Abstract
Background:
In adults, obesity-driven inflammation can lead to increased cardiovascular disease (CVD). However, information regarding childhood obesity and its inflammatory sequelae is less well defined. Serum amyloid-A (SAA) is an inflammatory molecule that rapidly associates with high-density lipoproteins (HDLs) and renders them dysfunctional. Therefore, SAA may be a useful biomarker to identify increased CVD potential in overweight and obese children.
Methods:
Young Hearts 2000 is a cross-sectional cohort study in which 92 children who were obese were matched for age and sex with 92 overweight and 92 lean children. HDL2 and HDL3 (HDL2&3) were isolated from plasma by a three-step rapid-ultracentrifugation procedure. SAA was measured in serum and HDL2&3 by an enzyme-linked immunosorbent assay procedure, and the activities of cholesterol ester transfer protein (CETP) and lecithin cholesteryl acyltransferase (LCAT) were measured by fluorimetric assays.
Results:
Trends across the groups indicated that SAA increased in serum and HDL2&3 as BMI increased, as did HDL2-CETP and HDL2-LCAT activities.
Conclusion:
These results have provided evidence that overweight and obese children are exposed to an inflammatory milieu that impacts the antiatherogenic properties of HDL and that could increase CVD risk. This supports the concept that it is important to target childhood obesity to help minimize future cardiovascular events.
Similar content being viewed by others
Main
Cardiovascular disease (CVD) is a leading cause of mortality and morbidity in the developed world (1), and its incidence is augmented with a concomitant increase in BMI (2,3). Furthermore, the global prevalence of childhood obesity has reached significant proportions, with more than one-third of children and adolescents being classed as either overweight or obese (4); this has led to fears that younger generations may experience the cardiovascular-associated complications identified in obese adult populations (3,5).
This is especially relevant because the obesity-related pathologies of atherosclerosis are known to begin in childhood (6) and track through into adult life (7), thus exposing these individuals to a continuous inflammatory milieu over their life-course. Conversely, minimizing obesity-related inflammation in young people may have far-reaching cardioprotective effects (5). This is important because obesity is characterized by adiposopathy and increased inflammation (8). One molecule that is released from hypertrophic adipocytes and that has been shown to predict CVD, is serum amyloid-A (SAA) (9,10). Furthermore, SAA has been suggested as a candidate molecule linking increased fat load and its comorbidities (11). Once in the circulation, SAA rapidly associates with high-density lipoproteins (HDLs), especially the smaller of the HDL subfractions, HDL3, rendering them dysfunctional (12) with numerous proinflammatory actions (13,14). Dysfunctional HDL has reduced reverse cholesterol transport capabilities (15) and antioxidant abilities (16). In addition, two enzymes involved in HDL remodeling, cholesteryl ester transfer protein (CETP) and lecithin cholesterol acyl transferase (LCAT), are altered to an atherogenic phenotype in the presence of SAA (16,17).
However, information regarding obesity-driven inflammation in younger generations is limited, with only a few studies having examined this process in serum. To date, studies have mainly focused on the more traditional markers of inflammation such as C-reactive protein, tumor necrosis factor-α, and interleukin-6 and -8 (18,19,20,21,22) showing consistent associations with obesity. We have also examined several of these inflammatory molecules in the cohort described here, showing that low-grade inflammation was associated with increased CVD risk (23). To our knowledge, only three studies have examined SAA in children. In the first, the authors found that SAA was not affected by the presence of increased weight, which we suggest may due to the young age of their prepubertal children (<10 y) (24). In a second study, both groups were obese, therefore limiting the likelihood of identifying any differences in SAA (25). On the other hand, in a third study, SAA was shown to be directly related to increased adiposity in a group of children with a mean age of 12 y (26). To date, an examination of the influence of SAA-related inflammation on HDL function in children is missing from the literature; this study addresses this shortcoming by examining age- and sex-matched obese, overweight, and normal-weight children. Inflammation was assessed by measuring SAA and its association with HDL’s two major subfractions, that is, HDL2 and HDL3 (HDL2&3), and its effect on the functioning of HDL2&3 was examined by measuring the activities of CETP and LCAT. These analyses will assist in determining if the function of HDL is altered to a more proatherogenic phenotype in overweight and obese children.
Results
Subject Characteristics
Subject characteristics are presented in Table 1 . The three groups were matched for age and gender, and, as expected, BMI increased across the groups (normal weight > overweight > obese, P ≤ 0.001). Similarly to BMI, triglyceride levels also increased across the groups (P ≤ 0.001), and total cholesterol was higher in the overweight and obese children as compared with the normal-weight children (P ≤ 0.05 for both comparisons). In addition, HDL cholesterol decreased with increasing BMI (P ≤ 0.001).
Serum Analyses
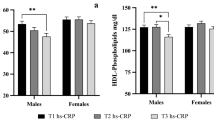
Both high-sensitive C-reactive protein and SAA increased with increasing BMI ( Table 2 ; P ≤ 0.001 for both analytes). However, the activities of serum CETP and LCAT were unaffected by BMI ( Table 2 ), although there was a trend for the activity of serum CETP to increase as BMI increased (P = 0.076).
HDL2 and HDL3 Analyses
SAA associated with HDL2&3 followed a similar trend to that of serum SAA, in which it increased within both HDL2&3, as BMI increased ( Table 3 , P ≤ 0.001 for both subfractions). In addition, SAA associated with HDL2 was significantly higher in the obese children as compared with both the overweight and normal-weight children (P ≤ 0.05 for both comparisons). SAA associated with HDL3 was significantly higher in both the overweight and obese children as compared with the normal-weight children (P ≤ 0.05 for both comparisons).
With regard to trends in the activities of CETP and LCAT within HDL2&3, these were also found to increase as BMI increased, although this was only statistically significant for HDL2 ( Table 3 , P ≤ 0.001 for both CETP and LCAT). In addition, the activity of HDL2-CETP was significantly higher in the obese children as compared with the overweight and normal-weight children (P ≤ 0.05 for both comparisons), whereas the activity of HDL2-LCAT was significantly higher in both the obese and overweight children as compared with the normal-weight children (P ≤ 0.05 for both comparisons).
Serum and HDL2&3 Correlations
Correlations in the complete cohort of 275 children (mean BMI, 24.6 (SD 5.18) kg/m2) showed that SAA positively correlated with HDL2&3-SAA (HDL2, r = 0.250, P ≤ 0.001; HDL3, r = 0.604, P ≤ 0.001). In addition, there were weak positive correlations between HDL2-SAA and the activity of HDL2-LCAT (r = 0.151, P = 0.022) and between HDL3-SAA and the activity of HDL3-CETP (r = 0.130, P = 0.036).
Correlations in each of the three weight groups showed that:
-
1
In the lean group (n = 92, mean BMI 19.9 (SD 2.2) kg/m2), SAA was positively correlated with HDL3-SAA (r = 0.410, P ≤ 0.001).
-
2
In the overweight group (n = 92, mean BMI 24.4 (SD 1.7) kg/m2), SAA was positively correlated with HDL3-SAA (r = 0.680, P ≤ 0.001).
-
3
In the obese group (n = 92, mean BMI 30.4 (SD 2.8) kg/m2), SAA was positively correlated with HDL2&3-SAA (r = 0.458, P ≤ 0.001; r = 0.554, P ≤ 0.001, respectively). In addition, in this group, HDL2-SAA positively correlated with the activity of CETP and LCAT in HDL2 (r = 0.325, P = 0.011; r = 0.356, P = 0.005, respectively). HDL3-SAA positively correlated with the activity of HDL3-CETP (r = 0.260, P = 0.014).
Discussion
To our knowledge, this is the first report to show an association between the inflammatory molecule SAA and HDL2&3 in overweight and obese children. Furthermore, this study is also the first study to identify changes within HDL2&3 within this cohort, which augmented the atherogenic potential of these subfractions. Therefore, these results significantly add to and are in support of our previous findings, in which low-grade inflammation was associated with increased CVD risk in overweight and obese children (23).
The inflammatory molecule SAA has consistently been shown to be a sensitive marker of obesity-related inflammation in adult populations (11,27), although its role in children is largely undefined. However, one study (24) found little utility for SAA as a marker of obesity-related inflammation, which we suggest may be due to the prepubertal status of their cohort (children <10 y). This lack of effect was also described by Neuman et al. (25), however, the BMIs of their study groups were similar, and would therefore display a similar level of obesity-related inflammation. However, our results were in support of Gómez-Ambrosi et al. (26), who reported that SAA was associated with increasing BMI in children. Therefore, we suggest that SAA is a sensitive marker for detecting obesity-related inflammation in children, similar to that identified in adults (11,27).
In addition to this, we have shown that this increase in SAA augmented its association with HDL; we found that SAA positively correlated with HDL3-SAA in the overweight group (r = 0.680, P ≤ 0.001) and with HDL2&3-SAA in the obese group (HDL2 r = 0.458, P ≤ 0.001; HDL3 r = 0.554, P ≤ 0.001), which supports the concept that SAA primarily associates with HDL in the circulation (15). In adults, this association renders HDL proatherogenic, with a loss or reduction of many of its functions, including its reverse cholesterol transport and antioxidant properties (15,28,29,30). Therefore, we suggest that this would also be the case in SAA-enriched HDL from overweight and obese children; in support of this concept, we have shown that the activities of HDL2-CETP and HDL2-LCAT were also influenced in these overweight and obese cohorts.
CETP is an important enzyme that is involved in the remodeling of HDL and apo B lipoproteins (16,31). However, an increase in its activity is considered proatherogenic because it can reduce HDL concentrations and influence adipocyte morphology (16,32,33). Therefore, our results suggest that these overweight and obese children were exposed to a similar CETP-mediated proatherogenic phenotype to that of overweight and obese adults, which may be negated by weight loss as documented in adults (34).
With regard to LCAT, an increase in the activity of this enzyme is seen as antiatherogenic because it is responsible for esterifying free cholesterol within pre-β–HDL and HDL3, with HDL2 being the final mature product. Therefore, LCAT is more closely associated with the former HDL particles, whereas HDL2 contains minimum LCAT levels. However, this is not the case in HDL2 from obese adults, where in fact LCAT is increased, which suggests that these subjects have more immature HDL2 particles than are found in lean subjects (35). Therefore, our results support this finding; we also showed that LCAT’s activity was increased in HDL2 from both the overweight and obese children vs. their lean comparators, therefore suggesting that their HDL2 was more immature. However, this finding is not consistent with the concept that LCAT’s activity is normally reduced in SAA-enriched HDL (28), which suggests that obesity, independent of SAA, had influenced the activity of LCAT. However, this anomaly would require further investigation, and a mass assay for LCAT, alongside this activity assay may be more informative.
Overall, this study has provided evidence that children who are overweight or obese are exposed to an inflammatory milieu that impacts functional aspects of HDL2&3 and would potentially increase their CVD risk. Therefore, the growing epidemic of childhood obesity needs to be addressed to help prevent premature mortality in later life.
Methods
Study Population
The cohort utilized in this analysis has been described elsewhere (23). In brief, the Young Hearts 2000 study was a cross-sectional study that took place between 1999 and 2001. A total of 2,017 subjects, aged 12 and 15 y, were recruited from 36 nationally representative schools from across Northern Ireland. Height and weight measurements were used to assess BMI (kg/m2), and this was used to classify each subject as normal-weight, overweight, or obese on the basis of age- and sex-specific cut-off points for BMI linked to adult cut-off points (36) ( Table 4 ). The prevalence of overweight and obese subjects in the complete cohort was 16.2 and 4.7%, respectively. All of the obese adolescents were selected (95 subjects). However, due to the inability to obtain blood from three of these subjects, the final subject number was reduced to 92. These were then matched according to age, sex, and smoking status with 92 overweight and 92 normal-weight children.
Ethical approval was obtained from the Research Ethics Committee of Queens University Belfast. Written informed consent was signed by the participants and the participants’ parent or guardian. All investigations conformed to the principles in the Declaration of Helsinki.
Blood Sampling
Fasting peripheral venous blood was obtained by venipuncture and collected into serum or K2-EDTA tubes. Serum and plasma were harvested following centrifugation at 1,100g for 10 min at 4 °C within 4 h of collection.
Lipid Analyses
Serum total cholesterol, HDL cholesterol, and triglycerides were determined using enzymatic assays (Boehringer, Mannheim, Germany) on a Cobas Fara-automated analyzer (Roche Diagnostics, West Sussex, UK).
Isolation of HDL2 and HDL3
HDL2&3 were isolated from plasma by rapid ultracentrifugation according to a method previously established in our laboratory (37), which is a three-step, 6-h procedure. First, crude HDL was isolated by a 2-h rapid sedimentation method, second, this crude HDL was subfractionated into HDL2&3 by two separate 2-h sequential rapid flotation ultracentrifugation procedures.
C-Reactive Protein Concentration
C-reactive protein has previously been reported (23), but for comparison with SAA, its results are presented here. This was measured in plasma by a latex-enhanced immune-turbidimetric assay (Wako Chemicals, Neuss, Germany) using a Cobas Fara automated analyzer.
SAA Concentration
SAA, in serum and associated with HDL2&3, was analyzed in duplicate by an enzyme-linked immunosorbent assay (Invitrogen, Paisley, UK) per the manufacturer’s instructions. Analysis was performed on a TRITURUS-automated ELISA system (Grifols, Ghezzano Pisa, Italy).
CETP and LCAT Activity
The activity of CETP and LCAT was measured in duplicate in serum and in HDL2&3 using commercially available fluorometric assays per manufacturer’s instructions (CETP and LCAT; Roar Biomedical, New York, NY).
Total Protein Determination
The protein concentration of HDL2&3 was determined in duplicate by a spectrophotometric assay, as previously described (38). Total protein concentration was utilized to standardize SAA, CETP, and LCAT within HDL2&3.
Statistical Analysis
Statistics were completed using the statistical software package SPSS for Windows, version 17 (SPSS, Chicago, IL). Variables were assessed for normality and logarithmically transformed where required. Differences between BMI groups were assessed by a one-way ANOVA followed by the Newman–Keuls multiple range comparison test when the data were normally distributed, or by the Kruskal–Wallis test when the data were not normally distributed, even after logarithmic transformation. Differences between the BMI groups in categorical variables were assessed using χ2 test. Correlations between HDL2&3-SAA levels and the activities of LCAT and CETP within HDL2&3 were assessed using Pearson’s correlation coefficient. All variables were summarized as mean and SD when normally distributed, as geometric mean (interquartile range) when normally distributed after logarithmic transformation, and as median (interquartile range) when logarithmic transformation did not normalize the distribution. The significance level was set as P < 0.05.
Statement of Financial Support
This work was supported by the Department for Employment and Learning, All Island Fund, UK. The original Young Hearts 2000 was supported by the British Heart Foundation, UK, and the Wellcome Trust, UK.
References
Lloyd-Jones D, Adams R, Carnethon M, et al.; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics–2009 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 2009;119:e21–181.
Forman D, Bulwer BE . Cardiovascular disease: optimal approaches to risk factor modification of diet and lifestyle. Curr Treat Options Cardiovasc Med 2006;8:47–57.
Jones PH . Management of obesity in the prevention of cardiovascular disease. Methodist Debakey Cardiovasc J 2010;6:33–6.
Kipping RR, Jago R, Lawlor DA . Obesity in children. Part 1: Epidemiology, measurement, risk factors, and screening. BMJ 2008;337:a1824.
Splansky GL, Corey D, Yang Q, et al. The Third Generation Cohort of the National Heart, Lung, and Blood Institute’s Framingham Heart Study: design, recruitment, and initial examination. Am J Epidemiol 2007;165:1328–35.
McGill HC Jr, McMahan CA, Herderick EE, Malcom GT, Tracy RE, Strong JP . Origin of atherosclerosis in childhood and adolescence. Am J Clin Nutr 2000;72:Suppl 5:1307S–15S.
Lloyd LJ, Langley-Evans SC, McMullen S . Childhood obesity and adult cardiovascular disease risk: a systematic review. Int J Obes (Lond) 2010;34:18–28.
Shoelson SE, Herrero L, Naaz A . Obesity, inflammation, and insulin resistance. Gastroenterology 2007;132:2169–80.
Ogasawara K, Mashiba S, Wada Y, et al. A serum amyloid A and LDL complex as a new prognostic marker in stable coronary artery disease. Atherosclerosis 2004;174:349–56.
Zhao Y, He X, Shi X, et al. Association between serum amyloid A and obesity: a meta-analysis and systematic review. Inflamm Res 2010;59:323–34.
Yang RZ, Lee MJ, Hu H, et al. Acute-phase serum amyloid A: an inflammatory adipokine and potential link between obesity and its metabolic complications. PLoS Med 2006;3:e287.
Baranova IN, Vishnyakova TG, Bocharov AV, et al. Serum amyloid A binding to CLA-1 (CD36 and LIMPII analogous-1) mediates serum amyloid A protein-induced activation of ERK1/2 and p38 mitogen-activated protein kinases. J Biol Chem 2005;280:8031–40.
Badolato R, Wang JM, Murphy WJ, et al. Serum amyloid A is a chemoattractant: induction of migration, adhesion, and tissue infiltration of monocytes and polymorphonuclear leukocytes. J Exp Med 1994;180:203–9.
Furlaneto CJ, Campa A . A novel function of serum amyloid A: a potent stimulus for the release of tumor necrosis factor-alpha, interleukin-1beta, and interleukin-8 by human blood neutrophil. Biochem Biophys Res Commun 2000;268:405–8.
Artl A, Marsche G, Lestavel S, Sattler W, Malle E . Role of serum amyloid A during metabolism of acute-phase HDL by macrophages. Arterioscler Thromb Vasc Biol 2000;20:763–72.
Park KH, Shin DG, Kim JR, Hong JH, Cho KH . The functional and compositional properties of lipoproteins are altered in patients with metabolic syndrome with increased cholesteryl ester transfer protein activity. Int J Mol Med 2010;25:129–36.
Cho KH . Biomedicinal implications of high-density lipoprotein: its composition, structure, functions, and clinical applications. BMB Rep 2009;42:393–400.
Pirkola J, Vääräsmäki M, Ala-Korpela M, et al. Low-grade, systemic inflammation in adolescents: association with early-life factors, gender, and lifestyle. Am J Epidemiol 2010;171:72–82.
Beauloye V, Zech F, Tran HT, Clapuyt P, Maes M, Brichard SM . Determinants of early atherosclerosis in obese children and adolescents. J Clin Endocrinol Metab 2007;92:3025–32.
Herder C, Schneitler S, Rathmann W, et al. Low-grade inflammation, obesity, and insulin resistance in adolescents. J Clin Endocrinol Metab 2007;92:4569–74.
Balagopal P, George D, Patton N, et al. Lifestyle-only intervention attenuates the inflammatory state associated with obesity: a randomized controlled study in adolescents. J Pediatr 2005;146:342–8.
Balagopal PB, de Ferranti SD, Cook S, et al.; American Heart Association Committee on Atherosclerosis Hypertension and Obesity in Youth of the Council on Cardiovascular Disease in the Young; Council on Nutrition, Physical Activity and Metabolism; Council on Epidemiology and Prevention. Nontraditional risk factors and biomarkers for cardiovascular disease: mechanistic, research, and clinical considerations for youth: a scientific statement from the American Heart Association. Circulation 2011;123:2749–69.
Wijinstok NH, Twisk JWR, Young IS, et al. Inflammation markers are associated with cardiovascular diseases risk in adolescents: The Young Hearts Project 2000. J Adolesc Health 2010;47:346–51.
Economou EV, Malamitsi-Puchner AV, Pitsavos CP, Kouskouni EE, Magaziotou-Elefsinioti I, Creatsas G . Low-grade systemic inflammation profile, unrelated to homocysteinemia, in obese children. Mediators Inflamm 2005;2005:337–42.
Neuman G, Sagi R, Shalitin S, Reif S . Serum inflammatory markers in overweight children and adolescents with non-alcoholic fatty liver disease. Isr Med Assoc J 2010;12:410–5.
Gómez-Ambrosi J, Azcona C, Patiño-García A, Frühbeck G . Serum Amyloid A concentration is increased in obese children and adolescents. J Pediatr 2008;153:71–5.
Poitou C, Coussieu C, Rouault C, et al. Serum amyloid A: a marker of adiposity-induced low-grade inflammation but not of metabolic status. Obesity (Silver Spring) 2006;14:309–18.
Van Lenten BJ, Hama SY, de Beer FC, et al. Anti-inflammatory HDL becomes pro-inflammatory during the acute phase response. Loss of protective effect of HDL against LDL oxidation in aortic wall cell cocultures. J Clin Invest 1995;96:2758–67.
Brites FD, Bonavita CD, De Geitere C, et al. Alterations in the main steps of reverse cholesterol transport in male patients with primary hypertriglyceridemia and low HDL-cholesterol levels. Atherosclerosis 2000;152:181–92.
Kontush A, de Faria EC, Chantepie S, Chapman MJ . Antioxidative activity of HDL particle subspecies is impaired in hyperalphalipoproteinemia: relevance of enzymatic and physicochemical properties. Arterioscler Thromb Vasc Biol 2004;24:526–33.
Tall AR . Plasma cholesteryl ester transfer protein. J Lipid Res 1993;34:1255–74.
Kontush A, Chapman MJ . Functionally defective high-density lipoprotein: a new therapeutic target at the crossroads of dyslipidemia, inflammation, and atherosclerosis. Pharmacol Rev 2006;58:342–74.
Zhou H, Li Z, Hojjati MR, et al. Adipose tissue-specific CETP expression in mice: impact on plasma lipoprotein metabolism. J Lipid Res 2006;47:2011–9.
Asztalos BF, Swarbrick MM, Schaefer EJ, et al. Effects of weight loss, induced by gastric bypass surgery, on HDL remodeling in obese women. J Lipid Res 2010;51:2405–12.
Magkos F, Mohammed BS, Mittendorfer B . Plasma lipid transfer enzymes in non-diabetic lean and obese men and women. Lipids 2009;44:459–64.
Cole TJ, Bellizzi MC, Flegal KM, Dietz WH . Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ 2000;320:1240–3.
McPherson PA, Young IS, McKibben B, McEneny J . High density lipoprotein subfractions: isolation, composition, and their duplicitous role in oxidation. J Lipid Res 2007;48:86–95.
McEneny J, McMaster C, Trimble ER, Young IS . Rapid isolation of VLDL subfractions: assessment of composition and susceptibility to copper-mediated oxidation. J Lipid Res 2002;43:824–31.
Acknowledgements
I.S.Y., L.M., and C.B. were involved in the original Young Hearts 2000 study, which provided the samples for this additional analysis. High-sensitive C-reactive protein has been reported in the original analysis of this study (Wijistok NH et al., J Adoles Health 2010;47:337–42) and is included here to compare with serum amyloid-A.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McEneny, J., Blair, S., Woodside, J. et al. High-density lipoprotein subfractions display proatherogenic properties in overweight and obese children. Pediatr Res 74, 279–283 (2013). https://doi.org/10.1038/pr.2013.93
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pr.2013.93
This article is cited by
-
Anthracycline-induced cardiotoxicity: targeting high-density lipoproteins to limit the damage?
Lipids in Health and Disease (2022)
-
Increased Cholesteryl Ester Transfer Protein and Lipoprotein-Associated Phospholipase A2 Activities in Children and Adolescents Presenting High Triglyceride/High-Density Lipoprotein Cholesterol (TG/HDL-C) Ratio
Indian Journal of Pediatrics (2021)