Abstract
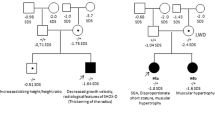
Although ACAN heterozygous loss-of-function variants often cause idiopathic short stature (ISS) phenotype, there is no report describing ISS phenotype caused by ACAN biallelic loss-of-function variants. We encountered a 4 1/12-year-old Japanese girl with a height of 80.4 cm (−5.2 SD), a weight of 11.4 kg (−1.9 SD), a head circumference of 48.7 cm (−0.6 SD), and an arm span/height ratio of 1.0 (+1.1 SD). Endocrine studies and bone survey showed no abnormal findings. Whole exome sequencing revealed biallelic rare variants in ACAN, i.e., NM_013227.4:c.4214delC:p.(Pro1405Leufs*3) derived from her father and paternal grandfather with short stature (−2.9 and −2.0 SD, respectively) and NM_013227.4:c.7124 A>G:p.(Gln2375Arg) inherited from her mother and maternal grandmother with short stature (−2.1 and −3.0 SD, respectively). The frameshift variant underwent nonsense mediated mRNA decay, and the missense variant was assessed to have high pathogenicity. The results imply for the first time that ACAN biallelic loss-of-function variants can cause severe ISS phenotype.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gibson BG, Briggs MD. The aggrecanopathies; an evolving phenotypic spectrum of human genetic skeletal diseases. Orphanet J Rare Dis. 2016;11:86. https://doi.org/10.1186/s13023-016-0459-2.
Hauer NN, Sticht H, Boppudi S, Büttner C, Kraus C, Trautmann U, et al. Genetic screening confirms heterozygous mutations in ACAN as a major cause of idiopathic short stature. Sci Rep. 2017;7:12225. https://doi.org/10.1038/s41598-017-12465-6.
Anzo M, Ito Y, Kanzaki S, Satoh K, Satoh M, Shinoda K, et al. on behalf of Bone Age Committee in the Japanese Association for Human Auxology and the Japanese Society for Pediatric Endocrinology. Bone Age for the Japanese assessed by the Tanner-Whitehouse 2 Methods. Medical Review, Tokyo, 2018 (in Japanese).
Nogayama T, Inokuchi M, Hasegawa T. Arm span, arm span-height ratio, and difference centiles for Japanese children. J Jpn Pediatr Soc. 2017;121:343.
Matsuura S, Kajii T. Radiographic measurements of metacarpophalangeal lengths in Japanese children. Jpn J Hum Genet. 1989;34:159–168. https://doi.org/10.1007/BF01912484.
Nakashima M, Tohyama J, Nakagawa E, Watanabe Y, Siew CG, Kwong CS, et al. Identification of de novo CSNK2A1 and CSNK2B variants in cases of global developmental delay with seizures. J Hum Genet. 2019;64:313–322. https://doi.org/10.1038/s10038-018-0559-z.
Kuzmiak HA, Maquat LE. Applying nonsense-mediated mRNA decay research to the clinic: progress and challenges. Trends Mol Med. 2006;12:306–316. https://doi.org/10.1016/j.molmed.2006.05.005.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. ACMG Laboratory Quality Assurance Committee. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–424. https://doi.org/10.1038/gim.2015.30.
Tompson SW, Merriman B, Funari VA, Fresquet M, Lachman RS, Rimoin DL, et al. A recessive skeletal dysplasia, SEMD aggrecan type, results from a missense mutation affecting the C-type lectin domain of aggrecan. Am J Hum Genet. 2009;84:72–79. https://doi.org/10.1016/j.ajhg.2008.12.001.
Fukuhara Y, Cho SY, Miyazaki O, Hattori A, Seo JH, Mashima R, et al. The second report on spondyloepimetaphyseal dysplasia, aggrecan type: a milder phenotype than originally reported. Clin Dysmorphol. 2019;28:26–29. https://doi.org/10.1097/MCD.0000000000000241.
Lundell A, Olin AI, Moergelin M, al-Karadaghi S, Aspberg A, Logan DT. Structural basis for interactions between tenascins and lectican C-type lectin domains: evidence for a crosslinking role for tenascins. Structure. 2004;12:1495–1506. https://doi.org/10.1016/j.str.2004.05.021.
Gkourogianni A, Andrew M, Tyzinski L, Crocker M, Douglas J, Dunbar N, et al. Clinical Characterization of Patients With Autosomal Dominant Short Stature due to Aggrecan Mutations. J Clin Endocrinol Metab. 2017;102:460–469. https://doi.org/10.1210/jc.2016-3313.
Gerver WJM, Gkourogianni A, Dauber A, Nilsson O, Wit JM. ArmSpan. and Its Relation to Height in a 2- to 17-Year-Old Reference Population and Heterozygous Carriers of ACAN Variants. Horm Res Paediatr. 2020;93:164–172. https://doi.org/10.1159/000508500.
Nilsson O, Guo MH, Dunbar N, Popovic J, Flynn D, Jacobsen C, et al. Short stature, accelerated bone maturation, and early growth cessation due to heterozygous aggrecan mutations. J Clin Endocrinol Metab. 2014;99:E1510–1518. https://doi.org/10.1210/jc.2014-1332.
Sentchordi-Montané L, Aza-Carmona M, Benito-Sanz S, Barreda-Bonis AC, Sánchez-Garre C, Prieto-Matos P, et al. Heterozygous aggrecan variants are associated with short stature and brachydactyly: Description of 16 probands and a review of the literature. Clin Endocrinol. 2018;88:820–829. https://doi.org/10.1111/cen.13581.
Bonioli E, Tarò M, La Rosa C, Citana A, Bertorelli R, Morcaldi G, et al. Heterozygous mutations of growth hormone receptor gene in children with idiopathic short stature. Growth Horm IGF Res. 2005;15:405–410. https://doi.org/10.1016/j.ghir.2005.08.004.
Noorian S, Khonsari NM, Savad S, Hakak-Zargar B, Voth T, Kabir K. Whole-Exome Sequencing in Idiopathic Short Stature: Rare Mutations Affecting Growth. J Pediatr Genet. 2020;10:284–291. https://doi.org/10.1055/s-0040-1716400.
Acknowledgements
We are grateful to the patient and her family members for their cooperation. We would like to thank Ms. Fumiko Kato, Ms. Aya Kitamoto, and Mr. Naoki Adachi for their support in molecular analyses.
Funding
This work was supported by the grants from Japan Agency for Medical Research and Development (AMED) (JP19ek0109297 to HS and JP21ek0109549 to TO).
Author information
Authors and Affiliations
Contributions
YM, YF, MF and HS performed molecular studies. YO, GN, TU and TO obtained clinical data and blood samples. TO designed the study, and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Masunaga, Y., Ohkubo, Y., Nishimura, G. et al. ACAN biallelic variants in a girl with severe idiopathic short stature. J Hum Genet 67, 481–486 (2022). https://doi.org/10.1038/s10038-022-01030-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-022-01030-3