Abstract
Despite extensive research, the pathogenesis of neonatal necrotizing enterocolitis (NEC) remains elusive. The aim of our work was to investigate the role of bacterial strains involved in NEC in gnotobiotic quails as experimental model. Six groups of germ-free quails that were fed a lactose diet were associated with Klebsiella pneumoniae, Clostridium perfringens, C. difficile, C. paraputrificum, or C. butyricum (two strains). Implantation level, incidence of cecal lesions, production of short-chain fatty acids, and histologic lesions of the cecal wall were investigated. Whatever the strain, the implantation level was high (109 UFC/g). Neither K. pneumoniae nor C. difficile induced any cecal lesions. In contrast, the four other clostridial strains led to cecal NEC-like lesions with a variable occurrence: four of 12 quails for C. perfringens, eight of 12 quails for C. paraputrificum, and the same highest value, nine of 12 quails and eight of 10 quails for both C. butyricum strains. Gross aspects of the lesions may be linked to the short-chain fatty acid profiles and/or concentrations: thickening of the cecal wall (C. butyricum and C. perfringens) with high proportion of butyric acid, hemorrhages (C. paraputrificum) with high proportion of iso-butyric acid, and presence of other iso-acids. In addition, C. butyricum was characterized by pneumatosis, linked to a high gas production. Microscopic aspects confirmed the presence of edemas and intramucosa hemorrhages. Clostridia species, whose role is controversial, seem to be strongly implicated in NEC through excessive production of butyric acid as a result of colonic lactose fermentation. These results call for anaerobe detection in feces of infants who have NEC.
Similar content being viewed by others
Main
Necrotizing enterocolitis (NEC) is a devastating disease with high morbidity and mortality and the most common gastrointestinal emergency encountered in the neonatal period, affecting predominantly preterm infants. Despite extensive research, the exact pathogenesis remains incompletely understood. Three major factors—bacterial colonization, enteral feeding, and gut immaturity—are recognized to coalesce for promoting an inflammatory cascade that leads to the disease (1–3). Digestive bacterial colonization seems to be a prerequisite. In fact, NEC occurred neither before colonization of the intestine by bacteria nor in germ-free animal models. Furthermore, if NEC usually occurs sporadically, then reports of outbreaks have encouraged speculations about specific transmissible agents. Implication of bacteria is thought to be due to fermentation of nonhydrolyzed lactose, a consequence of the immaturity of the intestinal lactasic equipment in preterm infants (2). High production of bacterial metabolites (hydrogen, butyric, and iso-butyric acids) may be responsible for the onset of the intestinal lesions and for pneumatosis (4–6).
Various microorganisms are involved in NEC, including bacteria and virus [see Foglia (3) for review]; however, no specific pathogens have been found (7). The most often implicated bacteria are enterobacteria, particularly Klebsiella pneumonia and Escherichia coli, but also anaerobes species, specifically clostridia species. The most common clostridia species involved are C. butyricum and C. perfringens. C. paraputrificum was scarcely identified. Recently, a novel Clostridium species, C. neonatale, was associated with a NEC outbreak (8). All of these bacteria belong to the endogenous intestinal microflora, and the aberrant gut colonization observed in preterm infants (9,10) may be a risk factor (2). Analyses of the premature infant's fecal flora by molecular techniques were seldom used. Millar et al. (11), who compared preterm infants with and without NEC, did not observed any difference. In contrast, De La Cochetière et al. (12) described recently a significant relationship between early colonization by C. perfringens and the later development of NEC. However, the causative role of clostridia remains controversial.
Gnotobiotic quails that were fed a lactose diet were previously used as a suitable experimental model to mimic human premature neonates in terms of gastrointestinal and physiologic characteristics, because they are a natural alactasic species and their ceca, a pair of blind ending ducts, favors bacterial stasis (13,14). Development of cecal NEC-like lesions in quails requires the combination of two major factors that are present in preterm human infants: lactose in diet and colonization by lactose-fermenting bacteria. Indeed, experimental infection of germ-free quails with either C. butyricum strains (monobiotic quails) or fecal specimens (polybiotic quails), both originating from preterm infants with NEC, reproduced many aspects of the pathology, such as thickening of the cecal wall with gas cysts, hemorrhagic ulcerations, necrotic areas, and intestinal pneumatosis (13–16). Up to now, only the key role of C. butyricum was clearly demonstrated in monobiotic quails associated with strains from various origins (13,14). Polybiotic quails were associated with fecal specimens that comprise several bacterial species implicated in NEC: C. perfringens, C. paraputrificum, C. difficile, and K. pneumoniae (15,16). However, these species were also isolated from healthy preterm infants.
The aim of the present study was to examine and compare the role of these bacterial species isolated from NEC cases—C. perfringens, C. paraputrificum, C. difficile, and K. pneumoniae—in our experimental model. With the use of monobiotic quails, the involvement of these bacterial species was studied through the bacterial implantation, the incidence of intestinal lesions, and the production of short-chain fatty acids (SCFAs) and compared with two C. butyricum strains isolated from infants with NEC. In case of cecal lesions, histologic examinations were performed.
METHODS
Bacterial strains.
Six bacterial strains that were isolated from preterm infants who had NEC and were hospitalized in different French pediatric units were studied (Table 1). All infants had definite or advanced NEC (Bell's stage IIc, IIIa, or IIIb). K. pneumoniae was isolated from an infant (NEC-stage IIIa) during an outbreak of NEC (12 cases). C. perfringens, C. difficile, and C. paraputrificum were isolated from fecal specimens of two preterm infants who had NEC (stage IIc) (15). C. butyricum MPP 195 and C. butyricum CB 1002–5 were isolated from two fatal NEC cases (stage IIIb). C. butyricum CB 1002–5 was included as a reference strain that produced NEC-like lesions in quails (13).
Experimental design.
Germ-free quails (Coturnix coturnix subspecies japonica) were obtained and maintained in germ-free isolators as already described (13). Quails were fed an experimental semisynthetic diet that contained lactose 7% (wt/wt) to mimic the proportion of lactose in human milk. The diet was sterilized by gamma irradiation at 40 kGy. After their germ-free status was checked, 2-wk-old germ-free quails were transferred in six experimental isolators (n = 7 to 12 quails per group) and mono-associated with the different bacterial strains. Each group was inoculated orally with 100 μL of cultures that contained 108 viable cells of one of the various strains. Bacterial establishment was checked in fresh droppings at weekly intervals. All quails were killed 3 wk after the bacterial inoculation.
Sampling.
Ceca were collected immediately after death. After macroscopic examinations, the cecal contents were removed for pH measurement and bacteriologic counts. For biochemical determinations, samples of cecal contents were immediately frozen with saturated mercuric chloride solution (10%, vol/vol) before analysis. Cecal wall samples were placed in a 10% formalin solution for histologic examinations.
Bacterial counts.
Cecal contents were homogenized and diluted in peptone broth for bacterial counts. Using the Spiral System (F-35270; AES Laboratoires, Combourg, France), dilutions were spread on Columbia agar media with 5% sheep blood for anaerobic bacteria and incubated for 48 h at 37°C in jars under an anaerobic gas phase (H2:CO2, 95:5, vol/vol). For K. pneumoniae, the dilutions were spread on Trypcase agar and incubated in aerobic atmosphere for 24 h. Bacterial counts were expressed as the log10 CFU/g of wet feces or cecal content.
Biochemical determination.
SCFA determinations and dosage were performed on in vitro cultures of the six bacterial strains and on the cecal contents. In case of in vitro experiments, bacteria were grown in TYH broth that contained lactose 1% (vol/vol) for 48 h in anaerobic or aerobic atmosphere, depending on the strain. SCFA analysis were carried out on a Perkin-Elmer Autosystem Gas Chromatography (St Quentin en Yvelines, France) fitted with a capillary (15 m × 0.53 mm) Nukol column (Supelco, St Germain-en-Laye, France) and a flame ionization detector as previously described (15). 2-Ethyl butyric acid (Sigma Chemical Co., L'Isle d'Abeau, France) was used as internal standard, and total SCFA concentrations were calculated in μmol/L or μmol/g of wet cecal content. The relative distribution of acetic (C2), propionic (C3), butyric (C4), valeric (C5), caproic (C6), and iso-acids (iC4, iC5, and iC6) was calculated as the percentage of the total concentration. pH was measured when the fecal specimen quantity was sufficient (>300 mg).
Macroscopic and Histologic Examinations of the Cecal Wall
Gross findings.
Ceca were categorized as normal, thickening, pneumatosis, and hemorrhagic contents. The weight of the cecal wall was expressed as the cecal wall weight:body weight ratio. Quails with normal ceca were referred to as healthy quails, and quails with one or more cecal modifications (thickening, pneumatosis, or hemorrhagic content) were referred to as sick quails.
Microscopic findings.
All of the specimens were fixed in formalin, then embedded in paraffin, serially sectioned, and stained with hematoxylin and eosin. The elementary histologic lesions were studied and classified as follows: erosion/ulceration, edema, foci of necrosis, infiltrates, gas cysts, and peritonitis. Biochemical determinations and macroscopic and histologic examinations were performed blinded to the investigators.
Statistical analysis.
Data presented as the mean ± SEM were evaluated statistically by the t test (cecal pH, cecal wall weight) and the Mann-Whitney nonparametric test (SCFA concentrations and profiles).
RESULTS
In vitro bacterial SCFA profiles.
SCFA concentrations varied from 11 to 68 mM/L, depending on the strain (Table 2). The production of SCFAs by K. pneumoniae consisted mainly of acetate, and gas production was moderate. C. difficile differed from the other species by a high production of SCFAs, a complex SCFA profile, and an absence of gas production. The four other clostridia strains shared a similar butyric acid profile and an abundant gas production. Among them, only C. perfringens produced propionic acid in a small amount.
In vivo experimental data.
The strains were established at a high level in the six groups of the monobiotic quails, ranging from 8.5 ± 0.2 to 9.3 ± 0.4 CFU/g of cecal content (Table 3). K. pneumoniae did not induce any cecal lesions. Among clostridia species, only C. difficile did not induce cecal lesions. In contrast, the four other clostridia strains led to cecal NEC-like lesions in quails with a variable occurrence: four (50%) of eight quails for C. perfringens, eight (67%) of 12 quails for C. paraputrificum, and the same highest value nine (75%) of 12 quails and eight (80%) of 10 quails for both C. butyricum strains.
Cecal contents in non–lesion-inducing strains.
The two strains differed from one another in cecal pH and SCFAs. With K. pneumoniae, SCFA concentrations were twice as high than with C. difficile and associated with a lower pH (Table 3). The C. difficile profile was more complex than the K. pneumoniae profile, with a significantly higher percentage of propionic, valeric, isocaproic, and caproic acids (Table 4).
Cecal contents in NEC-like lesion–inducing strains.
pH values were not significantly different between groups whatever the bacterial status or pathologic state (Table 3). Total SCFA concentrations were higher with C. perfringens and C. paraputrificum groups than with both C. butyricum groups. The relative contribution in the total SCFA amounts differed with the strain. As far as the profile was concerned, percentages of butyric acid were significantly higher in both C. butyricum groups as compared with the other groups, and percentages of iso-butyric acid were significantly higher in both C. perfringens and C. paraputrificum groups. Small amounts of other SCFAs were detected in these last two groups (Table 4). There was no significant difference in SCFA profiles between sick (i.e. with cecal lesions) and healthy quails (i.e. without lesions) within the same bacterial association.
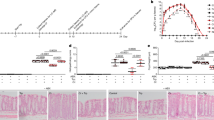
Pathologic findings were hemorrhagic fluid, predominant in the lumen of C. paraputrificum group (seven of the eight sick quails), and to a lesser extent in the three other clostridia groups (Fig. 1A).
Macroscopic (A) and microscopic (B) observations in quails associated with NEC-like lesion–inducing strains and displaying cecal lesions. T, thickening; H, hemorrhages; P, pneumatosis; L, lymphocytes; PN, heterophils and polynuclear cells; IH, intramucosal hemorrhages; E, edema; N, necrotic areas. Histologic examinations were not performed with C butyricum MPP195.
Cecal wall in non–lesion-inducing strains.
In quails associated with K. pneumoniae or C. difficile, the cecal wall weight to body weight ratio did not exceed 4.6 and 3.6, respectively (Table 3).
Cecal wall in NEC-like lesion–inducing strains.
Within the same bacterial status, cecal wall weight was significantly enhanced in sick quails as compared with healthy ones for the C. perfringens group (p = 0.01) and both C. butyricum groups (p = 0.03; Table 3). No thickening of the cecal wall was observed in the C. paraputrificum group.
The macroscopic pathologic findings were thickening and pneumatosis of the mucosa. Both C. butyricum strains led to the most dramatic effects, from thickening to the association of thickening and pneumatosis with or without hemorrhagic contents (Fig. 1A). In the case of C. perfringens, because of the small number of sick animals, a specific profile of lesions was difficult to identify. Thickening was very high, except for one sick quail, and associated with either pneumatosis or hemorrhage. In quails associated with C. paraputrificum, only one quail displayed thickening and pneumatosis.
Microscopic findings were necrotic areas, hemorrhages, and edema with infiltrate of mononuclear cells as macrophages, lymphocytes, and heterophils combined with polynuclear cells, and alteration in the brush border (Fig. 1B). C. butyricum led to the most dramatic alterations of both cecal mucosa and cecal brush border. Brush border was characterized by edema and villus atrophy. A hypertrophy of the cecal wall was observed to be associated with a large inflammatory infiltrate in the mucosa, submucosa, musculosa, and serous membrane. Gas cysts represented another common finding (Fig. 2A and B). In quails associated with C. paraputrificum, hemorrhages predominated (Fig. 2C and D), and necrotic areas that were observed in four quails were very scarce as compared with the C. perfringens and C. butyricum groups.
Various histologic examinations of the ceca of monobiotic quails (hematoxylin-eosin stain). (A and B) Cecum of quails monoassociated with C. butyricum CB 1002–5. Numerous gas cysts and infiltrates are in the submucosa of the cecum. (C and D) Cecum of quails monoassociated with C. paraputrificum. Pleomorphic infiltrates are in the cecal wall and hemorrhages. Bars = 100 μ. Magnification: ×100 in A and C; × 400 in B and D.
SCFA profiles and pathologic aspects.
Monobiotic quails associated with NEC-like lesion–inducing strains were characterized by significantly higher levels of either butyric or butyric and iso-butyric acids (Table 5). Moreover, the trend of major macroscopic aspects of the lesions may be linked to the major differences in SCFA profiles. Thickening (C. butyricum and C. perfringens) was linked with a higher proportion of butyric acid, whereas hemorrhages (C. paraputrificum) were linked with high proportion of iso-butyric acid and presence of other iso-acids (Table 4). In addition, the C. butyricum group was characterized by pneumatosis, linked to the high gas production by this strain.
DISCUSSION
The important role of enteral feeding and bacterial colonization in the pathogenesis of NEC has been well recognized for several years. However, no specific pathogens were found. Using an experimental model of NEC, i.e., monobiotic quails associated with bacterial species belonging to preterm neonates with NEC, we demonstrated the implication of clostridial species through the bacterial fermentation, except for C. difficile, and showed that K. pneumoniae did not interfere.
Klebsiella species is the most frequent aerobic species implicated in NEC, but most microbiologic analyses of NEC cases involving Klebsiella did not include anaerobic microflora cultures (17,18). Westra-Meijer et al. (19), comparing aerobic and anaerobic fecal flora in healthy preterm infants and infants with NEC, observed a higher colonization with Klebsiella species in infants with NEC but no difference in clostridia populations. Nevertheless, a careful analysis of the published results showed that the proportion of infants who were colonized with high levels of clostridia (≥108/g of feces) was higher in the two NEC groups (54%) than in the healthy group (25%). Moreover, the number of clostridia species, except C. difficile, that were isolated in the group with proved NEC (12 species isolated in 24 infants) was higher than in the suspected NEC groups (three species in 12 infants) and in the healthy group (14 species in 41 infants). In a clinical report of 12 cases of NEC, mild to moderate clinical course was associated with isolation of Klebsiella species, whereas severe disease was associated with the presence of clostridia (20).
Concerning C. difficile, our results with monobiotic quails were in accordance with our previous studies on polybiotic quails, which did not allow implication of C. difficile (15). In these studies, in quails associated with fecal specimens of premature neonates comprising C. difficile, a marked protective role by bifidobacteria through a sharp decrease in clostridia levels was demonstrated; however, both C. difficile counts and its toxins were not modified (mean 2.5 × 105 UFC/g). Our present results improve the understanding of the controversial role of this species commonly found in neonates, even in preterm infants (21). Indeed, in contrast to adults, high levels of toxins A and B may be detected in asymptomatic neonates (21). In studies involving C. difficile in NEC (22,23), the microbiologic methods performed were unable to isolate anaerobic bacteria other than C. difficile. Moreover, several authors did not find any relation between the isolation of C. difficile and/or detection of its toxins and NEC (21,24,25). In Westra-Meijer's study (19), C. difficile colonization was similar in the NEC groups and the healthy group (∼50%). However, in very rare cases, C. difficile might be involved in severe enterocolitis in infants, the most obvious relationship being in regards to enterocolitis in Hirschsprung's disease (26).
The three other clostridial species studied led to NEC-like digestive lesions in quails. The effects varied in terms of occurrence and seriousness of the cecal lesions according to the species, even implanted at the same high level. The most deleterious effects were observed with C. butyricum strains. The key role of C. butyricum in NEC development has been once again suggested through our experimental model. Cecal lesions, levels of bacterial implantation, and SCFA profile between quails that were infected with either the strain of C. butyricum (VPI 3266) that was isolated from the autochthonous microflora of pigs or various C. butyricum strains isolated from healthy neonates (three strains) or from neonates with NEC (three strains) in previous studies (13,14) were similar. In the present study, a new isolate of C. butyricum (MPP195), originating from a fatal NEC case, led to the same cecal lesions in monobiotic quails. Lesions were macroscopically and microscopically similar to that in neonates with NEC (3). Grossly, the lesions were a mural thickening of the bowel, with ulcerations, necrosis, and pneumatosis, which is a characteristic finding in NEC. Microscopically, an inflammatory reaction that consisted of lymphocytes and neutrophils was observed in the mucosa, with hemorrhages and edema of the submucosa. In severe lesions, necrotic areas and ulcerations were observed. Last, gas-filled cysts were often present in the bowel wall.
The role of this autochthonous nontoxigenic species was largely discussed 25 y ago [reviewed by Szylit et al. (14)]. Our experimental findings in previous studies showed its primary role in the onset of NEC-like lesions in quails through the carbohydrate fermentation products and the production of butyric acid. When quails were fed a diet deprived of lactose, butyric acid production was very low, whereas clostridia implantation was at a similar level and no digestive lesions were observed (13,14). Numerous gas cysts were always observed in the cecal wall of quails monoassociated with C. butyricum (13). The pathogenesis of the gas cysts probably consists of an accumulation of gas produced by bacteria in the cecal lumen through rupture of the epithelium integrity or through spaces between epithelium cells. Gas cysts in the mucosa were shown by other authors to consist largely of hydrogen produced by the proliferation of clostridia organisms (27).
C. perfringens, which also belongs to the autochthonous microflora in adults and neonates, was often associated with NEC [see Foglia (3) for review]. Moreover, NEC cases associated with C. perfringens had more severe clinical course (20). In our model, the main difference between C. perfringens– and C. butyricum–induced lesions was the lower occurrence of pneumatosis and intramucosal edema with C. perfringens. The intestinal damage and hemorrhages could be the result of the production of the numerous C. perfringens toxins. However, NEC was not linked to the production of the alpha toxin (28). Moreover, compared with C. butyricum, butyric acid is also a major end product of lactose fermentation by C. perfringens, and when quails were fed a diet deprived of lactose, no lesion was observed (16).
C. paraputrificum, like C. butyricum, may be found in healthy neonates (19,29,30) and does not produce any toxin. It is a lactose-fermenter species that produces acetic and butyric acids. Contrary to C. butyricum and to C. perfringens, C. paraputrificum did not induce any cecal wall thickening but led to intramucosa and intraluminal hemorrhages. Up to now, C. paraputrificum was scarcely studied as a NEC agent. One reason could be that usual bacteriologic methods do not allow growth of all anaerobes, which require specific methods that often are technically tedious. Recently, because a point-prevalence survey using rectal swabs was conducted, a NEC associated with C. paraputricum was diagnosed in one infant (8).
Our study confirms the role of bacterial fermentation end products, i.e. SCFAs and in particular butyric acid, in the onset of NEC-like lesions observed in our experimental model. However, implication of butyric acid in NEC is controversial. In fact, SCFAs and especially butyric acid are essential for the integrity of the colonic epithelium. Butyric acid stimulates the proliferation of the epithelial colonic cells and is the major source of energy for the enterocytes (31), but high concentrations can lead to deleterious effects. In vivo, high concentrations were shown to lower the pH of small intestine, causing mucosal breakdown and increasing susceptibility to bacterial translocation (32). Furthermore, gut luminal administration of acetic or butyric acid in newborn rats produced dose-dependant intestinal mucosal injury, resembling human NEC clinically and histologically (33). Deleterious effect of butyric acid may be a result of an inflammatory response to this bacterial fermentation end product. Indeed, butyric acid was demonstrated to increase IL-8 secretion in cultured enterocytes (34). In human pathology, IL-8 production has been observed in numerous digestive infections such as Shigella in colon (35). As an α-chemokine, IL-8 is a potent chemotactic factor for neutrophils and may be important in recruiting leucocytes into the gastrointestinal tract during inflammation (36). IL-8 also stimulates superoxide radical release in neutrophils and increases the permeability of vascular endothelium leading to tissue edema (36). In infants, high production of SCFAs associated with abnormal C4 ratio (above 40%) has previously been demonstrated as potentially deleterious (5). In premature infants, because of their potential deficiency in intestinal lactase, butyric acid can be overproduced in the distal ileum and colon and thus cause intestinal injury as hypothesized by Lin (6). Furthermore, SCFA concentrations in healthy extremely premature infants were very low as compared with premature infants and full-term infants, and butyric acid concentrations varied from 0 to 1.2 μmol/g of feces, suggesting a different relationship between SCFAs and the intestinal mucosa in preterm infants (37).
Furthermore, our results pointed out a relationship between SCFA production and the aspects of the lesions: thickening was associated with a high level of butyric acid and hemorrhages with a high level of iso-butyrate. Fermentation into butyric acid and/or iso-butyric acid seemed to be a prerequisite to the pathologic change. Moreover, in a previous study, the protective role of bifidobacteria through a decrease in clostridia level was associated with a dramatic decrease or a disappearance in butyric acid (15). The presence of iso-butyric acid showed also that these clostridia species were implicated in proteolysis, the end products of bacterial protein degradation known to be implicated in the intraluminal pathogenesis of NEC (38).
Last, the fermentation ability is likely to predominate over the characteristic of the strain itself. In fact, Carbonaro et al. (32) suggested that the increased ability for lactose fermentation of a K. pneumoniae strain and the ensuing production of SCFAs may be a factor in the onset of NEC. Such a peculiar strain was isolated from an infant with NEC, but other investigators never found a difference in β-galactosidase activity in Gram-negative bacteria that were isolated from 23 neonates with NEC (39).
Anaerobic research in fecal specimen of premature neonates, when performed, is frequently limited to C. difficile. The present microbiologic results, strengthened by the recently described outbreak of NEC associated with C. neonatale (8) and the relationship observed between early colonization by C. perfringens and NEC (12), call for the development of clostridia detection in stools of infants who are suspected of having NEC. This finding is in accordance with the conclusions of Duffy et al. (40) that Clostridium species were the most common bacterial pathogen associated with NEC in their study (p < 0.05). Thus, endogenous digestive flora, even in the absence of any known pathogenic factor, plays an ambiguous role toward the host. Excessive concentration of bacterial metabolites released during lactose fermentation—a consequence of the immaturity of the lactasis equipment—may have tremendous effects on mucosal integrity. The SCFA detection in feces might help notice a change in colonic microflora leading to the onset of the disease.
Abbreviations
- NEC:
-
neonatal necrotizing enterocolitis
- SCFA:
-
short-chain fatty acid
References
Caplan MS, Jilling T 2001 New concepts in necrotizing enterocolitis. Curr Opin Pediatr 13: 111–115
Claud EC, Walker WA 2001 Hypothesis: inappropriate colonization of the premature intestine can cause neonatal necrotizing enterocolitis. FASEB J 15: 1398–1403
Foglia RP 1995 Necrotizing enterocolitis. Curr Probl Surg 32: 757–823
Kien CL 1990 Colonic fermentation of carbohydrate in the premature infant: possible relevance to necrotizing enterocolitis. J Pediatr 117: S52–S58
Szylit O, Maurage C, Gasqui P, Popot F, Favre A, Gold F, Borderon JC 1998 Fecal short-chain fatty acids predict digestive disorders in premature infants. J Parenter Enteral Nutr 22: 136–141
Lin J 2004 Too much short chain fatty acids cause neonatal necrotizing enterocolitis. Med Hypotheses 62: 291–293
Peter CS, Feuerhahn M, Bohnhorst B, Schlaud M, Ziesing S, von der Hardt H, Poets CF 1999 Necrotising enterocolitis: is there a relationship to specific pathogens?. Eur J Pediatr 158: 67–70
Alfa MJ, Robson D, Davi M, Bernard K, Van Caeseele P, Harding GK 2002 An outbreak of necrotizing enterocolitis associated with a novel Clostridium species in a neonatal intensive care unit. Clin Infect Dis 35: S101–S105
Gewolb IH, Schwalbe RS, Taciak VL, Harrison TS, Panigrahi P 1999 Stool microflora in extremely low birthweight infants. Arch Dis Child Fetal Neonatal Ed 80: F167–F173
Schwiertz A, Gruhl B, Lobnitz M, Michel P, Radke M, Blaut M 2003 Development of the intestinal bacterial composition in hospitalized preterm infants in comparison with breast-fed, full-term infants. Pediatr Res 54: 393–399
Millar MR, Linton CJ, Cade A, Glancy D, Hall M, Jalal H 1996 Application of 16S rRNA gene PCR to study bowel flora of preterm infants with and without necrotizing enterocolitis. J Clin Microbiol 34: 2506–2510
de la Cochetière MF, Piloquet H, des Robert C, Darmaun D, Galmiche JP, Rozé JC 2004 Early intestinal bacterial colonization and necrotizing enterocolitis in premature infants: the putative role of Clostridium. Pediatr Res 56: 366–370
Bousseboua H, Le Coz Y, Dabard J, Szylit O, Raibaud P, Popoff MR, Ravisse P 1989 Experimental cecitis in gnotobiotic quails monoassociated with Clostridium butyricum strains isolated from patients with neonatal necrotizing enterocolitis and from healthy newborns. Infect Immun 57: 932–936
Szylit O, Butel MJ, Rimbault A 1997 An experimental model of necrotising enterocolitis. Lancet 350: 33–34
Butel MJ, Roland N, Hibert A, Popot F, Favre A, Tessèdre AC, Bensaada M, Rimbault A, Szylit O 1998 Clostridial pathogenicity in experimental necrotising enterocolitis in gnotobiotic quails and protective role of bifidobacteria. J Med Microbiol 47: 391–399
Butel MJ, Waligora-Dupriet AJ, Szylit O 2002 Oligofructose and experimental model of neonatal necrotising enterocolitis. Br J Nutr 87( suppl 2): S213–S219
Bell MJ, Feigin RD, Ternberg JL 1979 Changes in the incidence of necrotizing enterocolitis associated with variation of the gastrointestinal microflora in neonates. Am J Surg 138: 629–631
Gregersen N, Van Nierop W, Von Gottberg A, Duse A, Davies V, Cooper P 1999 Klebsiella pneumoniae with extended spectrum beta-lactamase activity associated with a necrotizing enterocolitis outbreak. Pediatr Infect Dis J 18: 963–967
Westra-Meijer CM, Degener JE, Dzoljic-Danilovic G, Michel MF, Mettau JW 1983 Quantitative study of the aerobic and anaerobic faecal flora in neonatal necrotising enterocolitis. Arch Dis Child 58: 523–528
Kosloske AM, Ulrich JA, Hoffman H 1978 Fulminant necrotising enterocolitis associated with clostridia. Lancet 2: 1014–1016
El-Mohandes AE, Keiser JF, Refat M, Jackson BJ 1993 Prevalence and toxigenicity of Clostridium difficile isolates in faecal microflora of preterm infants in the intensive care nursery. Biol Neonate 63: 225–229
Cashore WJ, Peter G, Lauermann M, Stonestreet BS, Oh W 1981 Clostridia colonization and clostridia toxin in neonatal necrotizing enterocolitis. J Pediatr 98: 308–311
Han VK, Sayed H, Chance GW, Brabyn DG, Shaheed WA 1983 An outbreak of Clostridium difficile necrotizing enterocolitis: a case for oral vancomycin therapy?. Pediatrics 71: 935–941
Sherertz RJ, Sarubbi FA 1982 The prevalence of Clostridium difficile and toxin in a nursery population: a comparison between patients with necrotizing enterocolitis and an asymptomatic group. J Pediatr 100: 435–439
Bartlett JG, Moon N, Chang TW, Taylor N, Onderdonk AB 1978 Role of Clostridium difficile in antibiotic-associated pseudomembranous colitis. Gastroenterology 75: 778–782
Urushihara N, Kohno S, Hasagawa S 1994 Pseudomembranous enterocolitis and hemorrhagic necrotizing enterocolitis in Hirschsprung's disease. Surg Today 24: 221–224
Engel RR, Virning NL, Hunt CE, Levitt MD 1973 Origin of mural gas in necrotizing enterocolitis. Pediatr Res 7: 292
Thomas DF, Fernie DS, Bayston R, Spitz L 1984 Clostridial toxins in neonatal necrotising enterocolitis. Arch Dis Child 59: 270–272
Stark PL, Lee A 1982 The microbial ecology of the large bowel of breast-fed and formula-fed infants during the first year of life. J Med Microbiol 15: 189–203
Favier CF, Vaughan EE, De Vos WM, Akkermans AD 2002 Molecular monitoring of succession of bacterial communities in human neonates. Appl Environ Microbiol 68: 219–226
Macfarlane GT 1997 Human colonic microbiota: ecology, physiology and metabolic potential of intestinal bacteria. Scand J Gastroenterol Suppl 222: 3–9
Carbonaro CA, Clark DA, Elseviers D 1988 A bacterial pathogenicity determinant associated with necrotizing enterocolitis. Microb Pathog 5: 427–436
Lin J, Nafday SM, Chauvin SN, Magid MS, Pabbatireddy S, Holzman IR, Babyatsky MW 2002 Variable effects of short chain fatty acids and lactic acid in inducing intestinal mucosal injury in newborn rats. J Pediatr Gastroenterol Nutr 35: 545–550
Fusunyan RD, Quinn JJ, Ohno Y, MacDermott RP, Sanderson IR 1998 Butyrate enhances interleukin (IL)-8 secretion by intestinal epithelial cells in response to IL-1β and lipopolysaccharide. Pediatr Res 43: 84–90
Sansonetti PJ, Arondel J, Huerre M, Harada A, Matsushima K 1999 Interleukin-8 controls bacterial transepithelial translocation at the cost of epithelial destruction in experimental shigellosis. Infect Immun 67: 1471–1480
Sanderson IR 1999 The physicochemical environment of the neonatal intestine. Am J Clin Nutr 69: 1028S–1034S
Favre A, Szylit O, Popot F, Catala I, Rondeau C, Maurage C, Gold F, Borderon JC, Butel MJ 2002 Diet, length of gestation and fecal short chain fatty acids in healthy premature neonates. JPEN J Parenter Enteral Nutr 26: 51–56
Clark DA, Miller MJ 1990 Intraluminal pathogenesis of necrotising enterocolitis. J Pediatr 117: S64–S67
Gupta S, Morris JG Jr, Panigrahi P, Nataro JP, Glass RI, Gewolb IH 1994 Endemic necrotizing enterocolitis: lack of association with a specific infectious agent. Pediatr Infect Dis J 13: 728–734
Duffy LC, Zielezny MA, Carrion V, Griffiths E, Dryja D, Hilty M, Cummings J, Morin F 2001 Bacterial toxins and enteral feeding of premature infants at risk for necrotizing enterocolitis. Adv Exp Med Biol 501: 519–527
Acknowledgements
We acknowledge Odette Szylit for advice and the Unité d'Ecologie et Physiologie du Système Digestif, CRZ-INRA (Jouy-en-Josas, France) for technical support with germ-free isolators. Patrick Avé is thanked for help with histologic examinations, Annie-Claude Tessedre for technical assistance in bacteriologic procedures, and Anne-Marie Marty for revising English.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Waligora-Dupriet, AJ., Dugay, A., Auzeil, N. et al. Evidence for Clostridial Implication in Necrotizing Enterocolitis through Bacterial Fermentation in a Gnotobiotic Quail Model. Pediatr Res 58, 629–635 (2005). https://doi.org/10.1203/01.PDR.0000180538.13142.84
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/01.PDR.0000180538.13142.84
This article is cited by
-
Particular genomic and virulence traits associated with preterm infant-derived toxigenic Clostridium perfringens strains
Nature Microbiology (2023)
-
A direct comparison of mouse and human intestinal development using epithelial gene expression patterns
Pediatric Research (2020)
-
Metabolomic signatures distinguish the impact of formula carbohydrates on disease outcome in a preterm piglet model of NEC
Microbiome (2018)
-
The initial prophylactic antibiotic usage and subsequent necrotizing enterocolitis in high-risk premature infants: a systematic review and meta-analysis
Pediatric Surgery International (2018)
-
Community analysis of bacteria colonizing intestinal tissue of neonates with necrotizing enterocolitis
BMC Microbiology (2011)