Abstract
While critical for normal development, the exact timing of establishment of the intestinal microbiome is unknown. For example, although preterm labor and birth have been associated with bacterial colonization of the amniotic cavity and fetal membranes for many years, the prevailing dogma of a sterile intrauterine environment during normal term pregnancies has been challenged more recently. While found to be a key contributor of evolution in the animal kingdom, maternal transmission of commensal bacteria may also constitute a critical process during healthy pregnancies in humans with yet unclear developmental importance. Metagenomic sequencing has elucidated a rich placental microbiome in normal term pregnancies likely providing important metabolic and immune contributions to the growing fetus. Conversely, an altered microbial composition during pregnancy may produce aberrant metabolites impairing fetal brain development and life-long neurological outcomes. Here we review the current understanding of microbial colonization at the feto-maternal interface and explain how normal gut colonization drives a balanced neonatal mucosal immune system, while dysbiosis contributes to aberrant immune function early in life and beyond. We discuss how maternal genetics, diet, medications, and probiotics inform the fetal microbiome in preparation for perinatal and postnatal bacterial colonization.
Similar content being viewed by others
Microbial Colonization In Utero
Although critically important for normal development of the intestinal mucosal immune system (1), the exact timing of bacterial colonization of the intestinal flora is unknown. Microbial invasion of the amniotic cavity has been well described in women with preterm rupture of membranes (2). In fact, intrauterine infection is considered the most common cause of spontaneous preterm birth (3). However bacteria can also be cultured from chorioamniotic tissue using sterile procedures during cesarean sections in cases of preterm labor with intact membranes, indicating a possible transmission of bacteria from the maternal blood stream into the amniotic cavity (3). Recently, the combination of bacterial culture techniques with culture independent molecular methods revealed that the amniotic cavity harbors a greater diversity of microbes than previously suspected (4), including uncultivated, previously uncharacterized taxa (5). In this cohort of pregnant women with intact membranes, a strong inverse correlation exists between intra-amniotic detection of bacteria via culture or polymerase chain reaction and gestational age at delivery. The type and number of bacteria in the amniotic cavity likely are important for pregnancy outcomes since amniotic fluid concentrations of white blood cells and interleukin-6 were highest when bacteria were detected by both culture and polymerase chain reaction.
Historically, Ureaplasma species of the Firmicutes phylum were implicated as culprits of perinatal infection and severe neonatal morbidity and mortality (6). Ureaplasma has been isolated from amniotic fluid as early as 16–20 wk gestation and its presence in the chorioamnion alone or in the company of other microbes has been strongly associated with histologic chorioamnionitis and preterm birth (7,8). Though originally suspected to ascend from the genitourinary tract secondary to maternal colonization, hematogenous transmission via placental infection or at delivery through an infected birth canal are considered an additional source of fetal and perinatal exposure (9). The exact role of Ureaplasma spp. in chorioamnionitis and adverse pregnancy outcomes remains controversial since up to 60–80% of women are colonized and only a small percentage of pregnancies develop histologically proven chorioamnionitis (10,11). In addition, clinical studies in which women were treated for Ureaplasma before and after birth demonstrated no differences in neonatal outcomes (11). Compelling data has recently emerged associating in utero Ureaplasma exposure with increased rates of bronchopulmonary dysplasia, intraventricular hemorrhage, and necrotizing enterocolitis (12,13,14). Causal relationships and exact timing of Ureaplasma exposure in relation to the postnatal sequelae require further investigation. As may be the case for Ureaplasma exposure, some degree of bacterial colonization in utero may underlie normal fetal development. Because not all microbes cause preterm labor, only specific bacteria within fetal membranes, other areas of the feto-maternal unit or even the fetal gastrointestinal tract may drive adverse pregnancy outcomes during critical developmental windows (15). It remains elusive whether “pathologic” bacterial colonization is always a primary factor in premature birth.
It is possible that maternal immune factors play an important role in bacterial invasion of the amniotic cavity and/or promote an inflammatory cascade resulting in preterm labor (16). For example, prostaglandin E2 expression is a potent negative regulator of innate immunity in the human placenta and has been shown to foster bacterial dissemination (17). Surprisingly, little is known about the innate immune capacities of intact fetal membranes, which serve as the final barrier between the external environment and the incubating fetus. Innate immune responses including antimicrobial peptide expression, such as human beta defensins (hβds), were studied ex vivo in human extraplacental membranes after inoculation with live Streptococcus agalactiae (GBS) (18). After GBS inoculation, hβd expression increased concomitant with markedly diminished recovery of GBS from fetal membranes. This robust innate immune response inhibited subsequent GBS colonization of the placenta and fetus. Additional studies are necessary to further examine the role of the maternal immune response in fetal microbial colonization and outcomes of pregnancy.
Despite an appreciation for in utero bacterial colonization in the setting of preterm labor and preterm rupture of membranes, the amniotic environment has traditionally been considered sterile in normal term pregnancies. Though this paradigm is conceptionally attractive, it’s long-held dogma is nevertheless surprising considering that maternal microbial transmission is a universal phenomenon in the animal kingdom and recently has been thought to play a critical role in evolutionary development (19,20). In addition to a potential “signature” fetal microbiome, a vibrant placental microbiome has been uncovered with the application of whole metagenomic shotgun sequencing. In studies with human placental tissue, investigators revealed a vibrant placental microbiome harboring specific metabolic functions (21). The overlap between the metabolic functions of the placenta and the developing fetus are largely unknown.
Multiple recent studies employing meconium as a proxy for in utero bacterial communities suggest that bacterial transmission from the mother to the fetus is a regular occurrence during human pregnancies. For example, traditional culture of meconium portions in 21 healthy term newborns obtained within 2 h of birth and prior to feeding revealed a diverse group of Gram-positive and Gram-negative bacteria (22). The most commonly isolated genus was Enterococcus (17 samples, 80%), followed by Staphylococcus (11 samples, 52%). Another piece of evidence stems from studies comparing the microbiome between meconium and sequentially collected fecal samples in neonates. When meconium collected from 14 preterm infants was evaluated and compared with postnatal fecal samples collected prospectively, the meconium microbiota was significantly different than that of feces obtained within the first week of life (23). Bacilli and other Firmicutes were the main bacteria groups detected in meconium while Proteobacteria dominated fecal samples. Culture-dependent techniques showed that Staphylococcus predominated in meconium, while Enterococcus, together with Gram-negative bacteria such as Escherichia coli, Escherichia fergusonii, Klebsiella pneumoniae, and Serratia marcescens, was more abundant in fecal samples. More severe prematurity correlated with an increased detection of microbiota in meconium and a microbial community present in amniotic fluid and rich in proinflammatory cytokines (24). Specific phyla were more abundant at lower gestation, however, the exact mechanism of these potentially immunoreactive bacteria in triggering a fetal inflammatory response syndrome followed by preterm birth remains to be studied.
Recently, we detected an unprecedented microbial diversity in intestinal tissue samples obtained directly from the operating room during resection for anatomic gut anomalies shortly after birth and prior to initiation of feeds in infants delivered via cesarean section (25). Diversity measures such as Simpson’s inverse index, Shannon-Wiener, and Chao 1 estimator were statistically significantly higher in the small intestinal mucosa compared to parallel collected fecal samples, suggesting a “top-to-bottom” pattern of microbial colonization that likely occurred in utero.
The Fetal Microbiome and Immune Development
Perhaps exposure to various microbial antigens in the human fetus explains the relative early development of a functional immune system. For example, CD4+ and CD8+ T cells can already be identified toward the end of the first trimester in the human fetus (26). More recently, Michaëlsson et al. (27). discovered that fetal T cells are highly responsive to stimulation and require control by an abundant and functional pool of FOXP3+ T regulatory (Treg) cells. Interestingly, fetal Treg cells appear to have been informed by substantial numbers of maternal cells that crossed the placenta and induced antigen-specific tolerance (28).
Maternal mononuclear blood cells carry whole bacteria or their genetic material at higher frequency and diversity during pregnancy (29). As some of these bacterial signatures are represented in infant feces, it is fascinating to speculate that in utero transfer of maternal microbial antigens during fetal development enables a balanced immune response in the newborn to the rapidly developing microbiome postpartum. This is especially intriguing in the context of bacteria known to be beneficial for immune regulation, such as Bifidobacteria and Lactobacilli (30,31). DNA from both of these probiotic bacteria has been detected repeatedly in the human placenta (32). This proposed balanced immune response to commensal flora in the fetus is in stark contrast to immune dysregulation observed after in utero exposure to potentially pathogenic bacteria. For example, colonization with Ureaplasma spp. may alter the mucosal and/or systemic immune milieu of the fetus and newborn and constitute a risk factor for adverse postnatal outcomes such as necrotizing enterocolitis, a common and often fatal complication of prematurity (14). It is not clear if bacterial products directly affect organ system development or if inflammatory responses at the feto-maternal interface lead to immunological imbalances and priming for harmful responses to postnatal triggers (33,34,35).
Maternal Influences on Fetal Intestinal Colonization
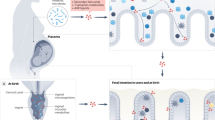
In this section, we describe how genetic predisposition, diet, medications, and other environmental influences shape the maternal microbiome, which informs the fetal microbiome in preparation for perinatal and postnatal bacterial colonization ( Figure 1 ).
Maternal influences on fetal microbial colonization and immune development. Maternal-fetal transmission of microbes is an evolutionary preserved phenomenon in the animal kingdom and likely an important mechanism for the development of a balanced immune system in the human fetus (19,69,70,71). (a) Maternal-fetal bacterial transfer is poorly understood and multiple mechanisms have been proposed (22,40,41,42,43,44,72). (b) Fetal and maternal host immunogenetic factors play a role in assemblage of the fetal and neonatal microbiome. Specific genes of interest are listed in Table 1 and reviewed by Spor et al. (73). (c) Ascending infection and ensuing chorioamnionitis have a major impact on the uterine environment and subsequent fetal development, including the intestinal microbiome (5,24). (d) Delivery mode (63,64,65) and diet (68,74) strongly influence early life microbial colonization. Other life events, especially introduction of antibiotics and solid food, are associated with major shifts in the microbiota composition (75). Despite taxonomic variation, the functionality of the newborn intestinal microbiota converges towards his/her mother’s microbiome, supporting the early and sustained role of the maternal environment on health (76).
Maternal Genotype
In order to define the effects of maternal genotype on the fetal and neonatal microbiome, environmental and fetal genetic influences have to be controlled for. Murine models have been particularly informative by transplanting pups to dams with different genetic origins in order to dissociate mouse pups from the influences of the microbiota from their respective families. In such studies, mice from different genetic lines that were born together after transplantation to a new dam had similar microbiota (36). This suggests a new maternal effect that is introduced by the birth mother.
While human twin studies have provided equivocal data on how host genetics may influence microbial assemblage (37,38,39), several single-gene studies have demonstrated a strong effect of specific host genes on microbial composition and several of these gene candidates are listed in Table 1 . As one would expect, the majority of these single gene effects are observed with genes that regulate immune function and metabolism.
Maternal Diet and Medications
Maternal diet seems to be a significant contributor to healthy pregnancy outcomes and prevention of premature birth (40). Specifically, consumption of probiotic containing food components may reduce the risk for spontaneous preterm delivery (41) and childhood allergic diseases (42). In a randomized trial in Finland a few years ago, pregnant mothers were given Bifidobacterium lactis, in combination with Lactobacillus rhamnosus GG (LGG) or placebo for 14 d before elective cesarean section at full term (43). Microbial DNA in amniotic fluid and placenta was associated with changes in innate immunity gene expression in placenta and meconium. Novel probiotic bacterial-derived proteins, LGG p75 (75 kilodaltons) and p40 (40 kilodaltons), are particularly promising as maternal therapies during pregnancy (44). In murine models of inflammatory bowel disease, they suppress cytokine-induced colonic epithelial apoptosis and injury. While other live probiotics have been associated with bacteremia in the very young (45), these soluble proteins do not pose the same risks yet still have potent anti-inflammatory activity at the level of the intestinal mucosa. Manipulation of the maternal microbiota with dietary supplements such as p40 and p75 could potentially temporize in utero fetal inflammatory responses triggered by chorioamnionitis and restore intestinal microbial homeostasis at the feto-maternal interface.
Antibiotics are one of the few medications whose effects on the maternal and fetal microbiomes have been studied (46). When given to pregnant dams, an altered neonatal gut microbiome, reduced intestinal host defense and increased risk for neonatal sepsis were observed. This was thought to be due to decreased transfer of bacteria during and/or shortly after delivery. However, it is likewise possible that maternal antibiotics limit bacterial gut colonization of the fetus even before birth resulting in anomalous immune priming.
A recent study demonstrates how high-fat maternal diet alters the offspring microbiome in primates (47). Therefore, while a shift in the intestinal microbiome during pregnancy due to hormonal or other factors may influence maternal and possibly fetal metabolism, conversely changes in pregnancy diet may shape the maternal and neonatal microbiome.
Maternal Microbiome
Transfer of bacteria from the pregnant mother to the fetus is universal in the animal kingdom. In vertebrates, one common example is transmission of Salmonella bacteria from contaminated chicken eggs. In an elegant experiment, Jiménez et al. (22) orally inoculated pregnant mice with genetically labeled Enterococcus that had been previously isolated from the breast milk of a healthy woman. The labeled bacteria were retrieved after term cesarean section delivery of pups from the internal meconium. However, labeled bacteria were not detected in the pups of control animals that had not been inoculated.
The exact mechanism of materno-fetal bacterial transfer is unknown. However, at least in mammals, the maternal fecal microbiome appears to shape the microbiota in the fetal intestine, and therefore, the maternal gastrointestinal tract is the most likely source (48,49,50). Bacterial translocation from the intestine to the maternal blood stream and from there to other organ systems is increased during pregnancy and lactation (29). Although not yet clear which process is active during human pregnancy, multiple pathways have been described for migration of luminal bacteria through the intact small intestinal epithelium. These include dendritic cells (51,52,53), microfold (M) cells (54), and goblet cells (55). In addition to transmigration of intact organisms, molecules such as polysaccharide A (PSA), produced by Bacteroides fragilis, influence the mucosal immune structure. PSA induces generation of FOXP3+ Treg cells in the intestinal mucosa, which are critical for tolerance to food antigens and prevention of inflammatory complications in the gut (56,57,58). Supplementation with Lactobacillus rhamnosus or Bifidobacterium lactis probiotics during pregnancy modulated the fetal immune response and enhanced immunoprotective components in breast milk (59). Exposure of the fetal intestine to commensal organisms or their products in swallowed amniotic fluid may be an important contributor to gut maturation. Indirect effects on the fetal microbiome may be achieved by other forms of maternal exposures such as gluten-free diet. Gluten-free diet fed during pregnancy changed gut microbiota in both mothers and offsprings resulting in shifts to a less proinflammatory immunological milieu in the gut and pancreas (60).
Aagard’s recent study, detailing a robust placental microbiome, suggests a different mode for transmission of bacteria to the fetus. The investigators found that the placenta microbiome resembled microbiome in an adult human’s mouth more than the vaginal, skin, gut, or other body microbiomes. However, comparisons between placental tissue and oral microbiota were not made between the same individuals and all oral samples were collected from nonpregnant patients. Given the shifts in the bacterial metabolic machinery (61) and systemic and mucosal immune functions (62) during pregnancy, the findings on the origin of the placenta microbiome require further validation in a cohort of pregnant women.
Maternal Environment
Immune stimulation such as during maternal viral infections can possibly also alter the intestinal microbiome during pregnancy resulting in potentially long-lasting consequences for the offspring that are not limited to the digestive system. One example of where immune activation and aberrant maternal flora affect fetal brain development and determine life-long neurological outcomes was recently reported in an elegant study by Hsiao et al. (63). In this study, when pregnant mice were injected with polyinosinic:polycytidylic acid (poly(I:C)) mimicking viral infection, their offspring exhibited autism-like behavior and neuropathology. In this model, maternal immune activation also reduced gut barrier integrity in offspring and significantly altered their microbial composition and metabolites. Oral treatment with Bacteroides fragilis corrected gut permeability, microbial composition, and improved autism-like behavior in pubs born to mothers with a history of immune activation during pregnancy. This study suggests that altering fetal exposure to microbial antigens can cause persistent microbiome changes with enduring consequences. Regulating the potentially adverse microbial metabolism after inflammatory exposures during pregnancy may be a powerful approach in the future to reduce the risk of chronic diseases.
Delivery Mode
Comprehensive studies of the succession of the microbiota immediately after delivery include those described by Dominguez-Bello et al. (64) in a cohort of maternal-newborn dyads. Infants born vaginally acquired bacterial communities resembling their own mother’s vaginal microbiota, while those delivered by cesarean section harbored bacterial communities similar to organisms found on maternal skin surfaces. Longitudinal fecal sampling reveals the striking persistence of delivery-imparted changes in the gut microbiota at 4 mo (65) and up until 2 y of age (66), including loss of bacterial richness and diversity in infants born via caesarean delivery. While health implications from inheritance of such divergent microbiota remain largely unknown, an association between cesarean sections and obesity in offsprings has been reported (67). In addition, while the fetus was assumed to be sterile in this study, fetal microbial colonization, as described above, may be setting the stage for intestinal microbial homeostasis or dysbiosis prior to delivery.
Conclusion
While long accepted in the animal kingdom, routine transmission of bacterial antigen from mother to fetus during normal human pregnancy remains to be confirmed. While evidence is accumulating that microbial programming begins in utero and is a central component for the development of a balanced mucosal immune system in the newborn, the current research is preliminary and more studies are needed. The specific types of bacteria along with their products and communication with maternal and fetal host factors likely determine the fetal immune composition as well as immediate and long-lasting health outcomes. Fetal programming is substantially modulated during and after birth by mode of delivery, perinatal antibiotics, and type of feeding. In addition, maternal and fetal host genetic and environmental factors may play a role and our understanding of these complex interactions is currently insufficient. More knowledge in this area could inform future clinical trials of microbial manipulation of the maternal diet as a possible strategy to prevent adverse pregnancy outcomes and to foster the development of a healthy microbiome in the offspring (68).
Statement of Financial Support
This project was supported by the American Academy of Pediatrics Marshall Klaus Perinatal Research Award and the Eunice Kennedy Shriver National Institute of Child Health & Human Development (NICHD) grants T32HD068256 and K08HD061607. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NICHD or the National Institutes of Health (NIH). The authors were also funded by the Vanderbilt University (Nashville, TN) Digestive Disease Research Center Grant P30DK058404 and the Vanderbilt CTSA Grant UL1 RR024975-01 from the National Center for Research Resources (NCRR/NIH).
Disclosure
The authors have no financial disclosures or potential conflicts of interest related to products described in this article.
References
Grönlund MM, Arvilommi H, Kero P, Lehtonen OP, Isolauri E . Importance of intestinal colonisation in the maturation of humoral immunity in early infancy: a prospective follow up study of healthy infants aged 0-6 months. Arch Dis Child Fetal Neonatal Ed 2000;83:F186–92.
DiGiulio DB, Romero R, Kusanovic JP, et al. Prevalence and diversity of microbes in the amniotic fluid, the fetal inflammatory response, and pregnancy outcome in women with preterm pre-labor rupture of membranes. Am J Reprod Immunol 2010;64:38–57.
Goldenberg RL, Hauth JC, Andrews WW . Intrauterine infection and preterm delivery. N Engl J Med 2000;342:1500–7.
Hitti J, Riley DE, Krohn MA, et al. Broad-spectrum bacterial rDNA polymerase chain reaction assay for detecting amniotic fluid infection among women in premature labor. Clin Infect Dis 1997;24:1228–32.
DiGiulio DB, Romero R, Amogan HP, et al. Microbial prevalence, diversity and abundance in amniotic fluid during preterm labor: a molecular and culture-based investigation. PLoS One 2008;3:e3056.
Kundsin RB, Driscoll SG, Monson RR, Yeh C, Biano SA, Cochran WD . Association of Ureaplasma urealyticum in the placenta with perinatal morbidity and mortality. N Engl J Med 1984;310:941–5.
Cassell GH, Davis RO, Waites KB, et al. Isolation of Mycoplasma hominis and Ureaplasma urealyticum from amniotic fluid at 16-20 weeks of gestation: potential effect on outcome of pregnancy. Sex Transm Dis 1983;10:Suppl 4:294–302.
Hillier SL, Martius J, Krohn M, Kiviat N, Holmes KK, Eschenbach DA . A case-control study of chorioamnionic infection and histologic chorioamnionitis in prematurity. N Engl J Med 1988;319:972–8.
Viscardi RM . Ureaplasma species: role in diseases of prematurity. Clin Perinatol 2010;37:393–409.
Sung TJ . Ureaplasma infections in pre-term infants: Recent information regarding the role of Ureaplasma species as neonatal pathogens. Korean J Pediatr 2010;53:989–93.
Carey JC, Blackwelder WC, Nugent RP, et al. Antepartum cultures for Ureaplasma urealyticum are not useful in predicting pregnancy outcome. The Vaginal Infections and Prematurity Study Group. Am J Obstet Gynecol 1991;164:728–33.
Kallapur SG, Kramer BW, Jobe AH . Ureaplasma and BPD. Semin Perinatol 2013;37:94–101.
Kasper DC, Mechtler TP, Böhm J, et al. In utero exposure to Ureaplasma spp. is associated with increased rate of bronchopulmonary dysplasia and intraventricular hemorrhage in preterm infants. J Perinat Med 2011;39:331–6.
Okogbule-Wonodi AC, Gross GW, Sun CC, et al. Necrotizing enterocolitis is associated with ureaplasma colonization in preterm infants. Pediatr Res 2011;69(5 Pt 1):442–7.
Steel JH, Malatos S, Kennea N, et al. Bacteria and inflammatory cells in fetal membranes do not always cause preterm labor. Pediatr Res 2005;57:404–11.
Jones HE, Harris KA, Azizia M, et al. Differing prevalence and diversity of bacterial species in fetal membranes from very preterm and term labor. PLoS One 2009;4:e8205.
Mason KL, Rogers LM, Soares EM, et al. Intrauterine group A streptococcal infections are exacerbated by prostaglandin E2. J Immunol 2013;191:2457–65.
Boldenow E, Jones S, Lieberman RW, et al. Antimicrobial peptide response to group B Streptococcus in human extraplacental membranes in culture. Placenta 2013;34:480–5.
Funkhouser LJ, Bordenstein SR . Mom knows best: the universality of maternal microbial transmission. PLoS Biol 2013;11:e1001631.
Brucker RM, Bordenstein SR . The hologenomic basis of speciation: gut bacteria cause hybrid lethality in the genus Nasonia. Science 2013;341:667–9.
Aagaard KM . Author response to comment on “The placenta harbors a unique microbiome”. Sci Transl Med 2014;6:254lr3.
Jiménez E, Marín ML, Martín R, et al. Is meconium from healthy newborns actually sterile? Res Microbiol 2008;159:187–93.
Moles L, Gómez M, Heilig H, et al. Bacterial diversity in meconium of preterm neonates and evolution of their fecal microbiota during the first month of life. PLoS One 2013;8:e66986.
Ardissone AN, de la Cruz DM, Davis-Richardson AG, et al. Meconium microbiome analysis identifies bacteria correlated with premature birth. PLoS One 2014;9:e90784.
Romano-Keeler J, Moore DJ, Wang C, et al. Early life establishment of site-specific microbial communities in the gut. Gut Microbes 2014;5:192–201.
Spencer J, Dillon SB, Isaacson PG, MacDonald TT . T cell subclasses in fetal human ileum. Clin Exp Immunol 1986;65:553–8.
Michaëlsson J, Mold JE, McCune JM, Nixon DF . Regulation of T cell responses in the developing human fetus. J Immunol 2006;176:5741–8.
Mold JE, Michaëlsson J, Burt TD, et al. Maternal alloantigens promote the development of tolerogenic fetal regulatory T cells in utero. Science 2008;322:1562–5.
Perez PF, Doré J, Leclerc M, et al. Bacterial imprinting of the neonatal immune system: lessons from maternal cells? Pediatrics 2007;119:e724–32.
Vlasova AN, Chattha KS, Kandasamy S, et al. Lactobacilli and bifidobacteria promote immune homeostasis by modulating innate immune responses to human rotavirus in neonatal gnotobiotic pigs. PLoS One 2013;8:e76962.
Foye OT, Huang IF, Chiou CC, Walker WA, Shi HN . Early administration of probiotic Lactobacillus acidophilus and/or prebiotic inulin attenuates pathogen-mediated intestinal inflammation and Smad 7 cell signaling. FEMS Immunol Med Microbiol 2012;65:467–80.
Satokari R, Grönroos T, Laitinen K, Salminen S, Isolauri E . Bifidobacterium and Lactobacillus DNA in the human placenta. Lett Appl Microbiol 2009;48:8–12.
Wolfs TG, Kallapur SG, Knox CL, et al. Antenatal ureaplasma infection impairs development of the fetal ovine gut in an IL-1-dependent manner. Mucosal Immunol 2013;6:547–56.
Kallapur SG, Presicce P, Senthamaraikannan P, et al. Intra-amniotic IL-1β induces fetal inflammation in rhesus monkeys and alters the regulatory T cell/IL-17 balance. J Immunol 2013;191:1102–9.
Kallapur SG, Presicce P, Rueda CM, Jobe AH, Chougnet CA . Fetal immune response to chorioamnionitis. Semin Reprod Med 2014;32:56–67.
Friswell MK, Gika H, Stratford IJ, et al. Site and strain-specific variation in gut microbiota profiles and metabolism in experimental mice. PLoS One 2010;5:e8584.
Turnbaugh PJ, Hamady M, Yatsunenko T, et al. A core gut microbiome in obese and lean twins. Nature 2009;457:480–4.
Stewart JA, Chadwick VS, Murray A . Investigations into the influence of host genetics on the predominant eubacteria in the faecal microflora of children. J Med Microbiol 2005;54(Pt 12):1239–42.
Turnbaugh PJ, Quince C, Faith JJ, et al. Organismal, genetic, and transcriptional variation in the deeply sequenced gut microbiomes of identical twins. Proc Natl Acad Sci USA 2010;107:7503–8.
Englund-Ögge L, Brantsæter AL, Sengpiel V, et al. Maternal dietary patterns and preterm delivery: results from large prospective cohort study. BMJ 2014;348:g1446.
Myhre R, Brantsæter AL, Myking S, et al. Intake of probiotic food and risk of spontaneous preterm delivery. Am J Clin Nutr 2011;93:151–7.
Bertelsen RJ, Brantsæter AL, Magnus MC, et al. Probiotic milk consumption in pregnancy and infancy and subsequent childhood allergic diseases. J Allergy Clin Immunol 2014;133:165–71.e1–8.
Rautava S, Collado MC, Salminen S, Isolauri E . Probiotics modulate host-microbe interaction in the placenta and fetal gut: a randomized, double-blind, placebo-controlled trial. Neonatology 2012;102:178–84.
Yan F, Cao H, Cover TL, Whitehead R, Washington MK, Polk DB . Soluble proteins produced by probiotic bacteria regulate intestinal epithelial cell survival and growth. Gastroenterology 2007;132:562–75.
Land MH, Rouster-Stevens K, Woods CR, Cannon ML, Cnota J, Shetty AK . Lactobacillus sepsis associated with probiotic therapy. Pediatrics 2005;115:178–81.
Deshmukh HS, Liu Y, Menkiti OR, et al. The microbiota regulates neutrophil homeostasis and host resistance to Escherichia coli K1 sepsis in neonatal mice. Nat Med 2014;20:524–30.
Ma J, Prince AL, Bader D, et al. High-fat maternal diet during pregnancy persistently alters the offspring microbiome in a primate model. Nat Commun 2014;5:3889.
Hu J, Nomura Y, Bashir A, et al. Diversified microbiota of meconium is affected by maternal diabetes status. PLoS One 2013;8:e78257.
Thum C, Cookson AL, Otter DE, et al. Can nutritional modulation of maternal intestinal microbiota influence the development of the infant gastrointestinal tract? J Nutr 2012;142:1921–8.
Tannock GW, Fuller R, Pedersen K . Lactobacillus succession in the piglet digestive tract demonstrated by plasmid profiling. Appl Environ Microbiol 1990;56:1310–6.
Rescigno M, Rotta G, Valzasina B, Ricciardi-Castagnoli P . Dendritic cells shuttle microbes across gut epithelial monolayers. Immunobiology 2001;204:572–81.
Macpherson AJ, Uhr T . Induction of protective IgA by intestinal dendritic cells carrying commensal bacteria. Science 2004;303:1662–5.
Lelouard H, Fallet M, de Bovis B, Méresse S, Gorvel JP . Peyer’s patch dendritic cells sample antigens by extending dendrites through M cell-specific transcellular pores. Gastroenterology 2012;142:592–601.e3.
Jang MH, Kweon MN, Iwatani K, et al. Intestinal villous M cells: an antigen entry site in the mucosal epithelium. Proc Natl Acad Sci USA 2004;101:6110–5.
McDole JR, Wheeler LW, McDonald KG, et al. Goblet cells deliver luminal antigen to CD103+ dendritic cells in the small intestine. Nature 2012;483:345–9.
Round JL, Mazmanian SK . Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc Natl Acad Sci USA 2010;107:12204–9.
Weitkamp JH, Koyama T, Rock MT, et al. Necrotising enterocolitis is characterised by disrupted immune regulation and diminished mucosal regulatory (FOXP3)/effector (CD4, CD8) T cell ratios. Gut 2013;62:73–82.
Dingle BM, Liu Y, Fatheree NY, Min J, Rhoads JM, Tran DQ . FoxP3+ regulatory T cells attenuate experimental necrotizing enterocolitis. PLoS One 2013;8:e82963.
Prescott SL, Wickens K, Westcott L, et al.; Probiotic Study Group. Supplementation with Lactobacillus rhamnosus or Bifidobacterium lactis probiotics in pregnancy increases cord blood interferon-gamma and breast milk transforming growth factor-beta and immunoglobin A detection. Clin Exp Allergy 2008;38:1606–14.
Hansen CH, Krych L, Buschard K, et al. A maternal gluten-free diet reduces inflammation and diabetes incidence in the offspring of NOD mice. Diabetes 2014;63:2821–32.
Koren O, Goodrich JK, Cullender TC, et al. Host remodeling of the gut microbiome and metabolic changes during pregnancy. Cell 2012;150:470–80.
Munoz-Suano A, Hamilton AB, Betz AG . Gimme shelter: the immune system during pregnancy. Immunol Rev 2011;241:20–38.
Hsiao EY, McBride SW, Hsien S, et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell 2013;155:1451–63.
Dominguez-Bello MG, Costello EK, Contreras M, et al. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc Natl Acad Sci USA 2010;107:11971–5.
Azad MB, Konya T, Maughan H, et al.; CHILD Study Investigators. Gut microbiota of healthy Canadian infants: profiles by mode of delivery and infant diet at 4 months. CMAJ 2013;185:385–94.
Jakobsson HE, Abrahamsson TR, Jenmalm MC, et al. Decreased gut microbiota diversity, delayed Bacteroidetes colonisation and reduced Th1 responses in infants delivered by caesarean section. Gut 2014;63:559–66.
Darmasseelane K, Hyde MJ, Santhakumaran S, Gale C, Modi N . Mode of delivery and offspring body mass index, overweight and obesity in adult life: a systematic review and meta-analysis. PLoS One 2014;9:e87896.
Fåk F, Ahrné S, Molin G, Jeppsson B, Weström B . Maternal consumption of Lactobacillus plantarum 299v affects gastrointestinal growth and function in the suckling rat. Br J Nutr 2008;100:332–8.
De Palma G, Capilla A, Nadal I, et al. Interplay between human leukocyte antigen genes and the microbial colonization process of the newborn intestine. Curr Issues Mol Biol 2010;12:1–10.
Khachatryan ZA, Ktsoyan ZA, Manukyan GP, Kelly D, Ghazaryan KA, Aminov RI . Predominant role of host genetics in controlling the composition of gut microbiota. PLoS One 2008;3:e3064.
Wen L, Ley RE, Volchkov PY, et al. Innate immunity and intestinal microbiota in the development of Type 1 diabetes. Nature 2008;455:1109–13.
Frank DN, Robertson CE, Hamm CM, et al. Disease phenotype and genotype are associated with shifts in intestinal-associated microbiota in inflammatory bowel diseases. Inflamm Bowel Dis 2011;17:179–84.
Spor A, Koren O, Ley R . Unravelling the effects of the environment and host genotype on the gut microbiome. Nat Rev Microbiol 2011;9:279–90.
Jost T, Lacroix C, Braegger CP, Chassard C . New insights in gut microbiota establishment in healthy breast fed neonates. PLoS One 2012;7:e44595.
Koenig JE, Spor A, Scalfone N, et al. Succession of microbial consortia in the developing infant gut microbiome. Proc Natl Acad Sci USA 2011;108:Suppl 1:4578–85.
Vaishampayan PA, Kuehl JV, Froula JL, Morgan JL, Ochman H, Francino MP . Comparative metagenomics and population dynamics of the gut microbiota in mother and infant. Genome Biol Evol 2010;2:53–66.
Mirpuri J, Raetz M, Sturge CR, et al. Proteobacteria-specific IgA regulates maturation of the intestinal microbiota. Gut Microbes 2014;5:28–39.
Lenman Y . Selective IgA deficiency. J Clin Immunol 2010;30:10–16.
Salzman NH, Hung K, Haribhai D, et al. Enteric defensins are essential regulators of intestinal microbial ecology. Nat Immunol 2010;11:76–83.
Wehkamp J, Salzman NH, Porter E, et al. Reduced Paneth cell alpha-defensins in ileal Crohn’s disease. Proc Natl Acad Sci USA 2005;102:18129–34.
Wehkamp J, Wang G, Kübler I, et al. The Paneth cell alpha-defensin deficiency of ileal Crohn’s disease is linked to Wnt/Tcf-4. J Immunol 2007;179:3109–18.
Barbosa T, Rescigno M . Host-bacteria interactions in the intestine: homeostasis to chronic inflammation. Wiley Interdiscip Rev Syst Biol Med 2010;2:80–97.
Acknowledgements
We apologize to colleagues whose work could not be cited due to space constraints.
Author information
Authors and Affiliations
Corresponding author
PowerPoint slides
Rights and permissions
About this article
Cite this article
Romano-Keeler, J., Weitkamp, JH. Maternal influences on fetal microbial colonization and immune development. Pediatr Res 77, 189–195 (2015). https://doi.org/10.1038/pr.2014.163
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pr.2014.163
This article is cited by
-
Investigating prenatal and perinatal factors on meconium microbiota: a systematic review and cohort study
Pediatric Research (2024)
-
In Utero Exposure to Antibiotics and Risk of Serious Infections in the First Year of Life
Drug Safety (2024)
-
Bacterial extracellular vesicles in the microbiome of first-pass meconium in newborn infants
Pediatric Research (2023)
-
The impact of the gut microbiota on T cell ontogeny in the thymus
Cellular and Molecular Life Sciences (2022)
-
Presence of distinctive microbiome in the first-pass meconium of newborn infants
Scientific Reports (2021)