaction mixture consisted of 1x Thermo Fisher PowerUP SYBR Master Mix, 2.0 mM of DTT, 270 nM of Tel-1b primer, and 900 nM of Tel-2b primer. The reaction proceeded for 1 cycle hold at 50 °C for 2 minutes and at 95 °C for 2 minutes, followed by 35 cycles at 95 °C for 15 seconds, and 54 °C for 2 minutes. The 36B4 reaction consisted of 1x Thermo Fisher PowerUP SYBR Master Mix, 300 nM of 36B4U primer, and 500 nM of 36B4D primer. The 36B4 reaction proceeded for 1 cycle hold at 50 °C for 2 minutes and at 95 °C for 2 minutes, followed by 40 cycles at 95 °C for 15 seconds, and 58 °C for 1 minute and 10 seconds. All samples for both the telomere and single-copy gene (36B4) reactions were performed in triplicate on different plates. Each 384-well plate also contained a 6-point standard curve from 0.625 ng to 20 ng using pooled buffy-coat derived genomic DNA.
The standard curve assessed and compensated for inter-plate variations in PCR efficiency. The slopes of the standard curve for both the telomere and 36B4 reactions were −3.33 ± 0.33 and the linear correlation coefficient (R2) values for both reactions were over 0.99. The T/S ratio (-dCt) for each sample was calculated by subtracting the average 36B4 Ct value from the average telomere Ct value. The relative T/S ratio (-ddCt) was determined by subtracting the T/S ratio value of the 5 ng standard curve point from the T/S ratio of each unknown sample [37]. Quality control samples were interspersed throughout the test samples in order to assess inter-plate and intra-plate variability of threshold cycle (Ct) values. A combined inter- and intra-assay coefficient of variation (CV) calculated from the relative T/S ratio (-ddCt) of quality control samples is 8.1%. The LTL are expressed in relative units with a mean of approximately 1.
Statistical analysis
All collected data were checked manually by the study supervisors and double-entered in either the Microsoft Access database or a cloud based mobile data collection platform Iformbuilder (https://www.zerionsoftware.com/iformbuilder). Data are presented as numbers and percentages (%) for categorical variables or means and standard deviations (SD) for continuous variables. We produced density plots of LTL separately for households reporting biomass fuel use for cooking and for those who did not, using the Epanechnikov Kernel function. Multiple linear regression analyses adjusting for potential confounding variables were done to estimate the association between LTL and biomass fuel use. Household and child characteristics (gender, stunting, exclusive breastfeeding, low birth weight, age of child and mother, presence of indoor smoking, anemia and WAMI index) were selected as potential confounders and included in the regression model based on clinical and epidemiological relevance as well as the availability of data. We also re-ran the regression analyses using the same covariates as in the main regression model in the following pre-defined subgroups: birthweight (<2500 g vs. ≥2500 g), exclusive breastfeeding (<3 months vs. ≥3 months), stunting (HAZ < −2 SD vs. ≥−2 SD), underweight (WAZ < −2 SD vs. ≥−2 SD), number of rooms in the house (≤2 rooms vs. >2 rooms), separate kitchen and bedroom and practice of indoor smoking (yes vs. no). We assessed whether these variables significantly modified the association between biomass and LTL by including interaction terms in the regression models. Regression coefficients and 95% CIs from these subgroup analyses were displayed in a forest plot. In the same manner, we present conditional regression coefficients from quantile regression analyses for the following percentiles of LTL: 95, 90, 80, 70, 60, 50, 40, 30, 20, 10, and 5. We used these quantile regression models to assess if there are any differences in the associations of biomass cooking fuel use and LTL in the different percentiles of the LTL distribution. All data analyses were done using Stata 16 (StataCorp LLC).
Ethics
We obtained ethical clearance from the Nepal Health Research Council (NHRC #233/2014), and from the Regional Committee for Medical and Health Research Ethics (REC # 2014/1528), Norway. After informing the caretaker (usually the mother) about the details of the study, we received written consent. During the study period, children with acute illnesses were treated according to the CB-IMNCI guidelines.
Results
LTL was measured in the last 497 (87%) of the 574 children who completed the one-year follow-up of the main trial. Almost half of the children were female, 6–7 months of age at enrollment, or living in joint families (Table 1). There were no substantial differences in baseline characteristics between children for whom we did LTL analysis (n = 497) and those we did not (n = 77). Upon enrolment, all but 14 mothers were still breastfeeding, and 52% had initiated breastfeeding within 1 hour of delivery. One in every 4 children had been introduced to solid or semi-solid foods by 2 months of age. Although the prevalence of anemia was high when the children were 6–11 months (64%), it decreased to 26% when children reached 18–23 months of age. Among the anemic children, both at baseline and at the one year follow up, 60% were mildly anemic (hemoglobin concentration 10–10.9 g/dL) and 40% had moderate anemia (hemoglobin concentration 7–9.9 g/dL).
Most of the families (78%) reported use of LPG as the primary fuel for cooking, and 18.7% families used biomass fuel. One in every second families reported practice of indoor smoking, probably due to high numbers of fathers who smoke regularly (35%). The LTL was normally distributed, and the relative mean (SD) was 1.03 (0.19) ranging from 0.32 to 1.59. The distribution of LTL of the children is graphically presented in Fig. 1 separately for those from biomass-use households and other households. In the unadjusted regression analysis, children from biomass fuel use households had 0.08 units shorter LTL (95% CI: 0.04 to 0.12) than children from households that did not use biomass fuel (Table 2). This association was unaltered after including potential confounders in the regression model.
We also depicted the associations between biomass fuel use and LTL in several pre-defined subgroups (Fig. 2). Nutritional status indicators of the children such as exclusive breastfeeding for 3 months, underweight, stunting and birth weight did not modify the association. The corresponding p-values for the interaction were 0.5, 0.41, 0.23, 0.9, respectively. However, the association between biomass use and LTL was stronger in children from households with ≤2 rooms in the house (p-value for interaction 0.07), or who did not have separate kitchen (p-value for interaction 0.16). In the quantile regression analyses, the association between biomass fuel use and LTL showed a tendency of an increase in percentile difference in LTL towards the lower end of the LTL distribution. In other words, there was a stronger association between biomass fuel use and LTL in those with a shorter LTL compared to those with a longer LTL (Fig. 3).
Discussion
In the present prospective cohort of Nepalese toddlers, approximately one in five households used biomass as primary fuel for cooking. We found that the LTL was shorter among children from these households compared to in children living in households using primarily LPG as cooking fuel. The negative impact of HAP mainly driven by biomass fuel use, is well established for several adverse health outcomes in both children and adults [3]. This is, to the best of our knowledge, the first report describing an association between biomass fuel use and LTL in young children.
The available literature on the relationship between telomere length and exposure to air pollutants is primarily from studies measuring the ambient air quality at a population level [24, 38]. In our study, we have individual data on the exposure to biomass fuel from each household and LTL measured when the children were 18–23 months. We also have comprehensive data on other exposures such as growth, household, and socioeconomic characteristics. The longitudinal design, the ability to measure several potential confounding variables, and the relatively large sample size, provide strengths to our findings of an association between the exposure of biomass fuel and LTL in young children [3, 39, 40].
An interesting observation in our analyses is that there were few other factors associated with LTL. In the adjusted model, we found that girls had on average longer LTL than boys, and that those with anemia at enrolment had longer LTL one year later than those who did not have anemia. Age was not associated with LTL, which is probably due to the narrow age range (18 to 23 months) when the blood sample was taken. Our finding of longer telomere lengths in female than male children is expected and was also found in a cohort study in 4000 children in New Zealand where the telomere length was measured in 4-year-olds [41]. Similar gender wise variation in LTL and an association with lower life expectancy at the age of 40 years was described among UK Biobank participants [42].
In the subgroup analyses, the negative association between biomass fuel use and LTL was even stronger in children living in households with fewer than 3 rooms as well as in those who did not have separate kitchen and bedroom. Both not having a separate kitchen and having few rooms in the household is likely to contribute to increased HAP exposure which could partly explain the modified association [43]. The quantile regression analysis (Fig. 3) indicates a stronger association between LTL and HAP in the lower than higher LTL distributions which is an indication of exposure to HAP mostly affected children with lower LTL percentiles.
The use of biomass fuels for cooking is still very common in LMIC including Nepal, particularly in rural areas [3]. Our study site, although located in an urban and peri-urban area, is an agricultural community, and biomass fuels, mainly crop residues, were still used by 18.7% of all households. However, the use of biomass fuel seems to be decreasing in Bhaktapur; while 25% of households reported to use biomass fuel for cooking in a survey in 2006–2007 [39], there were only 5% of the households reporting the same in a survey in 2018–2020 [40]. At the beginning of the study, our site was severely affected by a large earthquake. During the re-construction period, many people lived in temporary shelters with limited access to basic needs. Moreover, blockades at the Indian border for more than 6 months led to restricted LPG availability. Both the consequences from the earthquake and blockades could be causes for an increased biomass fuel use at the study site.
Although we collected data after the child was born, it is likely that these children were exposed to toxic pollutants from biomass fuel use already during the intra-uterine period. Both exposure on HAP questionnaire and LTL measurements were done in one time point, our study can not distinguish timing of exposure whether it was during pregnancy or after the birth. Indeed, exposure to such pollutants during pregnancy has also shown to be associated with shorter telomere lengths at birth [26] and with subsequent higher systolic blood pressure at four years of age [44]. Beside genetic and environmental determinants, variations in telomere length could also be due to exposure to oxidative stress and chronic inflammation in utero as a counter regulatory effect of the telomerase which is a reverse transcriptase enzyme maintaining telomere length [45].
This is a secondary analysis from a nutritional randomized trial enrolling mildly stunted infants [34] and caution is required for the generalizability of our findings. Micronutrient status may also affect LTL [46]; however, we did not find any association between vitamin B12 supplementation and the LTL. We used biomass fuel use for cooking as a proxy for HAP and lack information on the specific exposure of pollutants such as PM2.5, black carbon, or nitrous oxide which may be regarded as a limitation to our study. We also lack information on secondary cooking fuels in the households, as well as fuel used for heating. We know however, that in this area such energy sources are primarily used for cooking purpose [47] limiting the exposure to secondary fuel use for heating. Still the lack of information on the secondary cooking fuels may lead to exposure misclassification which we believe may bias the results towards the null [48]. Moreover, despite adjusting for several relevant confounding variables, there is a possibility that the observed association is due to residual confounding. Since we asked about primary fuel use at one time point (when children were 6–11 months of age), there could have been changes in stove/fuel use during the course of the study.
In this community-based cohort, children who were living in households with biomass fuel use had substantially shorter LTL compared with children from households where mostly LPG were used as the primary kitchen fuel. The immediate and long-term possible consequences of these shorter LTL should be explored in future studies.
Data availability
Data available on request. To meet ethical requirements for the use of confidential patient data, requests must be approved by the Nepal Health Research Council (NHRC) and the Regional Committee for Medical and Health Research Ethics in Norway. Requests for data should be sent to the authors, by contacting, the Department of Global Health and Primary Care at the University of Bergen (post@igs.uib.no) or Child Health Research Project, Department of Child Health, IOM, Nepal (chrp2015@gmail.com).
References
WHO. Enivironment and Health. Household air pollution data 2022. https://www.who.int/data/gho/data/themes/air-pollution/household-air-pollution.
Mock CN, Smith KR, Kobusingye O, Nugent R, Abdalla S, Ahuja RB, et al. Injury Prevention and Environmental Health: Key Messages from Disease Control Priorities, Third Edition. In: Rd, Mock CN, Nugent R, Kobusingye O, Smith KR, editors. Injury Prevention and Environmental Health. The World Bank; Washington (DC); 2017.
HEI. State of Global Air 2020. Special Report. Boston, MA: Health Effects Institute (HEI); 2020.
Lee KK, Bing R, Kiang J, Bashir S, Spath N, Stelzle D, et al. Adverse health effects associated with household air pollution: a systematic review, meta-analysis, and burden estimation study. Lancet Glob Health. 2020;8:e1427–e34.
Pokhrel AK, Bates MN, Shrestha SP, Bailey IL, Dimartino RB, Smith KR. Biomass stoves and lens opacity and cataract in Nepalese women. Optom Vis Sci. 2013;90:257–68.
Pant KP. Cheaper fuel and higher health costs among the poor in rural Nepal. Ambio. 2012;41:271–83.
Kinney PL, Asante KP, Lee AG, Ae-Ngibise KA, Burkart K, Boamah-Kaali E, et al. Prenatal and Postnatal Household Air Pollution Exposures and Pneumonia Risk: Evidence From the Ghana Randomized Air Pollution and Health Study. Chest. 2021;160:1634–44.
Lopuszanska U, Samardakiewicz M. The Relationship Between Air Pollution and Cognitive Functions in Children and Adolescents: A Systematic Review. Cogn Behav Neurol. 2020;33:157–78.
Kvestad I, Chandyo RK, Schwinger C, Ranjitkar S, Hysing M, Ulak M, et al. Biomass fuel use for cooking in Nepalese families and child cognitive abilities, results from a community-based study. Environ Res. 2022;212:113265.
DALYs GBD, Collaborators H, Murray CJ, Barber RM, Foreman KJ, Abbasoglu Ozgoren A, et al. Global, regional, and national disability-adjusted life years (DALYs) for 306 diseases and injuries and healthy life expectancy (HALE) for 188 countries, 1990-2013: quantifying the epidemiological transition. Lancet. 2015;386:2145–91.
Ghimire S, Mishra SR, Sharma A, Siweya A, Shrestha N, Adhikari B. Geographic and socio-economic variation in markers of indoor air pollution in Nepal: evidence from nationally-representative data. BMC Public Health. 2019;19:195.
NPC. National Reivew of Sustainable Developmental Goals. National Planning Commission (NPC). Kathmandu; Nepal; 2020. https://npc.gov.np/images/category/SDG_Review_Report_2020.pdf, accessed 27 August 2022.
Liu Y, Wang J, Huang Z, Liang J, Xia Q, Xia Q, et al. Environmental pollutants exposure: A potential contributor for aging and age-related diseases. Environ Toxicol Pharm. 2021;83:103575.
Blackburn EH, Epel ES, Lin J. Human telomere biology: A contributory and interactive factor in aging, disease risks, and protection. Science. 2015;350:1193–8.
Salpea KD, Humphries SE. Telomere length in atherosclerosis and diabetes. Atherosclerosis. 2010;209:35–8.
Buxton JL, Walters RG, Visvikis-Siest S, Meyre D, Froguel P, Blakemore AI. Childhood obesity is associated with shorter leukocyte telomere length. J Clin Endocrinol Metab. 2011;96:1500–5.
Albrecht E, Sillanpaa E, Karrasch S, Alves AC, Codd V, Hovatta I, et al. Telomere length in circulating leukocytes is associated with lung function and disease. Eur Respir J. 2014;43:983–92.
Alhareeri AA, Archer KJ, Fu H, Lyon DE, Elswick RK Jr, Kelly DL, et al. Telomere lengths in women treated for breast cancer show associations with chemotherapy, pain symptoms, and cognitive domain measures: a longitudinal study. Breast Cancer Res. 2020;22:137.
Masi S, Salpea KD, Li K, Parkar M, Nibali L, Donos N, et al. Oxidative stress, chronic inflammation, and telomere length in patients with periodontitis. Free Radic Biol Med. 2011;50:730–5.
Entringer S, Epel ES, Kumsta R, Lin J, Hellhammer DH, Blackburn EH, et al. Stress exposure in intrauterine life is associated with shorter telomere length in young adulthood. Proc Natl Acad Sci USA. 2011;108:E513–8.
Coluzzi E, Colamartino M, Cozzi R, Leone S, Meneghini C, O’Callaghan N, et al. Oxidative stress induces persistent telomeric DNA damage responsible for nuclear morphology change in mammalian cells. PLoS One. 2014;9:e110963.
Kaali S, Jack D, Delimini R, Hu L, Burkart K, Opoku-Mensah J, et al. Prenatal Household Air Pollution Alters Cord Blood Mononuclear Cell Mitochondrial DNA Copy Number: Sex-Specific Associations. Int J Environ Res Public Health. 2018;16:26–32.
Miri M, Nazarzadeh M, Alahabadi A, Ehrampoush MH, Rad A, Lotfi MH, et al. Air pollution and telomere length in adults: A systematic review and meta-analysis of observational studies. Environ Pollut. 2019;244:636–47.
Isaevska E, Moccia C, Asta F, Cibella F, Gagliardi L, Ronfani L, et al. Exposure to ambient air pollution in the first 1000 days of life and alterations in the DNA methylome and telomere length in children: A systematic review. Environ Res. 2021;193:110504.
Li S, Yang M, Carter E, Schauer JJ, Yang X, Ezzati M, et al. Exposure-Response Associations of Household Air Pollution and Buccal Cell Telomere Length in Women Using Biomass Stoves. Environ Health Perspect. 2019;127:87004.
Lee AG, Cowell W, Kannan S, Ganguri HB, Nentin F, Wilson A, et al. Prenatal particulate air pollution and newborn telomere length: Effect modification by maternal antioxidant intakes and infant sex. Environ Res. 2020;187:109707.
CBS. National Population and Housing Census. Central Bureau of Statistics (CBS). Government of Nepal. Preliminary report. Kathmandu; Nepal; 2021. https://censusnepal.cbs.gov.np/Home/Details?tpid=1&dcid=0f011f13-7ef6-42dd-9f03-c7d309d4fca3, accessed 27 August 2022.
Shrestha PS, Shrestha SK, Bodhidatta L, Strand T, Shrestha B, Shrestha R, et al. Bhaktapur, Nepal: the MAL-ED birth cohort study in Nepal. Clin Infect Dis 2014;59:S300–3.
Thygerson SM, Beard JD, House MJ, Smith RL, Burbidge HC, Andrus KN, et al. Air-Quality Assessment of On-Site Brick-Kiln Worker Housing in Bhaktapur, Nepal: Chemical Speciation of Indoor and Outdoor PM2.5 Pollution. Int J Environ Res Public Health. 2019;16:4114–9.
Neupane PR, Bajracharya I, Manandhar BR, Prajapati M, Sujakhu H, Awal P. Estimating Emission Load from Road Transportation within the Bhaktapur Municipality, Nepal. J Environ Public Health. 2020;2020:2828643.
AQM. Air Quality Monitoring: Government of Nepal, Ministry of Forests and Environment, Department of Environment; 2022. https://pollution.gov.np/.
Strand TA, Ulak M, Chandyo RK, Kvestad I, Hysing M, Shrestha M, et al. The effect of vitamin B12 supplementation in Nepalese infants on growth and development: study protocol for a randomized controlled trial. Trials. 2017;18:187.
Sudfeld CR, McCoy DC, Danaei G, Fink G, Ezzati M, Andrews KG, et al. Linear growth and child development in low- and middle-income countries: a meta-analysis. Pediatrics. 2015;135:e1266–75.
Strand TA, Ulak M, Hysing M, Ranjitkar S, Kvestad I, Shrestha M, et al. Effects of vitamin B12 supplementation on neurodevelopment and growth in Nepalese Infants: A randomized controlled trial. PLoS Med. 2020;17:e1003430.
Ulak M, Kvestad I, Chandyo RK, Ranjitkar S, Hysing M, Schwinger C, et al. The effect of infant vitamin B12 supplementation on neurodevelopment: A follow-up of a randomized placebo-controlled trial in Nepal. Br J Nutr. 2022:1–18.
Psaki SR, Seidman JC, Miller M, Gottlieb M, Bhutta ZA, Ahmed T, et al. Measuring socioeconomic status in multicountry studies: results from the eight-country MAL-ED study. Popul Health Metr. 2014;12:8.
Cawthon RM. Telomere measurement by quantitative PCR. Nucleic Acids Res. 2002;30:e47.
Durham T, Guo J, Cowell W, Riley KW, Wang S, Tang D, et al. Prenatal PM2.5 Exposure in Relation to Maternal and Newborn Telomere Length at Delivery. Toxics. 2022;10:13–18.
Bates MN, Chandyo RK, Valentiner-Branth P, Pokhrel AK, Mathisen M, Basnet S, et al. Acute lower respiratory infection in childhood and household fuel use in Bhaktapur, Nepal. Environ Health Perspect. 2013;121:637–42.
Siddharthan T, Pollard SL, Quaderi SA, Rykiel NA, Wosu AC, Alupo P, et al. Discriminative Accuracy of Chronic Obstructive Pulmonary Disease Screening Instruments in 3 Low- and Middle-Income Country Settings. JAMA. 2022;327:151–60.
Ly K, Walker C, Berry S, Snell R, Marks E, Thayer Z, et al. Telomere length in early childhood is associated with sex and ethnicity. Sci Rep. 2019;9:10359.
Codd V, Wang Q, Allara E, Musicha C, Kaptoge S, Stoma S, et al. Polygenic basis and biomedical consequences of telomere length variation. Nat Genet. 2021;53:1425–33.
Lin N, Mu X, Wang G, Ren Y, Su S, Li Z, et al. Accumulative effects of indoor air pollution exposure on leukocyte telomere length among non-smokers. Environ Pollut. 2017;227:1–7.
Kaali S, Jack D, Opoku-Mensah J, Bloomquist T, Aanaro J, Quinn A, et al. Prenatal Household Air Pollution Exposure, Cord Blood Mononuclear Cell Telomere Length and Age Four Blood Pressure: Evidence from a Ghanaian Pregnancy Cohort. Toxics. 2021;9:169–75.
Entringer S, de Punder K, Buss C, Wadhwa PD. The fetal programming of telomere biology hypothesis: an update. Philos Trans R Soc Lond B Biol Sci. 2018;373:1741–7.
Flannagan KS, Bowman AA, Mora-Plazas M, Marin C, Rentschler KM, Rozek LS, et al. Micronutrient status and leukocyte telomere length in school-age Colombian children. Eur J Nutr. 2020;59:1055–65.
Bates MN, Pokhrel AK, Chandyo RK, Valentiner-Branth P, Mathisen M, Basnet S, et al. Kitchen PM2.5 concentrations and child acute lower respiratory infection in Bhaktapur, Nepal: The importance of fuel type. Environ Res. 2018;161:546–53.
Clark ML, Peel JL, Balakrishnan K, Breysse PN, Chillrud SN, Naeher LP, et al. Health and household air pollution from solid fuel use: the need for improved exposure assessment. Environ Health Perspect. 2013;121:1120–8.
Acknowledgements
We thank the staff at the Child Health Research Project including the cognitive team, supervisors, data management team and all field workers. We are grateful to all participant children and their families for their valuable time to participate in the study. We are also grateful to Shyam Sunder Dhaubhadel and all staff at Siddhi Memorial Hospital.
Funding
This work was supported by Thrasher Research Fund (award # 11512), the GC Rieber foundation, the University of Bergen (UiB), and the Innlandet Hospital Trust (grant 150403).
Author information
Authors and Affiliations
Contributions
RKC conceptualised and designed the study, supervised the data collection, and performed the statistical analyses, interpretation of results and drafting of the initial manuscript. CS coordinated the statistical analyses, interpretation of the results, reviewed and revised the manuscript, approved the final manuscript. IK conceptualized and designed the study, supervised the data collection, coordinated the statistical analysis, reviewed, and revised the manuscript, and approved the final manuscript. TS conceptualised and designed the study, supervised the data collection, performed the statistical analysis and coordinated the laboratory analysis, interpretation of results, reviewed and revised the manuscript and approved the final manuscript. MU conceptualized and designed the study, coordinated, and supervised the data collection, reviewed, and revised the manuscript, and approved the final manuscript. SR coordinated and supervised the data collection, reviewed, and revised the manuscript, and approved the final manuscript. MS supervised the data collection, reviewed, and revised the manuscript, and approved the final manuscript. LN, DP and IV coordinated and analysed leukocyte telomere length, reviewed and revised the manuscript, and approved the final manuscript. LS coordinated the data collection, reviewed, and revised the manuscript, and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chandyo, R.K., Schwinger, C., Kvestad, I. et al. The association between household biomass fuel use and leukocyte telomere length among toddlers in Bhaktapur, Nepal. J Expo Sci Environ Epidemiol 33, 448–454 (2023). https://doi.org/10.1038/s41370-022-00474-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41370-022-00474-1