Abstract
Background
Little is known on amplitude-integrated electroencephalography (aEEG) during the first few days after birth in neonates with congenital heart disease (CHD). Our aim was, therefore, to assess electrocortical activity using aEEG within the first 72 h after birth in neonates diagnosed prenatally with CHD, and to define independent prenatal and postnatal predictors for abnormal aEEG.
Methods
Neonates with CHD who were admitted to the neonatal intensive care unit between 2010 and 2017 were retrospectively included. We assessed aEEG background patterns, sleep–wake cycling, and epileptic activity during the first 72 h after birth and defined prenatal and postnatal clinical parameters associated with aEEG patterns.
Results
Seventy-two neonates were included. Twenty-six (36%) had mildly abnormal and six (8%) had severely abnormal aEEG background patterns at some point during the study period. Sleep–wake cycling was present in 97% of the neonates. Subclinical seizures were common (15%), whereas none of the neonates had clinical seizures. Only treatment with sedatives was a significant predictor for abnormal aEEG background patterns, explaining 56% of the variance.
Conclusion
Abnormal aEEG background patterns are common and are strongly associated with treatment with sedatives in neonates with prenatally diagnosed CHD. Future studies should assess the association between early postnatal aEEG abnormalities and neurodevelopmental outcome.
Similar content being viewed by others
Main
Treatment of congenital heart disease (CHD) improved tremendously, and nowadays most children with CHD survive into adulthood and the adult population with CHD outnumbers the pediatric population (1). Focus of attention has therefore shifted from preventing mortality toward optimizing neurodevelopmental outcome (NDO). Up to 50% of the neonates with CHD experience neurodevelopmental impairments later in life (1, 2). Brain injury, responsible for neurodevelopmental impairments, might already occur during early postnatal or even intrauterine life.
Amplitude-integrated electroencephalography (aEEG) is a validated bedside clinical tool to continuously monitor electrocortical activity after birth (3). Previous studies have reported that neonates with CHD often have abnormal background patterns and absence of sleep–wake cycling (SWC) before surgery (4, 5, 6, 7). Furthermore, up to 23% of the neonates with CHD had intraoperative seizures and delayed recovery of background patterns and SWC after surgical procedures (6, 7). Although no clear associations between preoperative aEEG abnormalities and NDO have been demonstrated, postoperative aEEG abnormalities were associated with adverse NDO in infants with CHD. Delayed recovery of background patterns after surgery was associated with a 14-point decrease in motor composite score at 2 years of age (6). Furthermore, postoperative abnormal aEEG background patterns and failure to return to SWC were associated with lower IQ scores (up to 15 points) at 4 years of age (8).
Previous studies on aEEG in neonates with CHD included neonates who were diagnosed postnatally or only used aEEG monitoring for a short period immediately before surgery. Little is known about aEEG patterns during the first few days after birth in neonates with CHD. Transition from fetal to neonatal life is a very complex process of physiological adaptations. Neonates with CHD are at an increased risk for disturbances during this process, complicating the transition (9, 10). Furthermore, fetuses with CHD already show circulatory alterations from the second trimester onward that are associated with acute or chronic hypoxia and adverse NDO (11, 12, 13).
The first aim of this study was, therefore, to describe aEEG background patterns, presence, and onset of SWC, and epileptic activity within the first 72 h after birth in neonates with prenatally diagnosed CHD that were admitted to the neonatal intensive care unit (NICU) immediately after birth. The second aim was to assess prenatal and neonatal clinical parameters that were independently associated with aEEG. We hypothesized that neonates with CHD frequently have aEEG abnormalities during the first 3 days after birth as brain injury might already occur prenatally in patients with CHD. Furthermore, we hypothesized that abnormal prenatal Doppler flow patterns (i.e., low pulsatility index of the middle cerebral artery (MCA-PI), high pulsatility index of the umbilical artery (UA-PI), and low cerebroplacental ratio (CPR)) are associated with postnatal aEEG.
Methods
Participants
This was a retrospective study at the Fetal Medicine Unit and the tertiary NICU of the University Medical Center Groningen. All neonates with prenatally diagnosed CHD that were born in our hospital and admitted to the NICU after birth between January 2010 and January 2017 were considered for inclusion. Only those neonates with at least 24 h of aEEG monitoring within the first 72 h after birth were enrolled in this study. Neonates who were born before 36 weeks of gestational age were excluded from participation. The Medical Ethics Review Board University Medical Center Groningen approved the study.
Amplitude-Integrated Electroencephalography
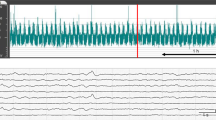
Amplitude-integrated aEEG was routinely recorded according to a local protocol using either the digital Olympic CFM 6000 or the Olympic Brainz Monitor. The nursing staff recorded clinical seizures, and all factors that could disturb aEEG signals such as nursing and medical procedures, and administration of medication. The aEEG recordings were assessed independently by two investigators (M.J.M. and N.J.E.O.). One investigator (N.J.E.O.) was blinded to the clinical condition of the patient. In case of disagreement, we consulted a third investigator (H.Ja.t.H.) who was also unaware of the clinical condition. Final decisions were made based on consensus. Background patterns, the presence of SWC, and epileptic activity were assessed. We distinguished five different types of background patterns according to the criteria of Toet et al. (3): continuous normal voltage (CNV), discontinuous normal voltage (DNV), continuous low voltage (CLV), burst suppression (BS), and flat trace (FT). CNV was considered to be a normal background pattern, DNV was considered to be mildly abnormal, and CLV, BS, and FT were considered to be severely abnormal background patterns. SWC was recognized by periodic changes in bandwidth of the aEEG tracing. For each included neonate, we assessed whether SWC was present and for neonates who were monitored within the first 12 h after birth, we also recorded the time of onset of SWC. Epileptic activity was classified as a single seizure, multiple seizures, and status epilepticus (>50% seizure activity during a 1-h epoch).
Clinical Parameters
During pregnancy, MCA-PI and UA-PI were repeatedly measured by Doppler ultrasonography, and CPR was calculated (CPR=MCA-PI/UA-PI) as part of routine prenatal care in fetuses with CHD. For this study, we selected the last available Doppler measurement before birth and converted all values into z scores to adjust for differences in gestational age.
After birth, all available clinical and biochemical parameters that could influence aEEG monitoring were collected. These variables included mode of delivery, Apgar score at 5 min, pH of the umbilical artery, lactate, whether the neonates underwent a Rashkind procedure, abnormality on cranial ultrasound (i.e., germinal matrix hemorrhage, slit-like ventricles, or choroid plexus cysts), and medical therapy (particularly sedatives and anticonvulsants). Additional information that was collected from the patients’ medical files included type of CHD, gestational age at birth, birth weight, gender, and mortality. For statistical purposes, the type of CHD was categorized in cyanotic and acyanotic cardiac defects. Coarctation of the aorta, aortic valve stenosis, aortic arch interruption, and hypoplastic left heart syndrome were considered to be acyanotic CHD. Transposition of the great arteries, double outlet right ventricle, pulmonary valve stenosis, pulmonary atresia with hypoplasia of the right ventricle, tetralogy of Fallot, atrioventricular septal defect, tricuspid atresia, truncus arteriosus, and complex monoventricular hearts were considered to be cyanotic CHD.
Statistical Analysis
We used SPSS version 23.0 (IBM Corp., Armonk, NY, USA) for statistical analyses and GraphPad PRISM version 5 (GraphPad Software Inc, CA, USA) for graphical display of our results. We used descriptive statistics to present the course of aEEG background patterns, SWC, and epileptic activity in neonates with prenatally diagnosed CHD that were admitted to the NICU. In addition, univariate and multiple logistic regression analyses were used to determine which variables were independently associated with aEEG background patterns, SWC, and epileptic activity. All parameters identified by univariate logistic regression analysis with a P value ≤0.05 were selected for multiple logistic regression analyses (enter method). A P value <0.05 was considered to be statistically significant.
Results
Participants
We identified 436 patients with prenatally diagnosed CHD between January 2010 and January 2017 (Figure 1). Of these patients, 160 required NICU admission after birth. Twenty-four neonates were excluded because of preterm birth and 46 neonates were not monitored with aEEG during the first 72 h after birth because of logistic reasons. As a result, 90 term-born neonates were eligible for inclusion, of whom 72 were monitored for more than 24 h within the first 72 h after birth. One of the included neonates was diagnosed with trisomy 21 after birth. Patient characteristics of the 72 included neonates are presented in Table 1. Patient characteristics of the 64 eligible but excluded neonates compared with the 72 included neonates are presented in Supplementary Table S1 online. Furthermore, a comparison between neonates who were monitored >24 h and neonates who were monitored <24 h within the first 72 h birth is presented in Supplementary Table S2.
Amplitude-Integrated Electroencephalography
The median (range) duration of aEEG recordings was 72.0 (24.4–277.8) hours and aEEG monitoring was initiated at 3.1 (0.8–40.0) hours after birth. In nine neonates (13%), aEEG monitoring was not continued until 72 h after birth. All these neonates had CNV and no signs of epileptic activity during the available monitoring time (median duration 45.5 h, range 24.8–47.4 h). Thirteen out of sixty-three neonates (21%) with 72 h of aEEG recordings had abnormal background patterns at 72 h after birth (12 DNV and one CLV). Duration of aEEG monitoring was shorter in neonates with normal aEEG background patterns in comparison with neonates with abnormal background patterns (median duration of 63.5 vs. 96.4 h, P<0.001).
The results regarding aEEG monitoring are summarized in Figure 2. Forty neonates (56%) had normal background patterns during the first 72 h after birth. Twenty-six neonates (36%) had mildly abnormal (DNV) and six neonates (8%) had severely abnormal background patterns (BS, CLV, and FT) at some point during the first 72 h after birth. Neonates with aEEG monitoring <24 h within the first 72 h after birth had similar background patterns (Supplementary Figure S1). All but two of the included neonates (3%) had SWC within the first 72 h after birth. In 57 neonates (79%), aEEG monitoring started within 12 h after birth. In these neonates, the first SWC occurred after 10.5 (3.0–44.5) hours, and 54 infants (95%) had their first SWC within 36 h after birth. The time of onset of SWC occurred earlier in neonates with normal aEEG background patterns in comparison with neonates with abnormal aEEG background patterns (6.0 vs. 16.0 h after birth, P<0.01). There were no clinical seizures reported during the study period. Subclinical seizures were present in 11 (15%) neonates. Four neonates had a single subclinical seizure during the study period, six neonates had multiple seizures, and one neonate had a status epilepticus. The majority of the seizures (67%) occurred on the second day after birth.
Amplitude-integrated electroencephalography background patterns. (a) Distribution of background patterns based on the % of the study population. (b) Distribution of background patterns based on the % of the study period. BS, burst suppression; CLV, continuous low voltage; CNV, continuous normal voltage; DNV, discontinuous normal voltage; N, normal (CNV); MA, mildly abnormal (DNV); SA, severely abnormal (BS, CLV). * indicates abnormal background pattern at some point during the study period.
Clinical Characteristics and Biochemical Parameters
The median (range) gestational age during the last Doppler measurement before birth was 34.1 (19.6–39.1) weeks. The median UA-PI was 0.89 (−2.76 to 4.01), MCA-PI was −0.08 (−3.94 to 2.71), and CPR was −0.91 (−4.06 to 3.18). Median gestational age at birth was 39.1 (36.0–41.9) weeks. Five neonates had Apgar scores <6 at 5 min, of whom one had CNV, three had DNV, and one had BS at some point during the study period. Thirteen (18%) neonates underwent a Rashkind procedure during the study period. Eighty-two percent of the neonates were treated with Prostaglandin E1 because of (suspected) duct-dependent congenital heart disease (mean starting dosage 0.031 μg/kg/min and end dosage 0.023 ug/kg/min) and 46% of the neonates were treated with sedatives during the first 72 h after birth. Neonates received sedatives for various reasons such as Rashkind procedures and discomfort due to respiratory insufficiency by increased pulmonary flow, or due to Prostaglandin E1 treatment. The majority received single treatment with morphine (54%), followed by combined treatment with morphine and midazolam (29%). Two neonates (3%) received treatment with phenobarbitone due to agitation.
Various clinical characteristics and biochemical parameters were associated with aEEG background patterns (Table 2). On the basis of univariate regression analyses, we selected Apgar score at 5 min, treatment with sedatives, type of CHD, and treatment with Prostaglandin E1 for multiple logistic regression analysis. Only treatment with sedatives remained significant in the final model, which explained 56% of the variance (Table 3). We were unable to demonstrate a significant association between the type of sedative (morphine or midazolam) and aEEG background pattern (P=0.36). As only two neonates did not have SWC, it was not possible to assess the association between clinical characteristics or biochemical parameters and the occurrence of SWC. Onset of SWC was associated with sedatives (P<0.01), type of CHD (P=0.02), treatment with Prostaglandin E1 (P=0.01), abnormality on cranial ultrasound (P=0.02), and Apgar score at 5 min (rho −0.29, P=0.03). There were no clinical characteristics associated with epileptic activity.
Discussion
To our knowledge, this is the first study to describe aEEG background patterns, the presence and onset of SWC, and epileptic activity within the first 72 h after birth in neonates who were admitted to the NICU with prenatally diagnosed CHD. We demonstrate that 44% of these neonates have mildly abnormal aEEG background patterns that seem to be, at least partly, due to treatment with sedatives. We also demonstrate that almost all neonates with prenatally diagnosed CHD have SWC within 36 h after birth and 15% of the neonates have subclinical seizures within the first 72 h after birth. In comparison with other studies on aEEG in neonates with CHD, we observed a lower frequency of abnormal background patterns, fewer neonates with severely abnormal background patterns (4, 5, 14), and a higher frequency of SWC (5). The time of onset of SWC was similar to healthy full-term neonates (15). As we only included neonates with prenatally diagnosed CHD and assessed aEEG abnormalities within the first 72 h after birth, we speculate that prenatal diagnosis and immediate treatment in neonates with CHD might be successful in preventing hypoxic–ischemic events, and could contribute to the preservation of brain function immediately after birth (16).
In comparison with previous studies, we observed only subclinical seizures, rather than electroclinical seizures (4, 5, 17). In neonates with CHD following surgery, however, similar rates of subclinical seizures have been reported compared with our study (18). The clinical significance of subclinical seizures is currently unknown (19). On one hand, one might speculate that brain function is preserved in infants with prenatally diagnosed CHD due to immediate treatment following birth. On the other hand, seizure activity can be very subtle in neonates and therefore may have been missed. In infants with hypoxic–ischemic encephalopathy; for example, only 30% of high-risk neonates had a clinical correlate with electroencephalographic seizure activity (20).
Brain injury can be reflected by abnormal aEEG background patterns, absence of SWC, and seizure activity. Seizures by themselves can also independently contribute to the development of brain injury through various mechanisms such as impairment of energy metabolism and neuronal integrity (19). In neonates with hypoxic–ischemic encephalopathy, monitoring brain function using aEEG is considered to be one of the most accurate methods to predict NDO (21). Persistence of severely abnormal background patterns and time of onset of SWC >36 h after birth is particularly predictive for adverse NDO in these neonates (21, 22, 23). Whether aEEG monitoring before surgery in neonates with CHD is also predictive for NDO is yet to be determined. A few studies that reported on preoperative aEEG and NDO in neonates with CHD were unable to demonstrate this association (6, 7). NDO in infants with CHD might be the result of multiple events related to the treatment of CHD and the perioperative course. Longitudinal studies, starting from the prenatal period, are needed to clarify this theory.
In our study, abnormal aEEG background patterns were strongly associated with treatment with sedatives. Both morphine and midazolam have been known to cause a temporary depression of electrocortical activity for up to 24 h after administration of the drug (24, 25, 26). This has also been demonstrated in neonates with CHD after cardiopulmonary bypass. Treatment with midazolam had a transient (up to 2 h) effect on aEEG background patterns and was associated with a delayed onset of SWC (26). Because of these effects, some authors state that interpreting aEEG is unreliable in neonates who are treated with sedatives (26, 27). It might also be, however, that treatment with sedatives is harmful for the newborn brain.
Contrary to our hypothesis, we were unable to demonstrate an association between prenatal Doppler flow patterns and postnatal aEEG. Fetuses with CHD often have abnormal Doppler flow patterns indicative of blood flow in favor of brain perfusion (11, 12, 13). This phenomenon might occur in response to acute or chronic hypoxia, and has been associated with adverse neurodevelopmental outcome in neonates with intrauterine growth restriction (28). For this study, we selected the last available measurement before birth with a wide range in gestational ages. It might be that the association between prenatal Doppler flow patterns and postnatal aEEG is only present in neonates with prolonged abnormal prenatal Doppler flow patterns, or only after a certain gestational age. Alternatively, our sample size might have been too small to detect a significant difference.
It is known that hypoxemia has a less profound effect on aEEG compared with hypoperfusion (29). Taking this into account, we were surprised to find an association between cyanotic CHD and aEEG abnormalities in our univariate logistic regression analysis. Previous studies reported lower seizure burden (5) and less neurological abnormalities (30) in infants with cyanotic CHD before surgery in comparison with infants with acyanotic CHD. Data from our multiple regression analysis suggest that the association between the type of CHD and aEEG background patterns was due to an association between the use of sedatives and type of CHD.
There were inevitable limitations to our study. First, not all neonates who were diagnosed prenatally with CHD were monitored by aEEG within the first 72 h after birth. Furthermore, not all neonates were monitored by aEEG during the entire study period. This has increased the risk for selection bias. Second, we do not have information on NDO in these neonates. The clinical relevance of mildly abnormal aEEG background patterns and subclinical seizures is yet to be determined. Third, we included different types of CHD that might be associated with a different rate and type of aEEG abnormalities. Fourth, Doppler flow profiles were assessed at a wide range of gestational ages. This might have influenced the association between prenatal Doppler flow patterns and postnatal aEEG.
In conclusion, this study demonstrates that neonates with prenatally diagnosed CHD who were admitted to the NICU frequently have mildly abnormal aEEG background patterns and often have subclinical seizures within the first 72 h after birth. Abnormal aEEG background patterns seem to be, at least partly, because of the use of sedatives. Prenatal Doppler flow patterns do not seem to be associated with postnatal aEEG. Whether mildly abnormal background patterns and subclinical seizures are associated with NDO in neonates with prenatally diagnosed CHD who were admitted to the NICU is yet to be determined.
References
Marino BS, Lipkin PH, Newburger JW et al. Neurodevelopmental outcomes in children with congenital heart disease: evaluation and management: a scientific statement from the American Heart Association. Circulation 2012;126:1143–72.
Schaefer C, von Rhein M, Knirsch W et al. Neurodevelopmental outcome, psychological adjustment, and quality of life in adolescents with congenital heart disease. Dev Med Child Neurol 2013;55:1143–9.
Toet MC, Lemmers PM . Brain monitoring in neonates. Early Hum Dev 2009;85:77–84.
Mulkey SB, Yap VL, Bai S et al. Amplitude-integrated EEG in newborns with critical congenital heart disease predicts preoperative brain magnetic resonance imaging findings. Pediatr Neurol 2015;52:599–605.
Ter Horst HJ, Mud M, Roofthooft MT, Bos AF . Amplitude integrated electroencephalographic activity in neonates with congenital heart disease before surgery. Early Hum Dev 2010;86:759–64.
Gunn JK, Beca J, Penny DJ et al. Amplitude-integrated electroencephalography and brain injury in infants undergoing Norwood-type operations. Ann Thorac Surg 2012;93:170–6.
Gunn JK, Beca J, Hunt RW, Olischar M, Shekerdemian LS . Perioperative amplitude-integrated EEG and neurodevelopment in infants with congenital heart disease. Intensive Care Med 2012;38:1539–47.
Latal B, Wohlrab G, Brotschi B, Beck I, Knirsch W, Bernet V . Postoperative amplitude-integrated electroencephalography predicts four-year neurodevelopmental outcome in children with complex congenital heart disease. J Pediatr 2016;178:55–60.e1.
Friedman AH, Fahey JT . The transition from fetal to neonatal circulation: normal responses and implications for infants with heart disease. Semin Perinatol 1993;17:106–21.
Hillman NH, Kallapur SG, Jobe AH . Physiology of transition from intrauterine to extrauterine life. Clin Perinatol 2012;39:769–83.
Masoller N, Sanz-Cortes M, Crispi F et al. Severity of fetal brain abnormalities in congenital heart disease in relation to the main expected pattern of in utero brain blood supply. Fetal Diagn Ther 2016;39:269–78.
Masoller N, Martinez JM, Gomez O et al. Evidence of second-trimester changes in head biometry and brain perfusion in fetuses with congenital heart disease. Ultrasound Obstet Gynecol 2014;44:182–7.
Hahn E, Szwast A, Cnota J et al. Association between fetal growth, cerebral blood flow and neurodevelopmental outcome in univentricular fetuses. Ultrasound Obstet Gynecol 2016;47:460–5.
El-Naggar WI, Keyzers M, McNamara PJ . Role of amplitude-integrated electroencephalography in neonates with cardiovascular compromise. J Crit Care 2010;25:317–21.
de Vries LS, Toet MC . Amplitude integrated electroencephalography in the full-term newborn. Clin Perinatol 2006;33:619–32 vi.
Peyvandi S, De Santiago V, Chakkarapani E et al. Association of prenatal diagnosis of critical congenital heart disease with postnatal brain development and the risk of brain injury. JAMA Pediatr 2016;170:e154450.
Limperopoulos C, Majnemer A, Rosenblatt B et al. Association between electroencephalographic findings and neurologic status in infants with congenital heart defects. J Child Neurol 2001;16:471–6.
Naim MY, Gaynor JW, Chen J et al. Subclinical seizures identified by postoperative electroencephalographic monitoring are common after neonatal cardiac surgery. J Thorac Cardiovasc Surg 2015;150:169–80.
Pinchefsky EF, Hahn CD . Outcomes following electrographic seizures and electrographic status epilepticus in the pediatric and neonatal ICUs. Curr Opin Neurol 2017;30:156–64.
Murray DM, Boylan GB, Ali I, Ryan CA, Murphy BP, Connolly S . Defining the gap between electrographic seizure burden, clinical expression and staff recognition of neonatal seizures. Arch Dis Child Fetal Neonatal Ed 2008;93:F187–91.
van Laerhoven H, de Haan TR, Offringa M, Post B, van der Lee JH . Prognostic tests in term neonates with hypoxic-ischemic encephalopathy: a systematic review. Pediatrics 2013;131:88–98.
Osredkar D, Toet MC, van Rooij LG, Van Huffelen A, Groenendaal F, De Vries LS . Sleep–wake cycling on amplitude-integrated electroencephalography in term newborns with hypoxic-ischemic encephalopathy. Pediatrics 2005;115:327–332.
ter Horst HJ, Sommer C, Bergman KA, Fock JM, van Weerden TW, Bos AF . Prognostic significance of amplitude-integrated EEG during the first 72 h after birth in severely asphyxiated neonates. Pediatr Res 2004;55:1026–33.
Young GB, da Silva OP . Effects of morphine on the electroencephalograms of neonates: a prospective, observational study. Clin Neurophysiol 2000;111:1955–60.
Bell AH, Greisen G, Pryds O . Comparison of the effects of phenobarbitone and morphine administration on EEG activity in preterm babies. Acta Paediatr 1993;82:35–9.
Bernet V, Latal B, Natalucci G, Doell C, Ziegler A, Wohlrab G . Effect of sedation and analgesia on postoperative amplitude-integrated EEG in newborn cardiac patients. Pediatr Res 2010;67:650–5.
Olischar M, Davidson AJ, Lee KJ, Hunt RW . Effects of morphine and midazolam on sleep-wake cycling in amplitude-integrated electroencephalography in post-surgical neonates >/= 32 weeks of gestational age. Neonatology 2012;101:293–300.
Meher S, Hernandez-Andrade E, Basheer SN, Lees C . Impact of cerebral redistribution on neurodevelopmental outcome in small-for-gestational-age or growth-restricted babies: a systematic review. Ultrasound Obstet Gynecol 2015;46:398–404.
Bunt JEH, Gavilanes AWD, Reulen JPH, Blanco CE, Vles JSH . The influence of acute hypoxemia and hypovolemic hypotension of neuronal brain activity measured by the cerebral function monitor in newborn piglets. Neuropediatrics 1996;27:260–4.
Limperopoulos C, Majnemer A, Shevell MI, Rosenblatt B, Rohlicek C, Tchervenkov C . Neurologic status of newborns with congenital heart defects before open heart surgery. Pediatrics 1999;103:402–8.
Acknowledgements
This study was part of the research program of the Graduate School of Medical Sciences, Research Institutes BCN-BRAIN and GUIDE, and University of Groningen.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Statement of Financial Support
M.J.M. and S.J.K. were financially supported by the Junior Scientific Master Class of the University of Groningen.
Supplementary material is linked to the version of the paper at
Supplementary information
Rights and permissions
About this article
Cite this article
Mebius, M., Oostdijk, N., Kuik, S. et al. Amplitude-integrated electroencephalography during the first 72 h after birth in neonates diagnosed prenatally with congenital heart disease. Pediatr Res 83, 798–803 (2018). https://doi.org/10.1038/pr.2017.311
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pr.2017.311
This article is cited by
-
Continuous electroencephalography (cEEG) in infants with congenital heart disease (CHD)
Pediatric Research (2023)
-
CeRebrUm and CardIac Protection with ALlopurinol in Neonates with Critical Congenital Heart Disease Requiring Cardiac Surgery with Cardiopulmonary Bypass (CRUCIAL): study protocol of a phase III, randomized, quadruple-blinded, placebo-controlled, Dutch multicenter trial
Trials (2022)