Abstract
Our objective was to study the probability of achieving undetectable viral load levels in HIV-1-infected children after 36 mo of highly active antiretroviral therapy (HAART). A prospective, multicenter, longitudinal study in 41 HIV-1-infected children on HAART was undertaken. Viral load was quantified using standard molecular assay. CD4+ and CD8+ T cell subsets were determined by flow cytometry. The probability of achieving undetectable viral load was determined using Kaplan-Meier curves according to groups by percentage CD8+ at baseline (CD8+ <25% or >25%). Lower baseline CD8+ T cell levels conditioned a less effective virological response to HAART in children, independent of baseline CD4+ T cell numbers and viral load levels. A greater number of children (81%) from CD8+ >25% group than from the CD8+ <25% (40%) presented undetectable viral load levels (p = 0.013). Additionally, the CD8+ >25% group showed a 4.5-fold higher (95% confidence interval: 1.1-19.2) relative proportion for achieving viral load <400 copies/mL than the CD8+ <25% group (p = 0.039). We concluded that monitoring CD8+ T cell numbers may be valuable in deciding when to start HAART in vertically HIV-1-infected children.
Similar content being viewed by others
Main
HAART has been shown to have a profound impact in terms of reducing mortality, AIDS-defining illnesses, and hospital admissions (1, 2). Several studies have been conducted to evaluate the predictive markers of initial virologic response to HAART in either previously antiretroviral naïve or experienced patients (3, 4). However, little is known about the relationship between the plasma VL response over the first 36 mo of HAART and the baseline CD8+ T-cell values measured at the time of starting HAART in HIV-1-infected children. Studies of immune response generated in HIV-infected individuals indicate that CD8+ T cells play an important role in controlling viremia (5), and the emergence of HIV-1-specific CD8+ cytotoxic T-lymphocyte activity coincides with the clearance of viremia during primary HIV-1 infection (6, 7).
We therefore analyzed data from a cohort of HIV-1-infected children on first-line HAART to evaluate the probability of achieving undetectable VL levels by 36 mo after initiation of HAART according to their baseline CD8+ T-cell numbers.
METHODS
Study subjects.
Forty-one vertically HIV-1-infected children were recruited in a prospective longitudinal study between December 1996 and October 2001, at the General University Hospital “Gregorio Marañón” in Madrid, and the “Virgen del Rocío” Hospital in Seville, Spain. All HIV-infected children had first-line HAART with a combination of two nucleoside analogues plus one PI for an average of 39.8 ± 1.5 mo. Response to therapy was evaluated by serial measurements of percentage CD4+ and percentage CD8+ T cells and VL every 3 mo until 18 mo of age and every 6 mo thereafter. Because CD4+ T-cell numbers fall naturally with age in uninfected infants, and, even adjusting for age (8), absolute counts in HIV-1-infected infants seem to be much less useful for predicting disease progression than in adults (9), we compared follow-up of both groups in terms of relative numbers. The institutional ethics committees approved the study. Therapeutic success was defined as achievement of undetectable VL levels. Informed written consent was obtained for all the pediatric patients. The study was conducted according to the Declaration of Helsinki and approved by the ethical committees of the hospitals involved. Clinical classification was based on the 1994 revised guidelines of the Centers for Disease Control (CDC) (10): A (asymptomatic), B (symptomatic moderate), and C (symptomatic severe or AIDS).
Quantification of T-cell subsets in peripheral blood.
T-lymphocyte subsets in peripheral blood were quantified by direct immunofluorescence using MAb of the T series and flow cytometry (FACScan, BD Biosciences, San Jose, CA, U.S.A.) as previously described (11).
Quantitative HIV-1 RNA assay.
Blood samples were collected in EDTA tubes and separated within 4 h; plasma was stored at −70°C. HIV-1 RNA was measured in 200 μL plasma using a quantitative RT-PCR assay (Amplicor monitor, Roche Diagnostics, Basel, Switzerland). Limit of detection was 400 copies/mL.
Statistics.
CD4+ and CD8+ numbers are expressed as percentages. In all analyses, VL was transformed to log10 scale to normalize its distribution. Differences among infants' characteristics according to a critical value of percentage CD8+ T cells at baseline (<25% or >25%) were analyzed using independent-samples two-tailed Mann-Whitney U test, Kaplan-Meier, and Cox regression analysis. The cut-off level of 25% CD8+ was chosen inasmuch as it has been previously reported to have prognostic value (12). The differences between groups and clinical categories were determined by Pearson X2 and Fisher exact text.
We determined the probability of achieving undetectable VL using Kaplan-Meier curves according to group percentage CD8+ and compared them by the log-rank test (Mantel-Haenszel), with follow-up curtailed at the last CD8+ T-cell determination. A Cox regression analysis to assess RP and 95% CI of undetectable VL, adjusted for basal values of percentage CD4+ and percentage CD8+ T cells, age, and log10 VL was applied, to compensate possible differences. All RP estimates were calculated with respect to the CD8+ >25% group.
RESULTS
We performed a prospective longitudinal study in 41 vertically HIV-1-infected children rated into two groups according to CD8+ T cells at baseline: 10 children with CD8+ <25% and 31 children with CD8+ >25%. Age, clinical category, mean CD4+, and percentage CD4+ T cells and VL were comparable between both groups of children, as shown in Table 1. The group with CD8+ >25% showed higher CD8+ T-cell counts, and a lower percentage of children in clinical category C (AIDS) than those with CD8+ <25% (Table 1). The antiretroviral therapy pre-HAART and the change in HAART drugs during follow-up are detailed in Table 2. HIVchildren with CD8+ <25% had more changes in the ART drugs during follow-up than the CD8+ >25% group.
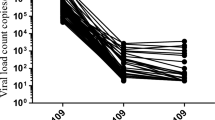
CD4+ and CD8+ T cells and VL were analyzed during follow-up, as illustrated in Figure 1. The CD8+ >25% group controlled VL quickly (by the third month after HAART). By contrast, the CD8+ <25% group showed a small decrease of VL (0.93 log10 VL versus 1.41 log10) within the first 3 mo of therapy, although the VL remained always above 4 log10 (Fig. 1C). A sharp decline in VL with slopes similar to those observed initially in the group CD8+ >25% was observed thereafter (Fig. 1C).
(A, B, and C) Values of the mean of CD4+ and CD8+ T cell percentages in peripheral blood, and plasma log10 VL (copies per milliliter) in the different groups of HIV-1-infected children during follow-up. HIV-1-infected children were separated into two groups according to CD8+ T cells baseline: 10 children CD8+ <25% and 31 children CD8+ >25%. Each point on the percentage CD4+, percentage CD8+, and VL lines represents the mean of the values observed for all children. (D) Kaplan-Meier estimates for undetectable VL evolution during follow-up. HIV-1-infected children were separated into two groups according to CD8+ T cells baseline.
Kaplan-Meier estimates and Cox regression analysis were used to assess time to achieve undetectable VL according to baseline CD8+ T cells. As shown in Figure 1D, a greater number of children (81%) from the CD8+ >25% group than the CD8+ <25% (40%) presented undetectable VL levels (p = 0.013). The CD8+ >25% group showed a 4.5-fold higher (95% CI: 1.1-19.2) relative proportion for achieving VL <400 copies/mL than CD8+ <25% group (p = 0.039). Moreover, we have not observed influences of the CDC clinical categories (A, B, and C) when including this variable together with the different categories. Interestingly, in those children, HAART allowed the recovery of percentage CD8 T cells to values near 40%. Neither baseline CD4+ T cell numbers nor VL were predictive of virological outcome after HAART (data not shown).
By multiple Cox regression analysis, CD8+ T cell numbers as a continuous variable was significant for therapeutical success (VL<400 copies/mL) and independent of CD4+ T cell numbers, VL, and age as covariates.
DISCUSSION
HIV-1 infection in pediatric patients shows distinctive characteristics from the adult features (12, 13), and these differences may have important consequences for the management of these patients. On the other hand, treatment with HAART modifies viral replication dynamics and induces a significant decrease of viral replication (14), although most therapeutic regimens have failed in achieving a complete suppression of HIV-1 replication in children (15, 16).
We have carried out a prospective, longitudinal, observational study in a pediatric population on HAART to define the predictive power of pre-HAART CD8+ T cells for virological response to HAART. We have studied the evolution of the most relevant markers of HIV infection progression (percentages of CD4+ and CD8+, and VL) in the two groups of children. We have observed that the probability of achieving an undetectable VL was strongly associated with pre-HAART percentage of CD8+ T cells. Thus, we have observed suppression of VL to undetectable levels in a higher proportion of children of the CD8+ >25% group than in the CD8+ <25% group. However, both groups showed a similar increase of CD4+ T cells over time, indicating a comparable immune reconstitution. On the other hand, the children in the CD8+ >25% group presented a faster decrease in VL compared with the CD8+ <25% group. Moreover, the predictive power of CD8+ T-cell numbers on virological success was independent of CD4+ T-cell numbers, VL, CDC clinical category, and age. These results suggest that the effect of HAART on the inhibition of viral replication (17, 18) is CD8+ T-cell dependent. There are, however, differences in the distribution of clinical categories, because children with baseline CD8+ T cells <25% were in a more advanced stage of the disease and there is a trend toward younger age, which might have biased the results.
HIV-1 infection by vertical transmission represents a unique mechanism of acute infection. Further studies are necessary to design more effective therapeutic strategies to suppress VL to undetectable levels and to increase percentage of CD4+ early and adequately in most of the treated children. In the management of HIV-1-infected children starting HAART, it is crucial to detect signs that are associated with incomplete suppression of VL so that more effective therapies can be implemented. HAART raises CD8+ T-cell numbers in HIV-1-infected patients (19). The CD8+ >25% group showed a lower median time (median, 10.0 mo; range, 3.5-16.5 mo) in achieving undetectable VL levels than the CD8+ >25% group (25.1 mo). It is possible that in several cases with high viral replication, earlier development of drug-resistant virus may occur and eventually result in virologic failure (20). However, our results suggest that children with low CD4+ (<15%), indicative of severe immunosuppression, but with CD8+ >25% may control HIV-1 replication. Thus, when the CD8+ T-lymphocyte numbers fall below a critical value, this may predict less effective virologic suppression and therefore may be used as a reliable marker as to whether to begin HAART. It is even more important to define which children are going to respond to HAART early and effectively in terms of VL, and our results indicate that CD8+ T-lymphocyte numbers have much to say in this regard.
Abbreviations
- ART:
-
antiretroviral therapy
- HAART:
-
highly active antiretroviral therapy
- VL:
-
viral load
- PI:
-
protease inhibitor
- RT-PCR:
-
reverse transcriptase PCR
- RP:
-
relative proportion
- 95% confidence interval:
-
95% CI
REFERENCES
Palella FJ Jr, Delaney KM, Moorman AC, Loveless MO, Fuhrer J, Satten GA, Aschman DJ, Holmberg SD 1998 Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. HIV Outpatient Study Investigators. N Engl J Med 338: 853–860.
Mocroft A, Barry S, Sabin CA, Lepri AC, Kinloch S, Drinkwater A, Lipman M, Youle M, Johnson MA, Phillips AN 1999 The changing pattern of admissions to a London hospital of patients with HIV: 1988-1997. Royal Free Centre for HIV Medicine. AIDS 13: 1255–1261.
Staszewski S, Miller V, Sabin C, Carlebach A, Berger AM, Weidmann E, Helm EB, Hill A, Phillips A 1999 Virological response to protease inhibitor therapy in an HIV clinic cohort. AIDS 13: 367–373.
Mocroft A, Gill MJ, Davidson W, Phillips AN 1998 Predictors of a viral response and subsequent virological treatment failure in patients with HIV starting a protease inhibitor. AIDS 12: 2161–2167.
Chun TW, Justement JS, Moir S, Hallahan CW, Ehler LA, Liu S, McLaughlin M, Dybul M, Mican JM, Fauci AS 2001 Suppression of HIV replication in the resting CD4+ T cell reservoir by autologous CD8+ T cells: implications for the development of therapeutic strategies. Proc Natl Acad Sci U S A 98: 253–258.
Borrow P, Lewicki H, Hahn BH, Shaw GM, Oldstone MB 1994 Virus-specific CD8+ cytotoxic T-lymphocyte activity associated with control of viremia in primary human immunodeficiency virus type 1 infection. J Virol 68: 6103–6110.
Koup RA, Safrit JT, Cao Y, Andrews CA, McLeod G, Borkowsky W, Farthing C, Ho DD 1994 Temporal association of cellular immune responses with the initial control of viremia in primary human immunodeficiency virus type 1 syndrome. J Virol 68: 4650–4655.
Study TEC 1992 Age-related standards for T lymphocyte subsets based on uninfected children born to human immunodeficiency virus 1-infected women. Pediatr Infect Dis J 11: 1018–1026.
Blanche S, Newell ML, Mayaux MJ, Dunn DT, Teglas JP, Rouzioux C, Peckham CS 1997 Morbidity and mortality in European children vertically infected by HIV-1. The French Pediatric HIV Infection Study Group and European Collaborative Study. J Acquir Immune Defic Syndr Hum Retrovirol 14: 442–450.
Center for Diseases Control Prevention 1994 Revised classification system for HIV-1 infection in children less than 13 years of age. MMWR Morb Mortal Weekly Rep 43: 1–13.
Munoz-Fernandez MA, Obregon E, Navarro J, Borner C, Gurbindo MD, Sampelayo TH, Fernandez-Cruz E 1996 Relationship of virologic, immunologic, and clinical parameters in infants with vertically acquired human immunodeficiency virus type 1 infection. Pediatr Res 40: 597–602.
Resino S, Gurbindo MD, Bellón JM, Sanchez-Ramón S, Muñoz-Fernández MA 2000 Predictive markers of clinical outcome in vertically HIV-1 infected infants. A prospective longitudinal study. Pediatr Res 47: 509–515.
Gurbindo D, Resino S, Sanchez-Ramon S, León JA, Munoz-Fernández MA 1999 Correlation of viral load and CD8 T-lymphocytes with development of neurological manifestations in vertically HIV-1-infected infants. A prospective longitudinal study. Neuropediatrics 30: 197–204.
Markowitz M, Saag M, Powderly WG, Hurley AM, Hsu A, Valdes JM, Henry D, Sattler F, La Marca A, Leonard JM 1995 A preliminary study of ritonavir, an inhibitor of HIV-1 protease, to treat HIV-1 infection. N Engl J Med 333: 1534–1539.
Finzi D, Hermankova M, Pierson T, Carruth LM, Buck C, Chaisson RE, Quinn TC, Chadwick K, Margolick J, Brookmeyer R, Gallant J, Markowitz M, Ho DD, Richman DD, Siliciano RF 1997 Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science 278: 1295–1300.
Wong JK, Hezareh M, Gunthard HF, Havlir DV, Ignacio CC, Spina CA, Richman DD 1997 Recovery of replication-competent HIV despite prolonged suppression of plasma viremia. Science 278: 1291–1295.
Rutstein RM, Feingold A, Meislich D, Word B, Rudy B 1997 Protease inhibitor therapy in children with perinatally acquired HIV infection. AIDS 11: 1487–1494.
Melvin AJ, Mohan KM, Arcuino LA, Edelstein RE, Frenkel LM 1997 Clinical, virologic and immunologic responses of children with advanced human immunodeficiency virus type 1 disease treated with protease inhibitors. Clin Exp Immunol 110: 15–21.
Pakker NG, Notermans DW, de Boer RJ, Roos MT, de Wolf F, Hill A, Leonard JM, Danner SA, Miedema F, Schellekens PT 1998 Biphasic kinetics of peripheral blood T cells after triple combination therapy in HIV-1 infection: a composite of redistribution and proliferation. Nat Med 4: 208–214.
Molla A, Korneyeva M, Gao Q, Vasavanonda S, Schipper PJ, Mo HM, Markowitz M, Chernyavskiy T, Niu P, Lyons N, Hsu A, Granneman GR, Ho DD, Boucher CA, Leonard JM, Norbeck DW, Kempf DJ 1996 Ordered accumulation of mutations in HIV protease confers resistance to ritonavir. J Virol 70: 4220–4227.
Acknowledgements
The authors thank to Dolores García Alonso and Consuelo Muñoz for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by grants from the Fondo de Investigación Sanitaria (00/0207), Fundación para la Investigación y la Prevención del SIDA en España (3008/99), Programa Nacional de Salud (SAF 99-0022), Bristol-Myers, S.A. (Grupo Bristol-Myers Squibb), and Comunidad de Madrid (08.5/0034/2001).
Rights and permissions
About this article
Cite this article
Resino, S., Bellón, J., Sánchez-Ramón, S. et al. CD8+ T-Cell Numbers Predict the Response to Antiviral Therapy in HIV-1-Infected Children. Pediatr Res 53, 309–312 (2003). https://doi.org/10.1203/01.PDR.0000047652.58958.96
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/01.PDR.0000047652.58958.96