Abstract
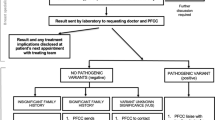
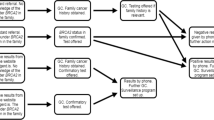
Genetic testing advances have enabled the provision of previously unavailable information on the pathogenicity of genetic variants, frequently necessitating the recontact of former patients by clinicians. In Japan, national health insurance coverage was extended to BRCA1/2 testing for the diagnosis of hereditary breast and ovarian cancer for patients who meet certain criteria in 2020, and conditions necessitating recontact were expected to increase. Studies and discussions regarding recontact have been conducted in the U.S. and Europe; however, in Japan, the national discussion around recontact remains undeveloped. We conducted a cross-sectional study by interviewing 73 facilities accredited by the Japanese Organization of Hereditary Breast and Ovarian Cancer regarding the practice of recontacting patients at these facilities. Sixty-six facilities responded that they recontact patients, but only 17 facilities had a protocol for this. The most common reason for recontact was that it could benefit the patient. Facilities that did not recontact stated that they lacked the necessary personnel or services. Most facilities indicated that a recontact system should be implemented in their practice. The increased burden on too few medical personnel, unestablished systems, patient confusion, and the right not to know were cited as barriers to implementing recontact. Although developing recommendations on recontact would be useful for providing equitable healthcare in Japan, there is an urgent need to deepen the discussion on recontacting, as negative opinions about recontacting patients were observed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Andorno R. The right not to know: an autonomy based approach. J Med Ethics. 2004;30:435–9.
Otten E, Plantinga M, Birnie E, Verkerk MA, Lucassen AM, Ranchor AV, et al. Is there a duty to recontact in light of new genetic technologies? A systematic review of the literature. Genet Med. 2015;17:668–78.
Carrieri D, Lucassen AM, Clarke AJ, Dheensa S, Doheny S, Turnpenny PD, et al. Recontact in clinical practice: a survey of clinical genetics services in the United Kingdom. Genet Med. 2016;18:876–81.
Sirchia F, Carrieri D, Dheensa S, Benjamin C, Kayserili H, Cordier C, et al. Recontacting or not recontacting? A survey of current practices in clinical genetics centres in Europe. Eur J Hum Genet. 2018;26:946–54.
Giesbertz NAA, van Harten WH, Bredenoord AL. A duty to recontact in genetics: context matters. Nat Rev Genet. 2019;20:371–2.
Mitchell C, Ploem C, Retèl V, Gevers S, Hennekam R. Experts reflecting on the duty to recontact patients and research participants; why professionals should take the lead in developing guidelines. Eur J Med Genet. 2020;63:103642.
Hirschhorn K, Fleisher LD, Godmilow L, Howell RR, Lebel RR, McCabe ER, et al. Duty to re-contact. Genet Med. 1999;1:171–2.
David KL, Best RG, Brenman LM, Bush L, Deignan JL, Flannery D, et al. Patient re-contact after revision of genomic test results: points to consider-a statement of the American College of Medical Genetics and Genomics (ACMG). Genet Med. 2019;21:769–71.
Bombard Y, Brothers KB, Fitzgerald-Butt S, Garrison NA, Jamal L, James CA, et al. The responsibility to recontact research participants after reinterpretation of genetic and genomic research results. Am J Hum Genet. 2019;104:578–95.
Carrieri D, Howard HC, Benjamin C, Clarke AJ, Dheensa S, Doheny S, et al. Recontacting patients in clinical genetics services: recommendations of the European Society of Human Genetics. Eur J Hum Genet. 2019;27:169–82.
Vora BB, Mountain H, Nichols C, Schofield L. Opinions and experiences of recontacting patients: a survey of Australasian genetic health professionals. J Community Genet. 2022;13:193–9.
van El CG, Cornel MC, Borry P, Hastings RJ, Fellmann F, Hodgson SV, et al. Whole-genome sequencing in health care: recommendations of the European Society of Human Genetics. Eur J Hum Genet. 2013;21:580–4.
The Japanese Association of Medical Sciences. Guidelines for Genetic Tests and Diagnoses in Medical Practice, 2011. https://jams.med.or.jp/guideline/genetics-diagnosis_e.pdf Accessed 29 Apr 2022.
Guidelines for Diagnosis and Treatment of Hereditary Breast and Ovarian Cancer 2021. https://johboc.jp/guidebook_2021/ Accessed 10 Dec 2022.
Actionability Summary Report Japanese Edition. http://www.idenshiiryoubumon.org/actionability_japan/index.html Accessed 29 Apr 2022.
Harrison SM, Rehm HL. Is ‘likely pathogenic’ really 90% likely? Reclassification data in ClinVar. Genome Med. 2019;11:72.
Esterling L, Wijayatunge R, Brown K, Morris B, Hughes E, Pruss D, et al. Impact of a cancer gene variant reclassification program over a 20-year period. JCO Precis Oncol. 2020;4:PO.20.00020.
Mersch J, Brown N, Pirzadeh-Miller S, Mundt E, Cox HC, Brown K, et al. Prevalence of variant reclassification following hereditary cancer genetic testing. JAMA. 2018;320:1266–74.
Acknowledgements
We thank the staff of all facilities accredited by the Japanese Organization of Hereditary Breast and Ovarian Cancer who participated in this study. We also thank Dr. Akihiro Sakurai, Sapporo Medical University, Japan, and Dr. Yumiko Kobayashi, Iwate Medical University, Japan, for their insight and suggestions. We would like to thank Editage for English language editing.
Author information
Authors and Affiliations
Contributions
Conceived and designed the study: TS, TT. Conducted a questionnaire: TS. Analyzed the data and conceived figures and tables: TS, TT, AY. Drafted the manuscript: TS. Revised the manuscript: TS, TT. Contributed to the writing of the manuscript: AY, KY, KO, DC, SEK, AF. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sakaguchi, T., Tokutomi, T., Yoshida, A. et al. Recontact: a survey of current practices and BRCA1/2 testing in Japan. J Hum Genet 68, 551–557 (2023). https://doi.org/10.1038/s10038-023-01149-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-023-01149-x