Abstract
Background:
Recent experimental evidence suggests that gut microbiota may alter function within the nervous system providing new insight on the mechanism of neuropsychiatric disorders.
Methods:
Seventy-five infants who were randomized to receive Lactobacillus rhamnosus GG (ATCC 53103) or placebo during the first 6 mo of life were followed-up for 13 y. Gut microbiota was assessed at the age of 3 wk, 3, 6, 12, 18, 24 mo, and 13 y using fluorescein in situ hybridization (FISH) and qPCR, and indirectly by determining the blood group secretor type at the age of 13 y. The diagnoses of attention deficit hyperactivity disorder (ADHD) and Asperger syndrome (AS) by a child neurologist or psychiatrist were based on ICD-10 diagnostic criteria.
Results:
At the age of 13 y, ADHD or AS was diagnosed in 6/35 (17.1%) children in the placebo and none in the probiotic group (P = 0.008). The mean (SD) numbers of Bifidobacterium species bacteria in feces during the first 6 mo of life was lower in affected children 8.26 (1.24) log cells/g than in healthy children 9.12 (0.64) log cells/g; P = 0.03.
Conclusion:
Probiotic supplementation early in life may reduce the risk of neuropsychiatric disorder development later in childhood possible by mechanisms not limited to gut microbiota composition.
Similar content being viewed by others
Main
Psychiatric disorders are already ranked among the leading causes of disability in industrialized countries. With the current progressive increase in the incidence, they may be expected to assume the first place also globally within the next few years (1,2). Attention-deficit hyperactivity disorder (ADHD), characterized by inattention, impulsivity, and hyperactivity, affects three to seven percent of children worldwide (3,4). Moreover, symptoms of inattention and hyperactivity are frequent in children with Asperger syndrome (AS), which is characterized by stereotyped behavior and deficient social interaction and communication skills (5). Besides the common behavioral features, shared biological pathways and neuroanatomical links between these diseases have been reported (5,6).
Despite intensive research on ADHD and AS, the precise chain of pathological events underlying them remains unknown. The available data indicate ADHD and AS to be multifactorial disorders, in which genetic risk predominates, reinforced by various environmental and biological factors such as fetal stress, prematurity, toxins, and diet (7). Recently, the search for etiologies has been expanded both within the central nervous system and beyond. Experimental data are accumulating to suggest that the presence of gut microbiota as such, as compared with the absence of it, and especially its certain beneficial bacteria, probiotics, make for altered function within the nervous system (8,9,10,11,12). As a recent empirical study indicates (13) probiotics may provide a tool to manipulate brain activity even in humans.
To test the hypothetical involvement of the gut brain-axis in the manifestation of ADHD and AS, we analyzed the association of compositional development of the gut microbiota, the blood group secretor type as an indirect evidence of gut microbiota involvement, and the impact of specific probiotic intervention on the emergence of these two neuropsychiatric disorders in a cohort followed until 13 y of age.
Results
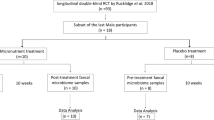
The 13-y follow-up was completed by 75 of the study subjects; 40 (53.3%) of these initially received Lactobacillus rhamnosus GG and 35 (46.7%) placebo ( Figure 1 ). Clinical characteristics of the study population were comparable between the intervention groups ( Table 1 ). Furthermore, all the clinical characteristics, except for length of exclusive breast-feeding, were comparable between the children who did and did not complete the 13-y follow-up ( Table 2 ).
Flow chart of the procedure.
ADHD was diagnosed in three (4.0%) children, AS in one (1.3%) child, and both ADHD and AS in two (2.7%) by the age of 13 y. Of these six children, four were on methylphenidate and two on daily ω-3 fatty acid supplementation. All the children with neuropsychiatric diagnoses were male. ADHD or AS had been diagnosed in 6/35 (17.1%) children in the placebo and none in the probiotic group by the age of 13 y (P = 0.008). When logistic regression analysis was controlled for gender, the result remained significant (P = 0.02). All the other clinical characteristics, as shown in Table 3 , were comparable between healthy children and those with ADHD or AS. Two children affected by AS were nonsecretors (33% of AS/ADHD children), while in nonaffected children this was the case in 12% (P = 0.20).
During the first 3 mo of life, the differences in gut microbiota composition between children who later developed ADHD or AS compared to healthy children were not manifested in bacterial numbers as analyzed by FISH ( Figure 2 ), but in species composition of Bifidobacterium, assessed by qPCR. The distinction was due to significantly lower median (IQR) numbers of Bifidobacterium longum at the age of 3 mo among the children with neuropsychiatric disorders than in healthy children (4.35 (3.99–10.40) log/g and 10.18 (8.88–10.88) log/g, respectively, P = 0.045).
The gut microbiota composition (mean (95% CI) log cells/g) development analyzed by FISH during the first 13 y of life among children with (circle) and without (diamond) neuropsychiatric disorders. a, Bifidobacterium; b, Clostridium histolyticum; c, Bacteroides-Prevotella; d, Lactobacillus-Enterococcus. Number of fecal samples analyzed in children with neuropsychiatric disorders: n = 2 at 0.5 mo, n = 4 at 3 mo, n = 5 at 6, 12, and 18 mo, n = 3 at 24 mo, n = 4 at 13 y. Number of samples analyzed in healthy children: n = 19 at 0.5 mo, n = 41 at 3 mo, n = 53 at 6 mo, n = 59 at 12 mo, n = 55 at 18 mo, n = 48 at 24 mo, n = 53 at 13 y.
At the age of 6 mo, at weaning, when probiotic intervention was completed, the mean (SD) number of cells belonging to the genus Bifidobacterium as measured with FISH was significantly lower among children with neuropsychiatric disorders than in those without (8.26 (1.24) log cells/g vs. 9.12 (0.64) log cells/g, respectively, P = 0.03). At the age of 18 mo, the mean (SD) numbers of Bacteroides and Lactobacillus-Enterococcus group bacteria were lower among children with ADHD/AS than healthy children (7.28 (0.85) log cells/g vs. 8.13 (0.51) log cells/g, P = 0.008; 7.71 (0.78) log cells/g vs. 8.40 (0.40) log cells/g, P = 0.01, respectively ( Figure 2 ). Furthermore, at the age of 24 mo, the mean (SD) numbers of cells belonging to the Clostridium histolyticum group were lower among children with ADHD or AS than in healthy children, (7.46 (0.44) log cells/g vs. 8.16 (0.55) log cells/g; P = 0.04).
At the age of 13, there were no statistically significant differences in gut microbiota composition, analyzed with FISH or qPCR, between children with or without neuropsychiatric disorders ( Figure 2 ).
Discussion
The results of our preliminary study demonstrate for the first time that specific probiotics may reduce the risk of the development of ADHD and AS possibly by mechanisms not directly associated with gut microbiota composition, since no single constant microbiota composition or their difference could be distinctive in children with or without neuropsychiatric disorders.
These findings must be viewed in the light of some caution as preliminary and initial observation into this novel area. First, our probiotic intervention study was originally designed and statistically powered for prevention of atopic eczema, not for prevention of more uncommon neuropsychiatric disorders ADHD and AS. Regardless of that, it is interesting to notice that allergic disease has been shown to be associated with an increased risk of ADHD and differences in neurodevelopment (14,15) suggesting a possibility for common environmental determinants. Second, the number of drop-outs was quite considerable during the follow-up. Therefore, we cannot discount the possibility that the issue would not have biased our findings. However the number of drop-outs was equally divided in the both intervention groups and base-line characteristics of the drop-outs and the study finishers were similar, except for the duration of exclusive breast-feeding, demonstrating that these two groups were unbiased in almost all of the known factors. On the other hand, the careful and prospective 13-y follow-up period is the strength of the study. A further strength of the study is that gut microbiota has been analyzed comprehensively by FISH and qPCR as well as indirectly by blood group secretor type analysis.
ADHD is a disease of substantial genetic predisposition, as suggested by a number of adoption, twins, and family studies (7,16,17). This notwithstanding, recent studies document up to 20% discordance in identical twins, leaving room for environmental and epigenetic determinants of ADHD. Thus far, prenatal exposure to maternal smoking (18), low birth weight, prematurity (19,20), and specific environmental exposures such as lead and organic pollutants (7) have been linked to ADHD. In contrast, research on dietary factors such as sugar, artificial food colorings, zinc, iron, and ω-3 fatty acids, has established only vague causality in ADHD (7), despite some hints of their therapeutic potential (21,22). An empirical elimination diet has indeed been shown to be effective in the treatment of ADHD (22). Moreover, breastfeeding, as a potent inducer of Bifidobacteria in the gut microbiota (23), has proved to be associated with lower levels of conduct disorder symptoms in middle childhood, which is in line with our findings in this study (24) demonstrating defiencies in early Bifidobacterium composition in those with later ADHD or AS.
Although the precise possible mechanisms of action of the gut microbiota in the gut-brain axis are unclear, a previous experimental study offers an interesting clue. In the study in question, mice with experimentally induced chronic colitis showed anxiety-like behavior. Treatment with Bifidobacterium longum abolished such behavior. However, the anxiolytic effect of Bifidobacterium longum was absent in vagotomized mice, suggesting that the effect was transmitted to the central nervous system by activating vagal pathways at the level of the enteric nervous system (9). It is thus intriguing to note that in our study the amount of the same species was found to be decreased in early life in children later developing ADHD or AS.
Our data demonstrate that early administration of Lactobacillus rhamnosus GG may reduce the risk of ADHD and AS. Lactobacillus rhamnosus GG has been shown to stabilize the gut permeability barrier by effects on tight junctions, mucin production and antigen-specific immunoglobulin A production (25). In addition, a recent experimental study has demonstrated that Lactobacillus rhamnosus regulates, again via the vagus nerve, emotional behavior and the central GABAergic system, which is also associated with neuropsychiatric disorders (11,26). Of note, a recent study with healthy women demonstrated that a consumption of a mixture of probiotic bacteria had significant effects on brain regions that control central processing of emotion and sensation (13). Probiotics had no significant effect on microbiota composition in the study suggesting that the effects on central nervous system were either induced by altered vagal afferent signaling or by systemic metabolic changes related to probiotic intake (13). Furthermore, another experimental study with mice showed that pretreatment with probiotic Lactobacillus rhamnosus prevents learning and memory dysfunction in Citrobacter rodentium-infected mice (27). It thus remains to be elucidated whether these effects of Lactobacillus rhamnosus are of importance in the development of neuropsychiatric disorders in humans. However, in accord with our clinical findings here it has been postulated that neural pathways may alter already early in development. If such an alteration takes place at a critical moment, the sequential dysfunction of the gut-brain axis may become relatively constant into adulthood (28,29).
Our findings demonstrate a possible preventive risk reducing effect of a probiotic LGG on later development of ADHD and AS. We also report an interconnection between the early gut microbiota and development of these neuropsychiatric disorders, although no single constant microbiota composition component or change was detected. However, keeping in mind the above-mentioned limitations of the study, we consider the findings preliminary but encouraging for further studies of the subject both in the area of well-powered clinical trials and experimental research.
Methods
Ethics Statement
The study was approved by the committees on ethical practice in Turku University Hospital and the Health Office of Turku. Written informed consent was obtained from the children and their parents.
Study Population and Design
The subjects included in this study are participants in an ongoing, randomized, double-blind, placebo-controlled prospective follow-up study involving perinatal Lactobacillus rhamnosus GG (ATCC 53103) intervention (http://www.clinicaltrials.gov/ct/gui/show/NCT00167700) as described in detail elsewhere (30,31). In brief, the original study population comprised 159 infants who had at least one family member with allergic disease. The mothers of these children were recruited in the antenatal clinics of the city of Turku between February 1997 and January 1998 and randomized in double-blind, placebo-controlled manner to receive 1x1010colony-forming units of Lactobacillus rhamnosus GG or placebo (microcrystalline cellulose) daily for 4 wk before expected delivery. After delivery, the capsule contents were given either to the children, or continuously to the mothers, if breast-feeding, for 6 mo.
Clinical Evaluation
All the 132 children (83% of the original cohort) completing the 2-y follow-up were invited to the 13-y follow-up visit. During the study visit between June 2011 and October 2011, a clinical examination was carried out by AP. Weight, height, mid-upper arm and waist circumference, and blood pressure were measured. Data were prospectively collected by the parents throughout the follow-up on breast-feeding, acute and chronic conditions, antibiotics and other medications, and probiotics and other diet. Data were also collected from the records of well-baby clinics and hospital.
Assessment of Early Behavior Patterns
During the 7th and 12th week of life parents recorded their infants’ behavior patterns (sleeping time, time awake and content, fussing, colic-type cry, other cry, and feeding), vomits, stools with their consistency, and skin condition, using a modified 24-h Barr chart (32) on 7 consecutive days (31,32). Fussing was defined as a state of irritability, “not quite crying but not awake and content”. Other cry was defined as crying responsive to intervention (feeding, diaper change, carrying, sucking a pacifier), and colic-type cry as crying not responsive to such intervention. Total distress, the amount of crying and fussing, was the sum of these various modes as reported by the parents.
Assessment of Neuropsychiatric Disorders
The clinical diagnosis of ADHD and AS was made by an experienced child psychiatrist or neurologist not involved in the study or follow-up and blinded with randomization. The diagnostic criteria of the International Classification of Diseases (ICD-10) were used. Accordingly, a person with ADHD must have at least six of the listed symptoms of inattention and at least three of the listed symptoms of hyperactivity and impulsivity, these having persisted for at least 6 mo to a degree that is maladaptive and inconsistent with the person’s development level. The symptoms must have begun before the age of 7 y, must present in at least two places, negatively affect school, social or occupational functioning, and are not better explained by an alternative disorder. According to the ICD-10, a person with Asperger syndrome shows no clinically significant general delay in spoken or receptive language or cognitive development but has qualitative abnormalities in reciprocal social interaction and unusually intense circumscribed interest or restrictive, repetitive and stereotyped patterns of behavior, interests, and activities. The disorder is not attributable to other varieties of pervasive developmental disorder.
Fecal Samples
A fecal sample was taken at 3 wk, 3, 6, 12, 18, 24 mo, and 13 y either by nursing staff at a scheduled visit or by the parents/children immediately before the visit. In the latter case, the sample was stored at 4 °C and delivered to the hospital within 24 h.
DNA Isolation and qPCR Assay
Fecal samples were stored at −80 °C until analyzed. They were pretreated and DNA was extracted using an automated KingFisher DNA extraction system (Thermo Fisher Scientific Oy, Vantaa, Finland) and InviMag Stool DNA kit (Stratec Molecular, Berlin, Germany), as previously described (33). The standard DNA for quantitative PCR (qPCR) was prepared as described elsewhere (33). All DNA samples were stored at −20 °C until analyzed. Quantitative PCRs were conducted as previously described (34,35). PCR amplification and detection were performed with an ABI PRISM 7300-PCR sequence detection system (Applied Biosystems, Foster City, CA).
FISH Assay
Bacterial cells were harvested and fixed and FISH carried out with fluorophore (indocarbocyanine Cy3)-labeled oligonucleotide probes, as previously described (36). Total cell numbers were determined by nucleic acid stain 4′, 6-diamidino-2-phenylindole (DAPI). Cells were counted visually using an Olympus SZX9 epifluorescence microscope (Olympus Optical Co LTD, Tokyo, Japan).
Blood Group Secretor Status
At the age of 13 y, histo-ABO blood group secretor status was determined by genotyping a single-nucleotide polymorphism (SNP) rs601338 (W143X, G428A) of FUT2 gene. Nonsecretor individuals have a nonsense mutation in rs601338 (genotype AA) leading to unfunctional fucosyltransferase 2 enzyme, and therefore the non-secretors do not express histo-ABO blood group antigens in intestinal mucosa or other secretions. The SNP rs601338 determines the nonsecretor status in Finnish population (37). The SNP rs601338 was genotyped by using Taqman chemistry (Life Technologies, Carlsbad, CA) (fwd primer: GGGAGTACGTCCGCTTCAC, rev primer: TGGCGGAGGTGGTGGTA, reporter 1-VIC: CTGCTCCTAGACCTT, reporter 2-FAM: CTGCTCCTGGACCTT) on LightCycler 480 Real-Time PCR Instrument (Roche, Switzerland) in the Institute for Molecular Medicine Finland (FIMM) as previously described (38).
Statistical Methods
The clinical characteristics of the study subjects for continuous variable are given as mean values with SD, and as a number and proportions for categorical variables. Comparisons between two continuous variables were made using t-test or Mann–Whitney U-test, as appropriate. Comparisons between two categorical variables were made using χ2 test or Fisher’s exact test, as appropriate. Univariate associations between the response variable (neuropsychiatric disorders) and continuous predictor variables were studied using logistic regression analysis. In all PCR assays, except with Bifidobacterium genus and Bifidobacterium longum, a large proportion of observations were below the detection limit. These observations were dichotomized as either above or below the detection limit for statistical analyses. In all other microbial assays, the proportion of observations below detection limit was at most seven percent. These observations were excluded from statistical analyses. Statistical analyses were made using SAS for Windows version 9.2. P values below 0.05 were considered statistically significant.
Statement of Financial Support
The study was supported by the Foundation for Pediatric Research and an EVO-grant (the Research Funds from Specified Government Transfers) from Turku University Hospital.
Disclosure
There is no competing interest related to this manuscript.
References
Ustün TB, Ayuso-Mateos JL, Chatterji S, Mathers C, Murray CJ. Global burden of depressive disorders in the year 2000. Br J Psychiatry 2004;184:386–92.
World Health Organization The world health report 2001: mental health: new understanding, new hope. 2001. http://www.who.int/whr/2001/en/whr01_ch1_en.pdf?ua=1.
Forsythe P, Sudo N, Dinan T, Taylor VH, Bienenstock J. Mood and gut feelings. Brain Behav Immun 2010;24:9–16.
American Psychiatric Association: Diagnostic and Statistical Manual of Mental Disorders. 4th edn. Washington, DC: American Psychiatric Association, 2000.
Gargaro BA, Rinehart NJ, Bradshaw JL, Tonge BJ, Sheppard DM. Autism and ADHD: how far have we come in the comorbidity debate? Neurosci Biobehav Rev 2011;35:1081–8.
Pasini A, D’Agati E, Pitzianti M, Casarelli L, Curatolo P. Motor examination in children with Attention-deficit/hyperactivity Disorder and Asperger Syndrome. Acta Paediatr 2012;101:e15–8.
Thapar A, Cooper M, Jefferies R, Stergiakouli E. What causes attention deficit hyperactivity disorder? Arch Dis Child 2012;97:260–5.
Bercik P, Verdu EF, Foster JA, et al. Chronic gastrointestinal inflammation induces anxiety-like behavior and alters central nervous system biochemistry in mice. Gastroenterology 2010;139:2102–2112.e1.
Bercik P, Park AJ, Sinclair D, et al. The anxiolytic effect of Bifidobacterium longum NCC3001 involves vagal pathways for gut-brain communication. Neurogastroenterol Motil 2011;23:1132–9.
Neufeld KM, Kang N, Bienenstock J, Foster JA. Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol Motil 2011;23:255–64, e119.
Bravo JA, Forsythe P, Chew MV, et al. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc Natl Acad Sci USA 2011;108:16050–5.
Messaoudi M, Lalonde R, Violle N, et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br J Nutr 2011;105:755–64.
Tillisch K, Labus J, Kilpatrick L, et al. Consumption of fermented milk product with probiotic modulates brain activity. Gastroenterology 2013;144:1394–401, 1401.e1–4.
Tsai MC, Lin HK, Lin CH, Fu LS. Prevalence of attention deficit/hyperactivity disorder in pediatric allergic rhinitis: a nationwide population-based study. Allergy Asthma Proc 2011;32:41–6.
Meldrum SJ, D’Vaz N, Dunstan JA, et al. Allergic disease in the first year of life is associated with differences in subsequent neurodevelopment and behaviour. Early Hum Dev 2012;88:567–73.
Sprich S, Biederman J, Crawford MH, Mundy E, Faraone SV. Adoptive and biological families of children and adolescents with ADHD. J Am Acad Child Adolesc Psychiatry 2000;39:1432–7.
Lichtenstein P, Carlström E, Råstam M, Gillberg C, Anckarsäter H. The genetics of autism spectrum disorders and related neuropsychiatric disorders in childhood. Am J Psychiatry 2010;167:1357–63.
Langley K, Rice F, van den Bree MB, Thapar A. Maternal smoking during pregnancy as an environmental risk factor for attention deficit hyperactivity disorder behaviour. A review. Minerva Pediatr 2005;57:359–71.
Bhutta AT, Cleves MA, Casey PH, Cradock MM, Anand KJ. Cognitive and behavioral outcomes of school-aged children who were born preterm: a meta-analysis. JAMA 2002;288:728–37.
Aarnoudse-Moens CS, Weisglas-Kuperus N, van Goudoever JB, Oosterlaan J. Meta-analysis of neurobehavioral outcomes in very preterm and/or very low birth weight children. Pediatrics 2009;124:717–28.
Pelsser LM, Frankena K, Toorman J, Savelkoul HF, Pereira RR, Buitelaar JK. A randomised controlled trial into the effects of food on ADHD. Eur Child Adolesc Psychiatry 2009;18:12–9.
Pelsser LM, Frankena K, Toorman J, et al. Effects of a restricted elimination diet on the behaviour of children with attention-deficit hyperactivity disorder (INCA study): a randomised controlled trial. Lancet 2011;377:494–503.
Harmsen HJ, Wildeboer-Veloo AC, Raangs GC, et al. Analysis of intestinal flora development in breast-fed and formula-fed infants by using molecular identification and detection methods. J Pediatr Gastroenterol Nutr 2000;30:61–7.
Shelton KH, Collishaw S, Rice FJ, Harold GT, Thapar A. Using a genetically informative design to examine the relationship between breastfeeding and childhood conduct problems. Eur Child Adolesc Psychiatry 2011;20:571–9.
Isolauri E, Kalliomäki M, Laitinen K, Salminen S. Modulation of the maturing gut barrier and microbiota: a novel target in allergic disease. Curr Pharm Des 2008;14:1368–75.
Enticott PG, Rinehart NJ, Tonge BJ, Bradshaw JL, Fitzgerald PB. A preliminary transcranial magnetic stimulation study of cortical inhibition and excitability in high-functioning autism and Asperger disorder. Dev Med Child Neurol 2010;52:e179–83.
Gareau MG, Wine E, Rodrigues DM, et al. Bacterial infection causes stress-induced memory dysfunction in mice. Gut 2011;60:307–17.
Cryan JF, O’Mahony SM. The microbiome-gut-brain axis: from bowel to behavior. Neurogastroenterol Motil 2011;23:187–92.
Neufeld KA, Kang N, Bienenstock J, Foster JA. Effects of intestinal microbiota on anxiety-like behavior. Commun Integr Biol 2011;4:492–4.
Kalliomäki M, Salminen S, Arvilommi H, Kero P, Koskinen P, Isolauri E. Probiotics in primary prevention of atopic disease: a randomised placebo-controlled trial. Lancet 2001;357:1076–9.
Kalliomäki M, Laippala P, Korvenranta H, Kero P, Isolauri E. Extent of fussing and colic type crying preceding atopic disease. Arch Dis Child 2001;84:349–50.
Barr RG, Kramer MS, Boisjoly C, McVey-White L, Pless IB. Parental diary of infant cry and fuss behaviour. Arch Dis Child 1988;63:380–7.
Nylund L, Heilig HG, Salminen S, de Vos WM, Satokari R. Semi-automated extraction of microbial DNA from feces for qPCR and phylogenetic microarray analysis. J Microbiol Methods 2010;83:231–5.
Collado MC, Isolauri E, Laitinen K, Salminen S. Distinct composition of gut microbiota during pregnancy in overweight and normal-weight women. Am J Clin Nutr 2008;88:894–9.
Scalabrin DM, Mitmesser SH, Welling GW, et al. New prebiotic blend of polydextrose and galacto-oligosaccharides has a bifidogenic effect in young infants. J Pediatr Gastroenterol Nutr 2012;54:343–52.
Kalliomäki M, Kirjavainen P, Eerola E, Kero P, Salminen S, Isolauri E. Distinct patterns of neonatal gut microflora in infants in whom atopy was and was not developing. J Allergy Clin Immunol 2001;107:129–34.
Wacklin P, Mäkivuokko H, Alakulppi N, et al. Secretor genotype (FUT2 gene) is strongly associated with the composition of Bifidobacteria in the human intestine. PLoS One 2011;6:e20113.
Parmar AS, Alakulppi N, Paavola-Sakki P, et al. Association study of FUT2 (rs601338) with celiac disease and inflammatory bowel disease in the Finnish population. Tissue Antigens 2012;80:488–93.
Acknowledgements
We thank the families participating in our study, Jaakko Matomäki, for statistical consultation, Robert MacGilleon, for language review of the manuscript, and Johanna Hvitfelt-Koskelainen for help with the follow-up of participants.
Author information
Authors and Affiliations
Corresponding author
PowerPoint slides
Rights and permissions
About this article
Cite this article
Pärtty, A., Kalliomäki, M., Wacklin, P. et al. A possible link between early probiotic intervention and the risk of neuropsychiatric disorders later in childhood: a randomized trial. Pediatr Res 77, 823–828 (2015). https://doi.org/10.1038/pr.2015.51
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pr.2015.51
This article is cited by
-
Gut Microbiota Modulation and Its Implications on Neuropathic Pain: A Comprehensive Literature Review
Pain and Therapy (2024)
-
Assessment of probiotic strain Lactobacillus acidophilus LB supplementation as adjunctive management of attention-deficit hyperactivity disorder in children and adolescents: a randomized controlled clinical trial
BMC Psychiatry (2023)
-
Altered Gut Microbiota and Short-chain Fatty Acids in Chinese Children with Constipated Autism Spectrum Disorder
Scientific Reports (2023)
-
Early-life gut microbiota and attention deficit hyperactivity disorder in preadolescents
Pediatric Research (2023)
-
Maternal obesity, gestational diabetes mellitus, and diet in association with neurodevelopment of 2-year-old children
Pediatric Research (2023)