Abstract
Background:
Programming of the hypothalamic-pituitary-adrenal (HPA) axis possibly explains the relation between intrauterine growth restriction (IUGR) and/or preterm birth and elevated blood pressure in later life. Very-low-birth-weight infants (birth weight <1,500 g) have high prevalence of raised blood pressure, already in early childhood. We investigated cortisol levels, relation to blood pressure and reliability of salivary cortisol in infancy and early childhood in very-low-birth-weight infants.
Methods:
We included 41 children, participating in the randomized controlled Neonatal Insulin Replacement Therapy in Europe (NIRTURE) trial. Serum and salivary samples for cortisol measurement (immunoassay) were taken simultaneously at 6 mo and separately at 2 y corrected age. Blood pressure was measured at 2 y corrected age.
Results:
Serum cortisol was significantly correlated to systolic and diastolic blood pressure in boys and in the early-insulin treated group. At 2 y corrected age serum cortisol was significantly higher in the early-insulin group compared to the standard care group. At 6 mo corrected age salivary cortisol was significantly correlated to serum cortisol.
Conclusion:
In very-low-birth-weight boys, the positive correlation between cortisol and blood pressure is present at 2 y corrected age. Early insulin therapy could affect programming of the HPA axis. Salivary cortisol mirrors serum levels at 6 mo corrected age.
Similar content being viewed by others
Main
The inverse relation between birth weight and blood pressure, and consequently the importance of fetal growth for later blood pressure, was first indicated by Barker et al. (1,2) and confirmed in many studies in children and adults, reviewed by Huxley et al. (3). Preterm born infants have impaired growth in early postnatal life, in a period comparable to the third trimester of pregnancy, and also have a higher blood pressure in later life (4).
One of the proposed mechanism underlying the association between IUGR and/or preterm birth and blood pressure is programming of the HPA axis. In adults birth weight is inversely associated with cortisol levels and cortisol levels are positively correlated to blood pressure (5,6). This was also shown in children between the ages of 4.9 and 15.5 y and born at a gestational age >32 wk (7). In preterm born young adult men, cortisol is also associated with high systolic blood pressure (8). There are no data about the association between cortisol and blood pressure in preterm born infants <32 wk in early childhood. We recently showed that at the corrected age of 2 y very-low-birth-weight (VLBW) infants (birth weight <1,500 g) have a high prevalence of raised blood pressure (systolic and/or diastolic ≥ 90th percentile for age, sex, and height) (9). Elevated blood pressure (compared to published reference standards) in early childhood in VLBW infants was also reported by Duncan et al. (10).
The aim of the present study was to measure cortisol levels in VLBW infants at 6 mo and 2 y corrected age and correlate cortisol levels at 2 y corrected age to blood pressure. Our second aim was to investigate the reliability of salivary cortisol measurements in this population. As the subjects were part of the Neonatal Insulin Replacement Therapy in Europe (NIRTURE) trial, our third aim was to evaluate the effect of early insulin therapy on salivary and serum cortisol levels at 6 mo and 2 y corrected age. The blood pressure values were presented earlier, as part of the evaluation of all the metabolic syndrome components in this population (9). However, the emphasis of the present study is on cortisol, programming of the HPA axis and cortisol measurement in saliva compared to serum.
Results
In our neonatal intensive care unit, 47 VLBW infants participated in the NIRTURE trial. Five infants died. One child was excluded because parents refused blood and salivary sampling at 6 mo corrected age; at 2 y corrected age this child was lost to follow-up. Forty-one children were included in the present study. Table 1 shows the characteristics and outcome of these children. Seventeen infants (9 male/8 female) were assigned to the early-insulin group and 24 infants (12 male/12 female) received standard neonatal care. In the standard care group, six infants were treated with insulin for 1 or 2 d because of hyperglycemia due to sepsis. Table 2 shows serum and salivary cortisol at 6 mo and 2 y corrected age and blood pressure at 2 y corrected age with the group divided in male and female children and divided in early-insulin therapy and standard care. At 2 y corrected age serum cortisol was significantly higher in children treated with insulin compared to children in the standard care group. Paired-samples t-test showed no differences between serum cortisol levels at 6 mo and 2 y corrected age (both taken in the afternoon).
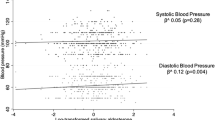
At 2 y corrected age serum cortisol (taken at the visit to the outpatient clinic early in the afternoon) was significantly correlated to both systolic blood pressure and diastolic blood pressure in boys (r = 0.79; P < 0.001 and r = 0.65; P = 0.004 resp.), but not in girls (r = 0.39; P = 0.15 and r = 0.47; P = 0.08 resp.) and in the early-insulin group (r = 0.80; P = 0.001 and r = 0.68; P = 0.008 resp.), but not in the standard care group (r = 0.33; P = 0.19 and r = 0.36; P = 0.14 resp.). These correlations are shown in Figure 1 . There was no correlation between salivary cortisol (taken at home early in the morning) and blood pressure.
Correlation between blood pressure (BP) and serum cortisol at 2 y corrected age with the group of very-low-birth-weight children divided in boys (•) and girls (▪) (a for systolic BP and c for diastolic BP) and divided in treatment group (standard care (○) and early-insulin therapy (□)) (b for systolic BP and d for diastolic BP).
Birth weight was not correlated to serum or salivary cortisol at any age or to blood pressure at 2 y corrected age.
At 6 mo corrected age salivary cortisol was significantly correlated to serum cortisol (r = 0.62; P = 0.001).
Discussion
The present study shows that in subgroups of VLBW infants the positive correlation between cortisol and blood pressure can be demonstrated as early as at 2 y corrected age. At 2 y corrected age insulin-treated children have higher serum cortisol levels than children in the standard care group. Salivary cortisol measurement is reliable in 6 mo-old-VLBW children.
The positive correlation between serum cortisol levels and blood pressure has been shown before in adults and older children (5,6,7). The present study confirms this association in early childhood in VLBW infants and supports the hypothesis that programming of the HPA axis may contribute to the high prevalence of raised blood pressure in this population (9,10). The correlation between cortisol and both systolic and diastolic blood pressure was significant only in boys. This is in accordance with the study of Szathmári et al. (8) in preterm born young adults, showing an association between cortisol and high systolic blood pressure only in men. In older children (between the ages of 4.9 and 15.5 y), including preterm born children >32 wk gestation, the association between cortisol level and blood pressure was not different between boys and girls (7). The age groups, which show the sex difference in this association (adulthood and early childhood) correspond to periods that the hypothalamic-pituitary-gonadal axis is active, as this is also active in the postnatal period (11,12).
VLBW children treated with insulin in the first postnatal week have higher serum cortisol levels at 2 y corrected age than children treated with standard care. In the insulin treated group there also was a significant correlation between serum cortisol levels and blood pressure (systolic and diastolic). These results suggest that early insulin treatment may affect the programming of the HPA axis, although the study population was small. Animal studies in offspring of diabetic mothers show that increased insulin concentrations within the immature hypothalamus may lead to irreversible malprogramming (with morphological changes) of regulation centres for metabolism and body weight (13). Proposed mechanism for the positive relation between increased HPA axis activity and blood pressure are reduced insulin sensitivity and activation of the central sympathetic nervous system (14).
The absence of a significant correlation between salivary cortisol and blood pressure at 2 y corrected age could be caused by the small population of our study. On the other hand, the fact that only afternoon (serum) and not morning (salivary) cortisol levels were correlated to blood pressure could also indicate that the failure to suppress cortisol during the day, resulting in sustained hypercortisolism, is related to elevated blood pressure.
In this study, we found a significant correlation between salivary and serum cortisol levels in VLBW infants at 6 mo corrected age. This finding confirms the reliability of salivary cortisol measurements shown before in premature infants (15) and older children (16). We did not find the inverse relation between birth weight and cortisol levels as shown in other studies in children (7,17).
Our study is limited by the small number of infants and the lack of data from term born children. The correlations between cortisol and blood pressure need to be confirmed in a larger group of VLBW infants. Cortisol levels and blood pressure have to be compared with those of term born children from our own population.
In conclusion, in VLBW boys, the positive correlation between cortisol and blood pressure is already present at 2 y corrected age, suggesting that programming of the HPA axis could contribute to the high prevalence of raised blood pressure in VLBW infants in early childhood. Early insulin treatment could affect this programming, resulting in higher cortisol levels. Salivary cortisol mirrors serum levels at 6 mo corrected age and has an important advantage as noninvasive method, especially in children.
Methods
Study Population
The subjects were part of the NIRTURE trial, an international multicenter randomized controlled trial investigating the role of early insulin therapy in VLBW infants (18). After written informed consent was obtained from the parents VLBW infants younger than 24 h of age and requiring intensive care were randomized to receive continuous intravenous infusion of insulin for the first 7 d of life or standard neonatal care with insulin treatment only in case of hyperglycemia. Exclusion criteria included maternal diabetes and major congenital anomalies. All 47 infants participating in the NIRTURE trial in our neonatal intensive care unit were eligible for the present study. Therefore the sample size of the present study was determined by the number of infants we included in the NIRTURE trial. After discharge, all 42 surviving infants were followed in the outpatient clinic with visits at expected date of delivery and at the corrected ages of 3 mo, 6 mo, 1 y, and 2 y. Approval from the ethics committee of the VU University Medical Center was obtained.
Data Collection
At every visit to the outpatient clinic body weight was measured using an electronic scale to the nearest 0.1 kg, length and headcircumference were measured to the nearest 0.1 cm and BMI was calculated. Standard deviation scores of weight, length, and BMI were calculated according to Dutch references (19,20). At 2 y corrected age blood pressure was measured in the calm state using an appropriately sized cuff and automated blood pressure measuring device (Dinamap, Critikon, Tampa, Florida); the mean value of two measurements was used for analysis and measurements in noncalm state were excluded. At 6 mo and 2 y corrected age blood and salivary samples were taken for measurement of cortisol. We only took blood samples at two of the visits to the outpatient clinic to limit the burden of blood sampling for the children. Saliva was collected by suction using a saliva aspiration set. At 6 mo corrected age this was performed by the research nurse in the outpatient clinic early in the afternoon and just before the blood sample was taken. At 2 y corrected age saliva was collected by the parents at home immediately after awakening in the morning; the blood sample was taken early in the afternoon. As the saliva sample at 2 y corrected age was the only sample taken in the morning, comparison of 2-y salivary cortisol to the other cortisol values, all taken in the afternoon, was not possible.
Assays
Serum cortisol was measured by competitive immunoassay (Advia Centaur, Siemens Medical Solutions Diagnostics, Malvern, PA). Lower limit of quantitation is 30 nmol/l, intra-assay coefficient of variation is 3% at 700 nmol/l, and interassay coefficient of variation is 6% at both 150 and 500 nmol/l and 8% at 1000 nmol/l.
Salivary cortisol was measured by automated immunoassay (Architect i2000, Abbott, North Chicago, IL). This assay is described in detail by Heijboer et al. (21).
Statistical Analysis
Statistical analyses were performed using the Statistical Package of Social Sciences software for Microsoft Windows version 19 (SPSS, Chicago, IL). Differences between subgroups were evaluated using Student’s t-test for normally distributed values and Mann–Whitney test and Wilcoxon signed-rank test for not normally distributed values. Bivariate correlation analysis was performed to study the relations between birth weight, cortisol concentrations and blood pressure and between serum and salivary cortisol concentrations. P values < 0.05 were considered as significant.
Statement of Financial Support
No financial support was provided for this study.
Disclosure
The authors have no conflicts of interest to disclose.
References
Barker DJ, Osmond C, Golding J, Kuh D, Wadsworth ME. Growth in utero, blood pressure in childhood and adult life, and mortality from cardiovascular disease. BMJ 1989;298:564–7.
Barker DJ, Bull AR, Osmond C, Simmonds SJ. Fetal and placental size and risk of hypertension in adult life. BMJ 1990;301:259–62.
Huxley RR, Shiell AW, Law CM. The role of size at birth and postnatal catch-up growth in determining systolic blood pressure: a systematic review of the literature. J Hypertens 2000;18:815–31.
de Jong F, Monuteaux MC, van Elburg RM, Gillman MW, Belfort MB. Systematic review and meta-analysis of preterm birth and later systolic blood pressure. Hypertension 2012;59:226–34.
Phillips DI, Barker DJ, Fall CH, et al. Elevated plasma cortisol concentrations: a link between low birth weight and the insulin resistance syndrome? J Clin Endocrinol Metab 1998;83:757–60.
Phillips DI, Walker BR, Reynolds RM, et al. Low birth weight predicts elevated plasma cortisol concentrations in adults from 3 populations. Hypertension 2000;35:1301–6.
Martinez-Aguayo A, Aglony M, Bancalari R, et al. Birth weight is inversely associated with blood pressure and serum aldosterone and cortisol levels in children. Clin Endocrinol (Oxf) 2012;76:713–8.
Szathmári M, Vásárhelyi B, Reusz G, Tulassay T. Adult cardiovascular risk factors in premature babies. Lancet 2000;356:939–40.
de Jong M, Lafeber HN, Cranendonk A, van Weissenbruch MM. Components of the metabolic syndrome in early childhood in very-low-birth-weight infants. Horm Res Paediatr 2014;81:43–9.
Duncan AF, Heyne RJ, Morgan JS, Ahmad N, Rosenfeld CR. Elevated systolic blood pressure in preterm very-low-birth-weight infants ≤3 years of life. Pediatr Nephrol 2011;26:1115–21.
Winter JS, Faiman C, Hobson WC, Prasad AV, Reyes FI. Pituitary-gonadal relations in infancy. I. Patterns of serum gonadotropin concentrations from birth to four years of age in man and chimpanzee. J Clin Endocrinol Metab 1975;40:545–51.
Winter JS, Hughes IA, Reyes FI, Faiman C. Pituitary-gonadal relations in infancy: 2. Patterns of serum gonadal steroid concentrations in man from birth to two years of age. J Clin Endocrinol Metab 1976;42:679–86.
Plagemann A. A matter of insulin: developmental programming of body weight regulation. J Matern Fetal Neonatal Med 2008;21:143–8.
Rosmond R, Dallman MF, Björntorp P. Stress-related cortisol secretion in men: relationships with abdominal obesity and endocrine, metabolic and hemodynamic abnormalities. J Clin Endocrinol Metab 1998;83:1853–9.
Matsukura T, Kawai M, Marumo C, et al. Diagnostic value of salivary cortisol in the CRH stimulation test in premature infants. J Clin Endocrinol Metab 2012;97:890–6.
Cetinkaya S, Ozon A, Yordam N. Diagnostic value of salivary cortisol in children with abnormal adrenal cortex functions. Horm Res 2007;67:301–6.
Jones A, Godfrey KM, Wood P, Osmond C, Goulden P, Phillips DI. Fetal growth and the adrenocortical response to psychological stress. J Clin Endocrinol Metab 2006;91:1868–71.
Beardsall K, Vanhaesebrouck S, Ogilvy-Stuart AL, et al. Early insulin therapy in very-low-birth-weight infants. N Engl J Med 2008;359:1873–84.
Fredriks AM, van Buuren S, Burgmeijer RJ, et al. Continuing positive secular growth change in The Netherlands 1955-1997. Pediatr Res 2000;47:316–23.
Fredriks AM, van Buuren S, Wit JM, Verloove-Vanhorick SP. Body index measurements in 1996-7 compared with 1980. Arch Dis Child 2000;82:107–12.
Heijboer AC, Martens F, Blankenstein MA. Measuring salivary cortisol with the Architect i2000 random access analyser. Ann Clin Biochem 2009;46(Pt 3):261–2.
Niklasson A, Ericson A, Fryer JG, Karlberg J, Lawrence C, Karlberg P. An update of the Swedish reference standards for weight, length and head circumference at birth for given gestational age (1977-1981). Acta Paediatr Scand 1991;80:756–62.
Author information
Authors and Affiliations
Corresponding author
PowerPoint slides
Rights and permissions
About this article
Cite this article
de Jong, M., Cranendonk, A. & van Weissenbruch, M. Salivary and serum cortisol and relation to blood pressure in infancy and early childhood in very-low-birth-weight infants. Pediatr Res 78, 476–479 (2015). https://doi.org/10.1038/pr.2015.128
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pr.2015.128