Abstract
Knowledge of human body composition at the dawn of the twentieth century was based largely on cadaver studies and chemical analyses of isolated organs and tissues. Matters soon changed by the nineteen twenties when the Czech anthropologist Jindřich Matiegka introduced an influential new anthropometric method of fractionating body mass into subcutaneous adipose tissue and other major body components. Today, one century later, investigators can not only quantify every major body component in vivo at the atomic, molecular, cellular, tissue-organ, and whole-body organizational levels, but go far beyond to organ and tissue-specific composition and metabolite estimates. These advances are leading to an improved understanding of adiposity structure-function relations, discovery of new obesity phenotypes, and a mechanistic basis of some weight-related pathophysiological processes and adverse clinical outcomes. What factors over the past one hundred years combined to generate these profound new body composition measurement capabilities in living humans? This perspective tracks the origins of these scientific innovations with the aim of providing insights on current methodology gaps and future research needs.
Similar content being viewed by others
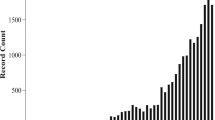
One-century ago the Roaring Twenties were in full swing, Europe was recovering from the destructive effects of World War I, and the Czech anthropologist Jindřich Matiegka had just published his classic treatise on the anthropometric fractionation of body mass into subcutaneous adipose tissue plus skin, skeletal muscle, skeleton, and remainder [1]. Up until Matiegka’s time information on human body composition, including adiposity, was sparse; the prevailing knowledge was built largely around earlier anatomic and chemical analyses of cadavers and excised tissues [2]. Investigators such as Camerer and Söldner had reported the chemical composition of fetuses as fat, nitrogen, major minerals, and water in 1900 [3], although similar notable experimental studies were published at a slow rate of only one or two every several years. Matters changed remarkably over the past one-hundred years: publications reporting human body composition studies are appearing in scientific literature at a rate of more than 3000 per year, totaling over 60,000 since Matiegka’s time.Footnote 1
Matiegka introduced his “somatotechnique” as a means of evaluating human physical efficiency following the “Great War” [1]. Deaths were declining from infectious diseases, although combat-related famines were soon to follow during World War II [4] and protein-calorie malnutrition prevailed during the mid-twentieth century in developing countries [5]. The physical effects, morbidity, and mortality related to semistarvation attracted a global cadre of eminent scientists whose careers were founded on the metabolic and body composition effects of undernutrition. Classic studies conducted in the aftermath of World War II such as the innovative Minnesota Starvation Experiment [6] still strongly influence investigators exploring topics related to body composition, metabolism, and obesity in humans [7, 8].
By the mid-1970s a different pattern of major killer diseases emerged: obesity appeared on the horizon [9] and heart disease replaced infectious diseases as the leading cause of death in the U.S. [10]. Adiposity phenotyping methods were still in an early stage of development; Spivak had introduced measurement of specific gravity in 1915 as a potential human vital sign [11] and by the 1960s the hydrodensitometry method for body fat estimation was still considered “far from accurate” [12]. Keys’ 1972 study of optimum adiposity power-type indices [13] leading to widespread use of body mass index relied largely on the sum of two skinfolds to quantify body fat in 7043 men of diverse backgrounds; 429 of the men also had body fat measured by hydrodensitometry. Major publications, such as the 1975 Reference Man [14], collated earlier data from the many studies reporting normative values acquired from cadavers and anthropometric measurements that were a hallmark of this era.
The emergence of obesity and related chronic diseases in the 1970s [9] prompted a new wave of body composition research aimed at developing methods of phenotyping people for adiposity and associated health risks. A second wave of body composition research emerged following Rosenberg’s 1989 report identifying sarcopenia as a major health concern [15]; sarcopenic-obesity soon appeared in other publications [16]. Developing methods for quantifying total body and regional adipose tissue and skeletal muscle mass became a major focus of research with specialized laboratories opening worldwide [2] and periodic international conferences devoted solely to advances in body composition methods and findings [17].
The broad research and clinical interest in human body composition is matched with a wide range of available measurement methods introduced over the past century. These methods are described in hundreds of publications, but most owe their origins to fundamental scientific discoveries during the nineteenth and twentieth centuries. The conceptual foundations for at least four families of methods are based on the Nobel-Prize winning discoveries of X-rays by Roentgen in 1895, radioactivity by Becquerel in 1896, the stable isotope deuterium by Urey in 1931, and nuclear magnetic resonance by Rabi in 1938 [18]. These fundamental discoveries led to downstream development of dozens of new methods for evaluating human body composition in general and specifically components related to adiposity [2]. The major families of new body composition methods and their historical foundations are visually depicted along with a timeline in Fig. 1 and an expanded summary in Table 1.
Expanded comments are provided in Table 1. ADP air displacement plethysmography, BIA bioimpedance analysis, DPA dual-photon absorptiometry, DXA dual-energy X-ray absorptiometry, INVA in vivo neutron activation analysis, MRI magnetic resonance imaging, MRS magnetic resonance spectroscopy, NMR nuclear magnetic resonance.
These sweeping discoveries in the short span of five decades and the seminal observations of several other investigators led to the “golden era” of human body composition method development between the 1930s and 1980s. Every major body component at the atomic, molecular, cellular, tissue-organ, and whole-body levels can now be measured in vivo using methods fully developed over the past century. Components such as adipose tissue and skeletal muscle mass have been fully characterized and can now be assessed in the whole body and selected regions across the full lifespan with great accuracy and precision. An important advance over recent decades is that body composition measurements can now be extended far past assessments of whole component volume and mass to chemical and cellular compositions. Examples include brown adipose tissue within the total body adipose tissue component [19], intermuscular adipose tissue and intramyocellular lipids within skeletal muscles [20, 21], intrahepatic lipid in liver [22], white and gray matter in the brain [23], muscle microarchitecture [24], and vast numbers of tissue metabolites [21]. These kinds of capabilities extend traditional body composition analysis to estimation of an organ or tissues “quality” in vivo. Many of the features of organ and tissue quality have been linked with metabolic and physiological disturbances in people with obesity.
In-depth analysis of component volume or mass and composition bring us closer to a deeper understanding of structure-function relationships. An example is the condition associated with obesity referred to as heart failure with preserved ejection fraction (HFpEF) [25]. Not only can total heart volume and mass (a component at the organ-tissue body composition level) be measured non-invasively, but evaluation of detailed myocardial structure, metabolism, and function can be quantified with methods such as magnetic resonance imaging/spectroscopy and echocardiography. Moreover, cardiac structure and function can be mechanistically associated with total adiposity, visceral adipose tissue, epicardial adipose tissue, and myocardial steatosis along with metabolic factors such as the level of insulin resistance [25]. These observations were recently amplified when Kosiborod et al. [26] showed that reductions in adiposity following administration of a glucagon-like peptide-1 receptor agonist significantly improved multiple physical functions in patients with obesity and HFpEF. Building structure-function-outcome relationships such as these were inconceivable only several decades ago and portend the future of research into “functional body composition” as suggested by Muller [27]. Discovery of new phenotypes is possible [28,29,30,31], particularly with advancing mathematical capabilities for analyzing large and complex data sets increasingly available in stored cloud sites [32].
These notable advances that thrust body composition research into a much wider sphere of basic and clinical research also bring in scientists from other disciplines with fresh points of view and ideas. Their publications likely account for the surge in human body composition reports appearing over the last few years. However, we are not yet at the “end” of body composition methodology research [33]. Regretfully, we still don’t have highly accurate, practical, and low-cost body composition methods that are widely applicable in clinical settings, epidemiological surveys, and even at home. Normative values for many methods are lacking, notably across samples varying widely in age, sex, and race/ethnicity. Devices often acquire manufacturer-specific measurements and employ non-disclosed population-specific empirical prediction algorithms. Comparisons of adiposity estimates across patients and studies is thus difficult and often impractical. Not all body components and their composition are yet measurable; to note several, the mass of the large gastrointestinal tract and its subcomponents, pancreatic beta-cell mass, and mitochondrial mass with associated energy expenditure still cannot be accurately quantified in vivo by body composition researchers. As shown in Fig. 1, most of the available current body composition methods evolved from only a few basic or seminal discoveries and current advances stem largely from refinements rather than innovation. Thus, there is work ahead for those of us whose interests lie in developing and applying methods for quantifying body composition in vivo.
Going back one century, could Matiegka working with a flexible tape and rigid ruler [1] have envisioned the advances in body composition and adiposity evaluation that we can now marshal at research centers with ease? Could Keys and Brozek studying adiposity changes with semi-starvation one-half century ago [13, 34] have imagined their sophisticated hydrodensitometry system for evaluating body fat percentage would be all but extinct in 2024? How will artificial intelligence, already sweeping into analyses of body composition data [35], create a new quantitative paradigm for the field? Can we, working at the forefront of body composition research, visualize what the field will look like in 2124? While clearly the future cannot be accurately predicted by most of us, we can only hope that our scientific descendants will experience another golden age in body composition and obesity research.
Notes
These data come from a PubMed search conducted January 2, 2024 using the search terms “human” and “body composition.”
References
Matiegka J. The testing of physical efficiency. Am J Phys Anthropol. 1921;4:223–30.
Wang Z, Wang ZM, Heymsfield SB. History of the study of human body composition: A brief review. Am J Hum Biol. 1999;11:157–65.
Camerer W, Soldner D. Die chemische Zusammensetzung des Neugeborenen. Z Biol. 1900;39:173–92.
Grzybowski A, Pawlikowska-Lagod K. Izrael Milejkowski and Hunger Disease Study in the Warsaw Ghetto. Clin Dermatol. 2023;41:159–65.
Nott J. Malnutrition in a Modernising Economy: The Changing Aetiology and Epidemiology of Malnutrition in an African Kingdom, Buganda c.1940-73. Med Hist. 2016;60:229–49.
Keys AB, Brozek J, Henschel A, Mickelsen O, Taylor HL. The biology of human starvation. Minneapolis: University of Minnesota Press; 1950.
Dulloo AG. Physiology of weight regain: Lessons from the classic Minnesota Starvation Experiment on human body composition regulation. Obes Rev. 2021;22:e13189.
Muller MJ, Enderle J, Pourhassan M, Braun W, Eggeling B, Lagerpusch M, et al. Metabolic adaptation to caloric restriction and subsequent refeeding: the Minnesota Starvation Experiment revisited. Am J Clin Nutr. 2015;102:807–19.
Bray GA. Beyond BMI. Nutrients. 2023;15:2254.
Hoyert DL. 75 years of mortality in the United States, 1935–2010. NCHS data brief, No. 88. Hyattsville, MD: National Center for Health Statistics; 2012.
Spivak CD. The Specific Gravity of the Human Body. Arch Int Med. 1915;15:628–42.
Billewicz WZ, Kemsley WF, Thomson AM. Indices of adiposity. Br J Prev Soc Med. 1962;16:183–8.
Keys A, Fidanza F, Karvonen MJ, Kimura N, Taylor HL. Indices of relative weight and obesity. J Chronic Dis. 1972;25:329–43.
Snyder WS, Cook MJ, Nasset ES, Karhausen LR, Howells GP, Tipton IH. Report of the task group on reference man. Oxford: Pergamon Press; 1975.
Rosenberg IH. Sarcopenia: origins and clinical relevance. J Nutr. 1997;127:990S–991S.
Heber D, Ingles S, Ashley JM, Maxwell MH, Lyons RF, Elashoff RM. Clinical detection of sarcopenic obesity by bioelectrical impedance analysis. Am J Clin Nutr. 1996;64:472S–477S.
International Symposium on Body Composition Research (ISBCR). 12th International Symposium on In Vivo Body Composition Studies. ISBCR. Available at: https://www.isbcr.org/. Last updated Accessed: 24 February 2024.
NobelPrize.org. All Nobel Prizes. Nobel Prize Outreach. Available at: https://www.nobelprize.org/prizes/lists/all-nobel-prizes/. Last updated 2024. Accessed: 12 January 2024.
Flanagan EW, Altazan AD, Carmichael OT, Hu HH, Redman LM. Practical application of in vivo MRI-based brown adipose tissue measurements in infants. Obesity. 2021;29:1676–83.
Gallagher D, Kuznia P, Heshka S, Albu J, Heymsfield SB, Goodpaster B, et al. Adipose tissue in muscle: a novel depot similar in size to visceral adipose tissue. Am J Clin Nutr. 2005;81:903–10.
Krssak M, Lindeboom L, Schrauwen-Hinderling V, Szczepaniak LS, Derave W, Lundbom J, et al. Proton magnetic resonance spectroscopy in skeletal muscle: Experts’ consensus recommendations. NMR Biomed. 2021;34:e4266.
Bosy-Westphal A, Braun W, Albrecht V, Muller MJ. Determinants of ectopic liver fat in metabolic disease. Eur J Clin Nutr. 2019;73:209–14.
Geisler C, Muller MJ. Is There a Contribution of Structural Brain Phenotypes to the Variance in Resting Energy Expenditure before and after Weight Loss in Overweight Females? Nutrients. 2019;11:2759.
Hooijmans MT, Schlaffke L, Bolsterlee B, Schlaeger S, Marty B, Mazzoli V. Compositional and Functional MRI of Skeletal Muscle: A Review. J Magn Reson Imaging. 2023. https://doi.org/10.1002/jmri.29091.
Borlaug BA, Jensen MD, Kitzman DW, Lam CSP, Obokata M, Rider OJ. Obesity and heart failure with preserved ejection fraction: new insights and pathophysiological targets. Cardiovasc Res. 2023;118:3434–50.
Kosiborod MN, Abildstrom SZ, Borlaug BA, Butler J, Rasmussen S, Davies M, et al. Semaglutide in Patients with Heart Failure with Preserved Ejection Fraction and Obesity. N. Engl J Med. 2023;389:1069–84.
Muller MJ, Bosy-Westphal A, Later W, Haas V, Heller M. Functional body composition: insights into the regulation of energy metabolism and some clinical applications. Eur J Clin Nutr. 2009;63:1045–56.
Linge J, Borga M, West J, Tuthill T, Miller MR, Dumitriu A, et al. Body Composition Profiling in the UK Biobank Imaging Study. Obesity. 2018;26:1785–95.
Linge J, Whitcher B, Borga M, Dahlqvist Leinhard O. Sub-phenotyping Metabolic Disorders Using Body Composition: An Individualized, Nonparametric Approach Utilizing Large Data Sets. Obesity. 2019;27:1190–9.
Muller MJ, Geisler C, Hubers M, Pourhassan M, Bosy-Westphal A. Body composition-related functions: a problem-oriented approach to phenotyping. Eur J Clin Nutr. 2019;73:179–86.
Prado CM, Siervo M, Mire E, Heymsfield SB, Stephan BC, Broyles S, et al. A population-based approach to define body-composition phenotypes. Am J Clin Nutr. 2014;99:1369–77.
Sobhiyeh S, Dunkel A, Dechenaud M, Mehrnezhad A, Kennedy S, Shepherd J, et al. Digital anthropometric volumes: Toward the development and validation of a universal software. Med Phys. 2021;48:3654–64.
Heymsfield SB, Pietrobelli A, Wang Z, Saris WH. The end of body composition methodology research? Curr Opin Clin Nutr Metab Care. 2005;8:591–4.
Brozek J, Grande F, Anderson JT, Keys A. Densitometric Analysis of Body Composition: Revision of Some Quantitative Assumptions. Ann N Y Acad Sci. 1963;110:113–40.
Santhanam P, Nath T, Peng C, Bai H, Zhang H, Ahima RS, et al. Artificial intelligence and body composition. Diabetes Metab Syndr. 2023;17:102732.
Wikipedia contributors. Alphonse Bertillon. Wikipedia, The Free Encyclopedia. Available at: https://en.wikipedia.org/w/index.php?title=Alphonse_Bertillon&oldid=1182268640. Last updated 28 October 2023. Accessed: 12 January 2024.
Jones PR, West GM, Harris DH, Read JB. The Loughborough anthropometric shadow scanner (LASS). Endeavour. 1989;13:162–8.
Stuart HCDP. The growth of bone, muscle, and overlying tissues in children 6t o 10 years of age as revealed by studies of roentgenograms of the leg area. Monog rSoc Res Child Dev. 1942;13:195.
Cormack AM. Nobel award address. Early two-dimensional reconstruction and recent topics stemming from it. Med Phys. 1980;7:277–82.
Hounsfield GN. Computerized transverse axial scanning (tomography). 1. Description of system. Br J Radio. 1973;46:1016–22.
Blake GM, Fogelman I. The role of DXA bone density scans in the diagnosis and treatment of osteoporosis. Postgrad Med J. 2007;83:509–17.
Oliphant ML, Harteck P, Rutherford E. Transmutation Effects observed with Heavy Hydrogen. Nature. 1934;133:413.
Landsberger S. Discovery of neutron activation analysis. Appl Radiat Isot. 2022;189:110422.
Anderson J, Osborn SB, Tomlinson RW, Newton D, Rundo J, Salmon L, et al. Neutron-Activation Analysis in Man in Vivo. A New Technique in Medical Investigation. Lancet. 1964;2:1201–5.
Mazess RB, Cameron JR, Sorenson JA. Determining body composition by radiation absorption spectrometry. Nature. 1970;228:771–2.
Behnke AR. Physiologic Studies Pertaining to Deep Sea Diving and Aviation, Especially in Relation to the Fat Content and Composition of the Body: The Harvey Lecture, March 19, 1942. Bull N Y Acad Med. 1942;18:561–85.
McCrory MA, Gomez TD, Bernauer EM, Mole PA. Evaluation of a new air displacement plethysmograph for measuring human body composition. Med Sci Sports Exerc. 1995;27:1686–91.
Hevesy G, Hofer E. Elimination of water from human body. Nature. 1934;134:879.
Rabi II, Millman S, Kusch P, Zacharias JR. The molecular beam resonance method for measuring nuclear magnetic moments. The magnetic moments of 3Li6, 3Li7, and 9F19. Phys Rev. 1939;55:526–35.
Bloch F. The Principle of Nuclear Induction. Science. 1953;118:425–30.
Purcell EM. Research in nuclear magnetism. In: Nobel lectures, physics: 1942–1962. Amsterdam: Elsevier; 1962, p 232.
Becker C. A sharper image. Father of the MRI honored for invention that forever changed diagnostics. Mod Health. 2001;31:44.
Shampo MA, Kyle RA. Karl Theodore Dussik-pioneer in ultrasound. Mayo Clin Proc. 1995;70:1136.
Pace N, Rathbun EN. Studies on body composition, III. Water and chemically contained nitrogen content in relation to fat content. J Biol Chem. 1945;158:685–91.
Heymsfield SB, Ebbeling CB, Zheng J, Pietrobelli A, Strauss BJ, Silva AM, et al. Multi-component molecular-level body composition reference methods: evolving concepts and future directions. Obes Rev. 2015;16:282–94.
Nyboer J, Kreider MM, Hannapel L. Quantitative studies of electrical conductivity of the peripheral body segments; basic and practical considerations. Ann West Med Surg. 1951;5:11–20.
Thomasset MA. [Bioelectric properties of tissue. Impedance measurement in clinical medicine. Significance of curves obtained]. Lyon Med. 1962;94:107–18.
Hoffer EC, Meador CK, Simpson DC. Correlation of whole-body impedance with total body water volume. J Appl Physiol. 1969;27:531–4.
Acknowledgements
The author acknowledges the support of Ms. Melanie Peterson in preparing this manuscript.
Funding
This review was partially supported by National Institutes of Health NORC Center Grants P30DK072476, Pennington/Louisiana, P30DK040561, Harvard.
Author information
Authors and Affiliations
Contributions
Author contributions to manuscript: SBH drafted the paper and had primary responsibility for final content.
Corresponding author
Ethics declarations
Competing interests
SBH serves on the Medical Advisory Boards of Tanita Corporation, Novo Nordisk, Abbott, and Medifast.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Heymsfield, S.B. Advances in body composition: a 100-year journey. Int J Obes (2024). https://doi.org/10.1038/s41366-024-01511-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41366-024-01511-9