Abstract
Fetal mineralization appears to be driven by the pregnancy-induced stimulation of intestinal Ca absorption. We thus hypothesized that mineralization would be impaired in fetuses of mice that lack the vitamin D receptor (VDR). Here we report on the maternal response to pregnancy, and the fetal mineralization, in mice with a homozygous disruption of the VDR gene (VDR −/−) mated with wild-type (wt) males. We found that VDR −/− mice show mild hypocalcemia, clear rickets and osteomalacia on bone histomorphometry, lower cortical bone density on quantitative tomography, and reduced concentrations of calbindin-D9k (CaBP-D9k) in duodenal mucosa and kidney. The skeletal response to pregnancy was comparable in wt and VDR −/− mice; duodenal CaBP-D9k concentrations increased during pregnancy in VDR −/− as in wt mice, but remained 40% lower than in wt mice. We confirmed our hypothesis that mineralization is defective in d18.5 VDR+/ − fetuses of VDR −/− mice, both by whole-body Ca determination and histomorphometric evaluation; the number of osteoclastic cells in bone was increased. The fetuses were hypercalcemic and had a 5-fold increase in circulating 1,25(OH)2D3. We then studied pregnancies in VDR −/− females, mated with wt males, fed a high Ca/P/lactose rescue diet during pregnancy. The rescue diet normalized the mineralization, the number of osteoclastic cells, and plasma Ca and 1,25(OH)2D3 concentrations in the fetuses. We interpret the data as evidence that, to ensure normal fetal mineralization, the maternal VDR-dependent intestinal Ca absorption can be substituted by passive Ca absorption entrained by a higher Ca intake. Alternatively or additionally, elevated 1,25(OH)2D3in utero may disturb bone development.
Similar content being viewed by others
Main
Increased intestinal Ca absorption during pregnancy appears to be a major determinant of fetal mineralization (1). The rise in Ca absorption is largely the result of a more efficient ATP-dependent, transcellular Ca absorption, which is vitamin D-receptor (VDR)-dependent. The concentrations of circulating 1,25(OH)2D3 and duodenal VDR increase during pregnancy in rats, as does the duodenal expression of the calbindin-D9k gene (CaBP-D9k) (2). CaBP-D9k is involved in the transport of Ca across the epithelia of the intestine (3), the distal tubules of the kidneys (4), and the syncytiotrophoblast (5). Following Ca influx through the apical membrane, which is facilitated by the epithelial Ca2+ channels 1 and 2, CaBP-D9κ serves as a “ferry” for Ca to cross the cytosol. Ca is then actively extruded at the basal membrane by a Ca2+-ATP-ase in the duodenum or placenta, and by a Ca2+-ATP-ase and a Na+-Ca2+-exchanger in the kidneys (3–6).
Several strains of mice with a disrupted VDR gene have been generated, including the Tokyo-strain (7), the Boston-strain (8) and the Leuven-strain (9). VDR-null mice show rickets and osteomalacia, with abnormal Ca homeostasis including hypocalcemia and hyperparathyroidism (7–9). The expression of the CaBP-D9k gene is reduced in the intestine and kidneys of VDR-null mice (7, 10, 11), confirming VDR-regulation (12). The abnormalities become manifest only in the late neonatal period or after weaning (7, 8). Reversing hypocalcemia and hyperparathyroidism, by a diet enriched with Ca, phosphate (P) and lactose, normalizes rickets and osteomalacia (13, 14).
Female VDR-null mice have a variable reduction of fertility: Tokyo-VDR-null mice are infertile (7, 15), whereas pregnancy is possible in Leuven-VDR-null mice. Data on pregnancy in mice lacking the VDR gene are elusive, and we hypothesized that fetal mineralization would be reduced, given the pivotal role of intestinal Ca absorption. The effect of VDR gene disruption on placental CaBP-D9k is uncertain. A study in normocalcemic nutritionally vitamin D (D)-deficient rats showed normal placental CaBP-D9k gene expression, compared with D-replete rats (16). Here we investigated the effect of VDR gene disruption on the maternal skeletal response, maternal and placental CaBP-D9k concentrations and fetal mineralization. In an additional experiment, we studied the effect of a ‘rescue' diet enriched with Ca, P and lactose on CaBP-D9k concentrations and fetal mineralization in VDR-null mice.
METHODS
Animals.
The experiments were approved by the ethical committee for animal research at the K.U. Leuven. Leuven VDR-null mice (VDR −/−) were generated by homozygous targeted ablation of exon 2 of the VDR-gene in embryonic stem cells, as described earlier (9). The animals received a standard murine diet, containing 1.04% Ca, 0.76% P, and 2000 IU D/kg (Carfil, Oud-Turnhout, Belgium). Six-week-old female VDR −/− mice, born from VDR+/ − mothers and VDR −/− fathers, were mated overnight with wild-type (wt) males, so that the fetuses were heterozygous for VDR (VDR+/ −). Pregnant wt mice and their fetuses, as well as nonpregnant female VDR −/− and wt mice of the same age served as controls. The animals received bone fluorochrome labeling with calcein (Sigma Chemical Co., St. Louis, MO, U.S.A.), 50 μg i.p., 4 d and 24 h before surgery. In a separate experiment, pregnant VDR −/− mice were fed a high lactose, and Ca-and P-enriched murine diet, containing 2.0% Ca, 1.25% P, 2200 IU D/kg, and 20% lactose (VDR −/− rescue diet group) (Teklad, Madison, WI, U.S.A.) from diagnosis of pregnancy (i.e., the presence of a copulation plug).
On d 18.5, the mice were anesthetized with an i.p. injection of sodium pentobarbital (0.6 μg/g body weight). In pregnant animals, the uterus was exposed from left to right, and the fetuses were delivered through small uterine incisions. They were bled by an axillary incision, with the umbilical cord still attached to the placenta; fetal blood was pooled per litter. All fetuses and placentas were subsequently weighed. Both tibiae of the first 3 fetuses were removed for histomorphometry. The remaining fetuses were eviscerated, and stored at −20°C for measurement of the Ca content. Maternal blood was sampled by cardiac puncture, and the mice were euthanized. The first placenta was fixed in p-formaldehyde 4% in cacodylate buffer 0.1 M (pH 7.2) for 24 h at 4°C, rinsed in 0.1 M cacodylate buffer (pH 7.2) for 24 h, and then kept in ethanol 70% at 4°C, until embedding for CaBP-D9k immunohistochemistry. The other placentas, and the maternal duodenum and left kidney were rapidly removed, snap frozen in liquid nitrogen and stored in −80°C for determination of CaBP-D9k protein content. The left tibia was kept in Burkhardt's fixative for 24 h at 4°C, and then kept in ethanol 100%, until embedding for histomorphometry; the right tibia was fixated in p-formaldehyde 2% in cacodylate buffer 0.1 M (pH 7.2) for 24 h, and then kept on cacodylate buffer 0.1 M (pH 7.2) at 4°C, until decalcification. The femora were stored for quantitative computed tomography (QCT).
Assays.
Total Ca in plasma was measured colorimetrically (Sigma Chemical Co. Diagnostics, St. Louis, MO, U.S.A.). 1,25(OH)2D3 was measured by a RIA-kit (Diasorin, Stillwater, MN, U.S.A.); plasma samples of wt adult mice and of the fetal mice were pooled in a consecutive 2-by-2 fashion for this assay. Osteocalcin was measured by an in-house RIA with mouse osteocalcin as the standard and a rabbit polyclonal antiserum (17). Intra- and inter-assay coefficients of variation (CV) are 5.9% (n = 10) and 5.2% (n = 9). CaBP-D9k protein content was measured in homogenates of the duodenum, kidney and placenta by RIA, using CaBP-D9k, purified from rat duodenum, as the standard (12) and a polyclonal antiserum raised in rabbits (batch R152489, kindly donated by M.E. Bruns, Charlottesville, VA, U.S.A.), as described previously (18); values were expressed per total protein, which was measured colorimetrically (Pierce BCA Protein Assay, Rockford, IL, U.S.A.). The detection limit is 2.5 ng/mL for CaBP-D9k and 0.1 mg/mL for total protein; intra- and inter-assay CV are 5.2% and 6.3%, respectively.
Placental CaBP-D9k immunohistochemistry.
Placental CaBP-D9k immunohistochemistry was performed as described (18); the anti-rat CaBP-D9k antiserum was diluted 1:32,000.
Bone studies.
The dry weight of the adult femora was obtained after dehydrating (in ethanol and diethylether for several days) and drying (24 h at 100°C) the bones. QCT of the adult femora was measured with a XCT Bone Scanner (Norland, Fort Atkinson, WI), as described (19). Trabecular bone density was measured in an inner area of 25% of the total cross-sectional area, at 0.75, 1.0 and 1.25 mm from the distal growth plate; the average value was calculated. Cortical bone density and the endocortical and pericortical perimeters were measured at 7 mm from the distal growth plate. Between-measurement CV is <1%.
Eviscerated fetuses were dried (24 h at 100°C) and ashed in a muffle furnace at 600°C. The ash of each fetus was weighed and dissolved in HCl; Ca was measured by colorimetry in diluted ash and the fetal Ca content was calculated as a percentage of fetal ash and per dry body weight after evisceration (18).
Histomorphometry of adult tibiae was performed as described previously for rat (20) and guinea pig tibiae (19, 21), with adaptations for mouse bones. The undecalcified left proximal tibia was embedded in methylmethacrylate, and 4 μm-thick longitudinal sections were cut. Three sections were assessed per animal, with 100 μm in between. Static and dynamic morphometry were performed using a KS 400 Image Analyzing Computer (Carl Zeiss, Hallbergmoos, Germany) or a VIDAS 21 Image Analyzing Computer (Kontron, Munich, Germany). Standardized measurements, nomenclature and abbreviations were used (22). Trabecular bone volume (BV/TV) was measured in von Kossa-stained sections, in 2 consecutive fields in the vertical axis of the central metaphysis at magnification ×300, excluding the primary spongiosa. Sections stained with a modified Goldner-trichrome were used for the measurement of osteoid surface (OS/BS) and thickness (O.Th) and the growth plate width (GPW). Growth plate width was measured over the full length of the growth plate at magnification ×300. The other measurements were performed in the secondary spongiosa, in as many fields as possible, at magnification ×300, excluding the endocortical and subcortical bone surfaces: 4.0 ± 0.1 fields (mean ± SEM, n = 156) were assessed per section, corresponding to 12,657 ± 782 μm trabecular bone surface. For the fluoromicroscopic measurement in unstained sections of double- (dLS/BS) and single-labeled (sLS/BS) surfaces and the mineral apposition rate (MAR), we measured 6 ± 1 double labels per section; the mineral apposition rate was measured at 3 equidistant points per double label. If a particular section contained no double fluorochrome labels, the mineral apposition rate was given a missing value. The mineral formation rate, surface-referent (MFR/BS), was calculated as the mineralizing surface (MS/BS = dLS/BS + 1/2 sLS/BS) × mineral apposition rate × π/4 (to correct for the obliquity of the sections).
The right proximal tibiae of adult mice were decalcified, embedded in paraffin, and 3 μm-thick sections were cut. The sections were stained for tartrate-resistant acid phosphatase (TRAP) as described previously (20), but with incubation for 1 h in Tris after deparaffination and with an incubation time of 40 min in naphthol AS-BI phosphate. The measurements (TRAPS/BS) were performed exactly as for the osteoid surface.
Fetal (undecalcified) tibiae were embedded in paraffin, and 3 μm-thick sections were produced. Three sections per fetus were assessed, with 18 μm in between. We measured the mineralized bone volume, relative to the total bone volume (BV/TV) in von Kossa-stained sections, at magnification ×150. In TRAP-stained sections, we counted the TRAP-positive cells in the trabecular bone area, at magnification ×600. We also measured the area occupied by hypertrophic chondrocytes relative to the total chondrocyte area of the growth plate at magnification ×150, as described previously in guinea-pig fetuses (19) and in VDR −/− mice (8). Dynamic histomorphometry could not be performed in fetuses, because the calcein labeling pattern was indistinct.
Data analysis.
Data analysis was performed with a software program (NCSS, Kaysville, UT, U.S.A.). In wt and VDR −/− adult animals, the effects of genotype and pregnancy were compared by two-factor ANOVA. If a significant interactive effect was detected (p < 0.05), one-way ANOVA was performed to confirm an overall significant effect, followed by Fisher's LSD multiple comparison test to detect differences between individual groups. Fetuses of the 3 groups (wt, VDR −/− and VDR −/− rescue) were analyzed by one-way ANOVA; if p < 0.05, Fisher's LSD multiple comparison test was used to detect significant differences between individual groups. Where appropriate, 2 individual groups were compared by unpaired t tests, taking into account the variance of the data.
RESULTS
Phenotype of VDR−/− mice.
VDR-ablation did not affect body weight, but, as expected, plasma Ca levels were lower while 1,25(OH)2D3 levels were markedly higher (Table 1). The femoral dry weight was not altered in VDR −/− mice. However, on QCT analysis, cortical thickness (−15%) and cortical bone density (−10%) were reduced, whereas trabecular bone density was unchanged (Table 2). Histomorphometry of trabecular bone showed the typical signs of rickets (increased growth plate width) and osteomalacia (increased osteoid surface and thickness); trabecular bone volume was also increased, as was the bone surface covered by TRAP-positive cells (Table 3). But we found no effect of VDR ko on mineralizing surface, mineral apposition rate or mineral formation rate on dynamic morphometry.
In wt mice, CaBP-D9k concentrations were 5-fold higher in the duodenum than in the kidney, and 3-fold higher than in the placenta (Fig. 1). VDR ko lowered duodenal CaBP-D9k by 35% to 40%, and renal CaBP-D9k by 90%.
CaBP-D9k in duodenum, kidney, and placenta of nonpregnant (npr) and d 18.5-pregnant (pr) wt (gray bars) and VDR-null (black bars) mice and pregnant VDR −/− rescue (hatched bars) mice. The number of data (n) is shown below the bars. Statistical analysis: in wt and VDR-null animals, the effects of genotype and pregnancy on duodenal and renal CaBP-D9k were studied by two-factor ANOVA. An interactive effect between pregnancy and genotype was detected for renal CaBP-D9k; therefore, one-way ANOVA was performed to confirm an overall significant difference, followed by Fisher's LSD multiple comparison test to detect differences between individual groups. Posthoc comparisons that were significantly different are denoted by different capital letters on top of the bars (p < 0.01). Pregnant VDR-null mice on the rescue diet were compared with VDR-null mice on the regular diet by unpaired t tests.
Changes during late pregnancy in wt and VDR−/− mice.
Plasma Ca was slightly increased in late pregnancy in both wt and VDR−/− mice (Table 1); 1,25(OH)2D3 levels rose during wt pregnancy (although this was not significant), whereas 1,25(OH)2D3 was lower in pregnant than in nonpregnant ko mice. Body weight, and femoral dry weight (Table 2) increased comparably in wt and VDR−/− mice. On QCT analysis, cortical thickness and bone density did not change, but (metaphyseal) trabecular bone density was increased in pregnant mice. Similarly, on histomorphometry, trabecular bone volume was increased in pregnant compared with nonpregnant mice (Table 3). Furthermore, histomorphometry of trabecular bone showed a pregnancy-associated decline in parameters indicative of bone formation and mineralization: osteoid surface and thickness, and mineralization surface and mineral formation rate. Growth plate width and TRAP-positive surface were unaffected by pregnancy. Overall, there were no significant interactions in the skeletal response to pregnancy between wt and ko mice.
Duodenal CaBP-D9k increased during pregnancy in both wt and VDR −/− animals, but the pregnancy-associated rise in renal CaBP-D9k, observed in wt mice, was absent in VDR −/− mice (Fig. 1).
Effects of the rescue diet in pregnant VDR−/− mice.
The Ca/P/lactose-enriched diet reversed the slight hypocalcemia in VDR −/− mice on the regular diet, as well as the increased 1,25(OH)2D3 concentrations (Table 1). Although body weight was lower in the mice on the rescue diet, femoral dry weight (Table 2) was comparable to that of ko mice on the regular diet. QCT analysis showed that the reduction in cortical bone thickness and density was reversed by the rescue diet; cortical density was also 3% higher than in pregnant wt mice. In addition, trabecular bone density was 55% and 66% higher than in pregnant wt and ko mice on the regular diet, respectively. The rescue diet further reduced duodenal and renal CaBP-D9k concentrations (Fig. 1).
Placental parameters in wt and VDR−/− pregnancies.
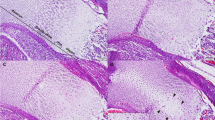
Placental weight was not different among wt, VDR −/− and VDR −/− rescue groups (ANOVA:p = 0.16). In addition, we found no significant differences in the placental CaBP-D9k concentrations among these 3 groups (Fig. 1). Immunohistochemistry of the placenta showed that CaBP-D9 K-staining was most intense in the intraplacental yolk sac. Staining was equally intense in the visceral and parietal layer of the intraplacental yolk sac, but appeared to be more abundant in the visceral layer. Weak staining was observed in the trophospongium and in the trophospongial islets within the labyrinth, as well as in the trophoblastic giant cells (not shown). On qualitative examination, we found no differences in the staining intensity or localization of CaBP-D9k in placentas of wt, VDR −/−, and VDR −/− rescue mice (Fig. 2).
Representative sections of CaBP-D9k immunohistochemistry in d 18.5-pregnant murine placentas, showing intense staining of the intraplacental yolk sac. Top: placenta of a wt mouse. Bottom: placenta of a VDR-null mouse. V, and P: visceral and parietal layer of the intraplacental yolk sac, respectively. Magnification × 100.
Fetal growth and mineralization, and effect of the rescue diet.
There was no difference among the 3 groups in the number of live fetuses per litter [6.9 (0.7) mean (SEM), 7.6 (0.7), and 6.6 (0.8) in wt, VDR −/−, and VDR −/− rescue groups, respectively; ANOVA:p = 0.65], or the number of early (resorptions) or late fetal deaths (ANOVA:p = 0.85 and p = 0.88, respectively). Compared with wt fetuses, there was an 8% reduction in body weight in VDR+/ − fetuses on the regular diet, and a 16% reduction in VDR+/ − fetuses of the rescue diet group (Table 4); thus, the rescue diet aggravated the fetal growth retardation. However, plasma Ca, 1,25(OH)2D3 and osteocalcin concentrations were higher in VDR+/ − fetuses than in wt fetuses, but were restored by the rescue diet to values measured in wt fetuses.
The Ca content, and the mineralized bone volume on histomorphometry, of VDR+/ − fetuses were 45% and 41%, respectively, of the values measured in wt fetuses (Fig. 3 and 4). In addition, the number of TRAP-positive cells per bone area was raised by 144%, compared with wt fetuses. The rescue diet normalized the mineralized bone volume and the number of TRAP-positive cells, and raised the fetal Ca content to 83% of wt values. No significant differences were found among the 3 groups in the hypertrophic chondrocyte area (ANOVA:p = 0.07; data not shown).
Bone data of fetuses of wt (gray bars), VDR-null (black bars), and VDR-null mice on the rescue diet (hatched bars): BV/TV, mineralized bone volume; TRAP, number of TRAP-positive cells. The number of data (n) is shown below the bars. Statistical analysis: fetuses of the different groups were analyzed by one-way ANOVA; if p < 0.05, Fisher's LSD multiple comparison test was used to detect significant differences between individual groups:Posthoc comparisons that were significantly different are denoted by different capital letters on top of the bars (all p < 0.01).
DISCUSSION
VDR −/− mice showed the anticipated abnormalities in bone mineral homeostasis. They were slightly hypocalcemic, with the expected compensatory increase in circulating 1,25(OH)2D3. Bone histomorphometry confirmed rickets (wide growth plates at the proximal tibia) and osteomalacia (abundant and wide osteoid seams at trabecular bone surfaces). The TRAP-positive surface was increased, suggestive of secondary hyperparathyroidism; in Tokyo- and Boston-VDR-null mice, histomorphometric indices of bone resorption were reportedly normal, as was the resorbing activity of osteoclasts generated in vitro(14, 23). Trabecular bone volume was slightly increased, confirming previous data (14, 23), but trabecular bone density on QCT was unaffected. However, cortical bone thickness and density on QCT was 10% to 15% lower than in wt mice, which is in line with our findings in nutritionally vitamin D-deficient guinea-pigs (19). Finally, concentrations of CaBP-D9k in duodenum and kidney were reduced, as previously shown in several VDR −/− strains at the mRNA and protein levels (7, 9, 11).
The maternal response to pregnancy was comparable in wt and VDR −/− mice. An interactive effect for pregnancy was observed only for renal CaBP-D9k, i.e., renal CaBP-D9k concentrations increased in wt but not in VDR −/− pregnancies. The skeletal response to pregnancy was comparable, and included a suppression of bone formation parameters on histomorphometry, both static and dynamic, and an increase in trabecular bone volume on histomorphometry and in trabecular bone density on QCT. We have reported that histomorphometric bone formation parameters are also reduced markedly during late pregnancy in guinea pigs (19, 21). The increase in trabecular bone volume/density - shown by two independent methods - is difficult to explain, given that bone resorption does not appear to be altered by pregnancy; clearly, further research into this issue is needed. Duodenal CaBP-D9k concentrations rose during pregnancy, compatible with the pregnancy-driven stimulation of active intestinal Ca absorption (2), but remained 40% lower in ko than in wt mice. These findings confirm data showing that the active intestinal Ca absorption increases during late pregnancy in both D-replete and D-deficient rats, but is still 40% lower in D-deficient rats (24).
Fetuses of VDR −/− mice were slightly growth-retarded, and were hypomineralized: both the fetal Ca content, corrected for weight, and the mineralized bone in tibial sections, were reduced. But the hypertrophic chondrocyte area was not affected, suggesting that, while bone maturation is normal, mineralization is impaired because of lower mineral availability, much the same as we have previously documented in fetuses of strontium-fed rats (18). In addition, the fetuses were hypercalcemic, with a 5-fold increase in 1,25(OH)2D3 concentrations. We surmise that the latter is the result of high maternal 1,25(OH)2D3 levels. Indeed, maternal and fetal 1,25(OH)2D3 concentrations are highly correlated in human (25), rat (18) and guinea pig pregnancies (19, 21). Whether the hypercalcemia is related to elevated 1,25(OH)2D3 levels in utero is uncertain, but cannot be excluded since the fetuses were genotypically heterozygous and thus potentially responsive to 1,25(OH)2D3. In fetal sheep, nephrectomy reduced both 1,25(OH)2D3 and Ca levels (26), whereas fetal nephrectomy in rats reduced 1,25(OH)2D3 but not Ca levels (27). The fetal hypercalcemia may also be explained by secondary hyperparathyroidism (19), since we found an increase in the osteoclastic cell number on histomorphometry of the tibia. However, we were unable to measure PTH reliably in the fetuses. Plasma osteocalcin levels were higher in fetuses of ko mice, inferring that osteoblast number or/and activity was increased as well; this could not be corroborated by histomorphometry, because of technical limitations.
A high Ca/P/lactose diet rescues the metabolic and skeletal abnormalities of VDR ko mice, including a normalization of histomorphometrical parameters (13, 14). We confirm these data in pregnant ko mice: the hypocalcemia was reversed, as well as the reduced cortical bone density on QCT. But the rescue diet lowered CaBP-D9k concentrations in duodenum and kidney further; the duodenum data confirm previous results in nonpregnant Leuven VDR-ko mice (9), whereas reversal of decreased CaBP-D9k was reported in Boston VDR ko mice on the rescue diet (10). Our results are compatible with the paradigm that the high Ca/P/lactose diet promotes the paracellular, nonsaturable Ca transport in the intestine, which is VDR- and CaBP-D9k-independent (3), and represses VDR-dependent Ca absorption.
In the fetuses, the rescue diet reversed the hypomineralization, the increased osteoclastic cell number in tibial sections, the increased plasma osteocalcin and 1,25(OH)2D3 levels, and the hypercalcemia. How to explain these data? First, since the fetuses were all genotypically heterozygous for the VDR, the genotype cannot be the sole explanation for the bone and mineral phenotype. Second, the rescue diet raises the maternal intestinal passive Ca absorption, which is a linear function of the Ca intake (3), and may thereby promote the Ca availability to the fetuses. Hence, the suboptimal VDR-dependent Ca absorption would be substituted by Ca intake-entrained VDR-independent Ca absorption to ensure normal fetal mineralization. Third, VDR+/ − fetuses on the regular diet had high 1,25(OH)2D3 levels, and there is some evidence for a ‘toxic' effect of 1,25(OH)2D3 on bone development in utero. Indeed, in experiments with 24-hydroxylase gene ko mice, homozygous mutant newborns of homozygous mutant mothers showed deficient mineralization, which was rescued in newborns that also lacked the VDR gene, inferring that high circulating 1,25(OH)2D3 acting through the VDR was responsible for the defective mineralization (28). Finally, the putative hyperparathyroidism of VDR+/ − fetuses on the regular diet may also be involved in the hypomineralization, through increased bone resorption (29).
The fetal growth retardation was not reversed by the rescue diet; in fact, VDR+/ − fetuses on the rescue diet weighed less than VDR+/ − fetuses on the regular diet. The data suggest that the VDR genotype may be a determinant of fetal growth in mice. In humans, there is some evidence that VDR allelic variants are associated with height at birth and postnatal growth (30). There may also be a link with the lower maternal body weight in the rescue diet group, a finding for which we have no explanation. In nutritionally D-deficient guinea pigs, we previously documented that a comparable rescue diet reversed both fetal growth retardation and hypomineralization (19).
From our data, placental CaBP-D9k expression does not appear to be VDR-dependent, although this needs to be confirmed at the mRNA transcript level; however, our results are consistent with previous data in nutritionally D-deficient rats (16). CaBP-D9k protein expression was particularly intense in the visceral layer of the intraplacental yolk sac -which may well be crucial for maternal-fetal Ca transfer in mice (31) - and to a lesser extent in the trophoblastic giant cells, as previously reported (32).
This study has several limitations. First, we did not include a rescue diet experiment in wt or nonpregnant VDR −/−- mice. Second, it would be of interest to expand the study and compare VDR+/ − and VDR −/− fetuses from matings of VDR −/− females and VDR+/ − males. Third, other proteins involved in intestinal Ca absorption, such as the epithelial Ca2+ channels, were not assessed here.
Experiments in VDR ko mice have demonstrated that the prime effect of the VDR is the stimulation of intestinal Ca absorption and Ca availability. Feeding VDR −/− mice a high Ca/P/lactose diet normalizes not only the hypocalcemia, hyperparathyroidism and the skeletal phenotype, but also fertility in some VDR ko strains (15) and the abnormalities in the immune system (33). The present data add to this body of knowledge, showing that fetuses of mothers with a disrupted VDR gene have a defective mineralization, and is reversed by a rescue diet that also normalizes maternal and fetal 1,25(OH)2D3 levels.
Abbreviations
- 1,25(OH)2D3:
-
1,25-dihydroxyvitamin D3
- CaBP-D9k:
-
calbindin-D9k
- CV:
-
coefficient of variation
- ko :
-
knock-out
- QCT:
-
quantitative computed tomography
- TRAP:
-
tartrate-resistant acid phosphatase
- VDR:
-
vitamin D receptor
- wt :
-
wild type
References
Ritchie LD, Fung EB, Halloran BP, Turnlund JR, Van Loan MD, Cann CE, King JC 1998 A longitudinal study of calcium homeostasis during human pregnancy and lactation and after resumption of menses. Am J Clin Nutr 67: 693–701
Zhu Y, Goff JP, Reinhardt TA, Horst RL 1998 Pregnancy and lactation increase vitamin D-dependent intestinal membrane calcium adenosine triphosphatase and calcium binding protein messenger ribonucleic acid expression. Endocrinology 139: 3520–3524
Bronner F, Pansu D, Stein WD 1986 An analysis of intestinal calcium transport across the rat intestine. Am J Physiol 250: G561–G569
Bronner F 1989 Renal calcium transport: mechanisms and regulation: an overview. Am J Physiol 257: F707–F711
Glazier JD, Atkinson DE, Thornburg KL, Sharpe PT, Edwards D, Boyd RDH, Sibley CP 1992 Gestational changes in Ca2+transport across rat placenta and mRNA for calbindin9K and Ca2+-ATPase. Am J Physiol 263: R930–R935
Hoenderop JG, Willems PH, Bindels RJ 2000 Toward a comprehensive molecular model of active calcium reabsorption. Am J Physiol 278: F352–360
Yoshizawa T, Handa Y, Uematsu Y, Takeda S, Sekine K, Yoshihara Y, Kawakami T, Arioka K, Sato H, Uchiyama Y, Masushige S, Fukamizu A, Matsumoto T, Kato S 1997 Mice lacking the vitamin D receptor exhibit impaired bone formation, uterine hypoplasia and growth retardation after weaning. Nat Genet 16: 391–396
Li YC, Pirro AE, Amling M, Delling G, Baron R, Bronson R, Demay MB 1997 Targeted ablation of the vitamin D receptor: an animal model of vitamin D-dependent rickets type II with alopecia. Proc Natl Acad Sci USA 94: 9831–9835
Van Cromphaut SJ, Dewerchin M, Hoenderop JG, Stockmans I, Van Herck E, Kato S, Bindels RJ, Collen D, Carmeliet P, Bouillon R, Carmeliet G 2001 Duodenal calcium absorption in vitamin D receptor-knockout mice: functional and molecular aspects. Proc Natl Acad Sci USA 98: 13324–13329
Li YC, Pirro AE, Demay MB 1998 Analysis of vitamin D-dependent calcium-binding protein messenger ribonucleic acid expression in mice lacking the vitamin D receptor. Endocrinology 139: 847–851
Li YC, Bolt MJG, Cao L-P, Sitrin MD 2001 Effects of vitamin D receptor inactivation on the expression of calbindins and calcium metabolism. Am J Physiol 281: E558–E564
Thomasset M, Parkes CO, Cuisinier-Gleizes P 1982 Rat calcium-binding proteins: distribution, development, and vitamin D dependence. Am J Physiol 243: E483–E488
Li YC, Amling M, Pirro AE, Priemel M, Meuse J, Baron R, Delling G, Demay MB 1998 Normalization of mineral ion homeostasis by dietary means prevents hyperparathyroidism, rickets, and osteomalacia, but not alopecia in vitamin D receptor-ablated mice. Endocrinology 139: 4391–4396
Amling M, Priemel M, Holzmann T, Chapin K, Rueger JM, Baron R, Demay MB 1999 Rescue of the skeletal phenotype of vitamin D receptor-ablated mice in the setting of normal mineral ion homeostasis: formal histomorphometric and biomechanical analyses. Endocrinology 140: 4982–4987
Johnson LE, DeLuca HF 2001 Vitamin D receptor null mutant mice fed high levels of calcium are fertile. J Nutr 131: 1787–1791
Glazier JD, Mawer EB, Sibley CP 1995 Calbindin-D9K gene expression in rat chorioallantoic placenta is not regulated by 1,25-dihydroxyvitamin D3 . Pediatr Res 37: 720–725
Verhaeghe J, Van Herck E, Van Bree R, Van Assche FA, Bouillon R 1989 Osteocalcin during the reproductive cycle in normal and diabetic rats. J Endocrinol 120: 143–151
Verhaeghe J, van Bree R, van Herck E, Rummens K, Vercruysse L, Bouillon R, Pijnenborg R 1999 Pathogenesis of fetal hypomineralization in diabetic rats: evidence for delayed bone maturation. Pediatr Res 45: 209–217
Rummens K, van Bree R, Van Herck E, Zaman Z, Bouillon R, Van Assche FA, Verhaeghe J 2002 Vitamin D-deficiency in guinea pigs: exacerbation of bone phenotype during pregnancy and disturbed fetal mineralization, with recovery by 1,25(OH)2D3 infusion or dietary calcium-phosphate supplementation. Calcif Tissue Int 71: 364–375
Verhaeghe J, Oloumi G, Van Herck E, van Bree R, Dequeker J, Einhorn TA, Bouillon R 1997 Effects of long-term diabetes and/or high-dose 17β-estradiol on bone formation, bone mineral density, and strength in ovariectomized rats. Bone 20: 421–428
Rummens K, Van Herck E, van Bree R, Bouillon R, Van Assche FA, Verhaeghe J 2000 Dietary calcium and phosphate restriction in guinea-pigs during pregnancy: fetal mineralization induces maternal hypocalcaemia despite increased 1α,25-dihydroxycholecalciferol concentrations. Br J Nutr 84: 495–504
Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR 1987 Bone histomorphometry: standardization of nomenclature, symbols, and units. J Bone Miner Res 2: 595–610
Takeda S, Yoshizawa T, Nagai Y, Yamato H, Fukumoto S, Sekine K, Kato S, Matsumoto T, Fujita T 1999 Stimulation of osteoclast formation by 1,25-dihydroxyvitamin D requires its binding to vitamin D receptor (VDR) in osteoblastic cells: studies using VDR knockout mice. Endocrinology 140: 1005–1008
Halloran BP, DeLuca HF 1980 Calcium transport in small intestine during pregnancy and lactation. Am J Physiol 239: E64–E68
Bouillon R, Van Assche FA, Van Baelen H, Heyns W, De Moor P 1981 Influence of the vitamin D-binding protein on the serum concentration of 1,25-dihydroxyvitamin D3 . J Clin Invest 67: 589–596
Moore ES, Langman CB, Favus MJ, Coe FL 1985 Role of fetal 1,25-dihydroxyvitamin D production in intrauterine phosphorus and calcium homeostasis. Pediatr Res 19: 566–569
Chalon S, Garel JM 1985 Plasma calcium control in the rat fetus. II. Influence of fetal hormones. Biol Neonate 48: 323–328
St-Arnaud R, Arabian A, Travers R, Barletta F, Raval-Pandya M, Chapin K, Depovere J, Mathieu C, Christakos S, Demay MB, Glorieux FH 2000 Deficient mineralization of intramembranous bone in vitamin D-24-hydroxylase-ablated mice is due to elevated 1,25-dihydroxyvitamin D and not to the absence of 24,25-dihydroxyvitamin D. Endocrinology 141: 2658–2666
Kovacs CS, Ho-Pao CL, Hunzelman JL, Lanske B, Fox J, Seidman JG, Seidman CE, Kronenberg HM 1998 Regulation of murine fetal-placental calcium metabolism by the calcium-sensing receptor. J Clin Invest 101: 2812–2820
Lorentzon M, Lorentzon R, Nordström P 2000 Vitamin D receptor gene polymorphism is associated with birth height, growth to adolescence, and adult stature in healthy Caucasian men: a cross-sectional and longitudinal study. J Clin Endocrinol Metab 85: 1666–1671
Kovacs C, Chafe LL, Woodland ML, McDonald KR, Fudge NJ, Wookey PJ 2002 Calcitropic gene expression suggests a role for the intraplacental yolk sac in maternal-fetal calcium exchange. Am J Physiol 282: E721–E732
Shamley DR, Veale G, Pettifor JM, Buffenstein R 1996 Trophoblastic giant cells of the mouse placenta contain calbindin-D9k but not the vitamin D receptor. J Endocrinol 150: 25–32
Mathieu C, Van Etten E, Cysemans C, Decallonne B, Kato S, Laureys J, Depovere J, Valckx D, Verstuyf A, Bouillon R 2001 In vitro and in vivo analysis of the immune system of vitamin D receptor knockout mice. J Bone Miner Res 6: 2057–2065
Acknowledgements
The authors are grateful to Professor D. Vanderschueren for the use of the QCT, and Professor R. Pijnenborg for his help with the CaBP-D9K immunohistochemistry. We also thank L. Vercruysse, R. Janssens, W. Coopmans, and I. Jans for their valuable advice and help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rummens, K., Van Cromphaut, S., Carmeliet, G. et al. Pregnancy in Mice Lacking the Vitamin D Receptor: Normal Maternal Skeletal Response, But Fetal Hypomineralization Rescued by Maternal Calcium Supplementation. Pediatr Res 54, 466–473 (2003). https://doi.org/10.1203/01.PDR.0000081302.06915.D3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/01.PDR.0000081302.06915.D3
This article is cited by
-
Maternal and fetal vitamin D and their roles in mineral homeostasis and fetal bone development
Journal of Endocrinological Investigation (2021)
-
The relationship between molar incisor hypomineralization, dental caries, socioeconomic factors, and polymorphisms in the vitamin D receptor gene: a population-based study
Clinical Oral Investigations (2020)
-
Vitamin D endocrine system and the intestine
BoneKEy Reports (2014)
-
Effect of the Vitamin D Receptor on Bone Geometry and Strength During Gestation and Lactation in Mice
Calcified Tissue International (2009)