Abstract
Recovery of the ability to digest and absorb lipids is essential to the maintenance of normal nutrition in infants with bowel damage. Two intrinsic microsomal enzymes, monoacylglycerol acyltransferase (MGAT) and diacylglycerol acyltransferase (DGAT), catalyze the major pathway for intestinal triacylglycerol biosynthesis. This study describes the effects of intestinal ischemia on epithelial DGAT and MGAT activities and their recovery in response to two luminal treatments: L-glutamine (Gln), the primary intestinal fuel, and transforming growth factor-α (TGF-α), a mitogenic hormone similar to epidermal growth factor present in breast milk. Ischemic damage and recovery were analyzed in mucosa from Thiry-Vella loops in the mid-ileum of 7-wk-old pigs. Loops were subjected to 2-h occlusion of local mesenteric arteries, followed by 6 or 72 h of recovery in the presence of luminal glucose(control), Gln, or TGF-α. Ischemic tissue followed by 6-h recovery exhibited an approximate 50% decrease in both MGAT and DGAT activities compared with nonischemic loop tissue. At 72 h, MGAT and DGAT recovery in Gln plus TGF-α treated loops was significantly greater than their corresponding 6-h peak damage levels (p < 0.05). From 6 to 72 h, MGAT increased 4-fold and DGAT increased 3.6-fold after Gln plus TGF-α treatment. With other treatments, MGAT and DGAT activities increased<2.5-fold from 6 to 72 h. This study shows that intestinal MGAT and DGAT activities decrease after ischemic damage, yet recover rapidly in bowel exposed to Gln and/or TGF-α. By stimulating the rate of recovery of the villi and lipid synthesizing enzymes, these treatments could improve the efficacy of enteral feeding in infants recovering from bowel damage.
Similar content being viewed by others
Main
Malabsorption and maldigestion of nutrients from the small intestinal lumen result from mucosal damage caused by a variety of infectious agents and carry a high rate of mortality in developing countries. We previously studied if specific nutrients known to stimulate intestinal growth would enhance recovery in porcine rotavirus infection(1). Unfortunately, in those studies, although rotavirus produced a dehydrating diarrhea and mortality, villi were reduced to only ≈50% of their normal height after viral infection. Subsequent intestinal repair in this rotavirus model was rapid and was uninfluenced by any of the treatments studied(1). Acute ischemia is seen in infants and toddlers, when they develop necrotizing enterocolitis, volvulus, and a number of nonocclusive disorders such as hemorrhagic shock(2, 3). Intestinal ischemia results in complete sloughing of the villi, followed by the ulceration of surface that was previously covered(4). The aim of the present study was to study whether nutrients can regulate the intestinal repair process in a model of severe bowel injury produced by ischemia.
One complex function of the small intestine is to metabolize nutrient lipids. Lipids, primarily in the form of TAG, comprise about 50 and 38% of the total calories in American infants' and children's diets, respectively(5, 6). By measuring lymph TAG output in rats, Fujimoto et al.(7) and Kerner et al.(8) demonstrated that ischemia causes lipid malabsorption, but it is unclear whether the malabsorption results from failure of fatty acid absorption, reesterification, or chylomycra assembly(7, 8). Because the microsomal enzymes MGAT (EC 2.3.1.22) and DGAT (EC 2.3.1.20) form the major pathway for TAG synthesis in the intestinal mucosa, we hypothesized that the intestinal malabsorption of TAG is due, in part, to a decrease in the activity of MGAT and DGAT. We therefore studied these enzymes during recovery from ischemia in the piglet intestine, an animal model with digestive and absorptive mechanisms similar to those of humans(9).
Gln and TGF-α have been shown to facilitate intestinal repair. Gln is the primary metabolic fuel of small intestinal enterocytes and constitutes more than 50% of the free amino acid pool in the body(10). Gln protects the epithelium from injury, serves as an essential metabolic component of the proliferative response of enterocytes, and reduces atrophy of intestinal mucosa in rats on total parenteral nutrition(11–13). TGF-α, a growth factor closely related to epidermal growth factor found in human milk, enhances proliferation of enterocytes, stimulates restitution, and hastens recovery of small intestinal villus surface area(14). We recently found that Gln plus epidermal growth factor had additive effects on intestinal cellular proliferation and activation of mitogen-activated protein kinases(15). We were therefore interested in the role of Gln and TGF-α in stimulating the postischemic recovery of microsomal MGAT and DGAT. The objectives of this study were to determine whether ischemia altered porcine ileal MGAT and DGAT activities and whether L-glutamine, alone or with TGF-α, improves the recovery of enzyme activities.
METHODS
Animals, surgery, and sample collection. All animal studies were approved by the Institute of Animal Care and Use Committee at North Carolina State University. Seven-week-old female or castrated male pigs(n = 46), weighing approximately 16 kg, were housed under controlled 12-h light cycles with free access to dried food and water. This age was chosen because it corresponds to the period of weaning and is an age at which piglets can survive the surgery for Thiry-Vella loop formation. Pigs were anesthetized with ketamine (11 mg/kg i.m.) and xylazine (1.5 mg/kg i.m.) and maintained during surgery on halothane vaporized in oxygen after orotracheal intubation. During the latter part of surgery, buprenorphine (0.05 mg/kg i.m.) was administered to minimize postsurgical pain.
Thiry-Vella loops (20 cm in length) were created in the mid-ileum, approximately 20 cm proximal to the ileocecal junction. A 10-cm segment of the loop was made ischemic by occluding the local mesenteric arteries for 2 h. Mesenteric vessels were individually occluded 1 cm from the margin of the bowel. Because Johns Hopkins bulldog clamps were used to clamp individual vessels, adjacent segments of the gut were not affected. Collateral supply from the adjacent control segment of the loop was prevented by cross-clamping the loop with a Doyden clamp. A pulse oximeter was applied to the ischemic side of the Thiry-Vella loop to ensure cessation of blood flow and to ensure reperfusion (arterial saturation returning to preischemic levels).
Our previous studies determined that 2 h of ischemia produced near-maximal ischemic damage (grade 4 out of 5 ischemic injury) that retained the ability to recover(16). This degree of injury provided a stringent test of the ability of Gln and TGF-α to stimulate repair and gave us the opportunity to examine the proliferative phase of repair.
The adjacent 10-cm segment of the Thiry-Vella loop was left as a nonischemic loop control. The loop was cannulated at each end with fabricated silicone cannulas that were exteriorized at the left flank. The remaining ileum was anastomosed using a simple continuous suture pattern (2.0 polyglactin 910).
On completion of experiments, the pigs were killed by i.v. injection of sodium pentobarbital (15 mg/kg). Ileal samples were immediately collected and washed with Ringer solution containing (in mEq/L) sodium 140, potassium 5.2, calcium 1.2, magnesium 1.2, chloride 119, HCO3- 25, H2PO4- 0.4, HPO42- 2.4, pH 7.4. Additional samples for histologic analysis were removed and fixed in formalin, embedded in paraffin, cut into 7-μm sections, stained with hematoxylin and eosin, and examined by light microscopy.
For analysis of microsomal lipid synthesizing enzymes, mucosa was scraped with a glass slide, snap frozen in liquid nitrogen, and stored at -70°C until further analysis. Mucosal samples were taken from the ischemic loop, the nonischemic portion of the loop, and a portion of ileum-in-continuity 10 cm proximal to the Thiry-Vella loop site. The ileum-in-continuity had been exposed to the pig diet.
Experimental plan. Four different solutions were manually infused in 120-mL aliquots once a day through the cannula and allowed to dwell for 6-24 h; they were replaced each 24 h for the animals surviving to 72 h. These treatments, in Ringer solution, were glucose (control, 2.5%), Gln (2%), glucose (2.5%) plus 60 μg/L TGF-α, or Gln (2%) plus 60 μg/L TGF-α. These concentrations were chosen so that the solutions were isotonic (280 mosmol). The Gln provided a negligible amount of protein to the piglets (0.03 g·kg-1·d-1). Thiry-Vella loops were treated for 6 h (n = 14 piglets) or 72 h (n = 32 piglets).
Materials. All chemicals were of reagent grade. Phosphatidylcholine (pig liver), phosphatidylserine (beef brain), lipid standards, sn-1,2-diC18:1-glycerol, and sn-2-monoC18:1-glycerol were purchased from Serdary Research Laboratories, Inc., Englewood Cliffs, NJ. [3H]Palmitoyl-CoA was synthesized enzymatically(17). [3H]Palmitate was purchased from DuPont, BSA (essential fatty acid-free) was purchased from Sigma Chemical Co., and silica gel G plates were purchased from Analtech, Newark, DE.
Subcellular fractionation. Microsomes were prepared from porcine ileal samples by differential centrifugation in medium 1 (0.25 M sucrose, 20 mM Tris-HCl, pH 7.4, 1 mM EDTA)(18). The microsomal pellet was resuspended in medium 1 containing 0.5 M KCl and centrifuged at 100 000 × g at 4°C for 1 h. The final KCl-washed intestinal microsomes were resuspended in medium 1 and stored in aliquots at -70°C. Protein concentrations were measured with BSA as the standard(19).
Assay of MGAT in microsomes. MGAT activity was measured using 25 μM [3H]palmitoyl-CoA, 50 μM sn-2-monoC18:1-glycerol, and 2-6 μg of microsomal protein(18). The reaction was initiated by adding [3H]palmitoyl-CoA. After thin layer chromatography on silica gel G plates in heptane/isopropyl ether/glacial acetic acid (60:40:4; vol/vol), the spots corresponding to diacylglycerol and TAG were scraped and counted. MGAT specific activity was calculated by subtracting half the counts/min that appeared in TAG. The enzyme activity dependent on endogenous sn-2-monoC18:1-glycerol was less than 3% and was not routinely measured. All assays contained optimal amounts of sn-2-monoC18:1-glycerol and [3H]palmitoyl-CoA and measured initial rates. Analysis of double reciprocal plots gave an apparent Km value of 14.7 μM for sn-2-monoC18:1-glycerol and a Vmax value of 12.3 nmol·min-1·mg-1(20). The sigmoidal nature of palmitoyl-CoA incorporation into diacylglycerol by MGAT did not permit kinetic calculations for palmitoyl-CoA.
Assay of DGAT in microsomes. DGAT activity was measured using 15 μM [3H]palmitoyl-CoA, 200 μM sn-1,2-diC18:1-glycerol, and 2-6 μg of microsomal protein(18). The reaction was initiated by adding[3H]palmitoyl-CoA. Because more than 75% of the labeled product was TAG, thin layer chromatography was not routinely performed. The enzyme activity using endogenous sn-1,2-diC18:1-glycerol was less than 4% and was not routinely measured. All assays contained optimal amounts of sn-1,2-diC18:1-glycerol and [3H]palmitoyl-CoA and measured initial rates. Double reciprocal plots gave an apparent Km value of 22.7 μM and a Vmax value of 26.8 nmol·min-1·mg-1 for sn-1,2-diC18:1-glycerol and a Km value of 4.1 μM and Vmax value of 24.6 nmol·min-1·mg-1 for [3H]palmitoyl-CoA(19).
Determination of sucrase specific activity. Sucrase was assayed according to methods developed by Dahlqvist(21) and reported as micromoles·g protein-1·min-1. Homogenates were incubated with sucrose for 60 min at 37°C, after which the glucose released by sucrase was determined by a glucose oxidase reaction. Protein was assayed with BSA as the standard(19).
Statistics. Effect of treatments at each time interval was compared with one-way ANOVA. For data that were normally distributed, one-way ANOVA was followed by a Tukey's test to determine the effects of individual treatments. For data that were not normally distributed, one-way ANOVA was performed on ranks, followed by a Dunn's test for individual comparison. p < 0.05 was considered statistically significant. All data are expressed as mean ± SE.
RESULTS
Histologic findings. The experimental protocol resulted in extensive epithelial damage of the ischemic side of the loop, characterized by epithelial sloughing (Fig. 1A). Six hours later, epithelial restitution was underway, with partial villus coverage by cells bearing a squamous shape. Villous contraction was also seen (Fig. 1B). No difference was noted between the appearance of loops treated with glucose (Fig. 1B), Gln, or Gln plus TGF-α (Fig. 1C). After 72 h, the mucosa appeared much differently, with villus coverage by columnar epithelial cells. In glucose-treated loops (Fig. 1D), villi were still rudimentary, with crypt hyperplasia. In Gln + TGF-α-treated loops, villi were taller and more distinct than in similar mucosa treated with glucose (Fig. 1E). Representative figures are shown for each treatment. Morphometric analysis of mucosal restitution and villus regrowth was undertaken and is the subject of a separate study (submitted for review).
(A) Histologic appearance of porcine ileal mucosa immediately after 2 h of ischemia. Note extensive sloughing of epithelium into the lumen, resulting in near-complete denuding of villi(hematoxylin and eosin, original magnification, ×400). (B) Appearance of porcine ileal mucosa from a Thiry-Vella loop, subjected to 2 h of ischemia, and recovered for 6 h after treatment with Ringer's solution containing glucose (2.5%). Note evidence of epithelial restitution (partial coverage of the proximal villus with flattened epithelium) and villous contraction; 1-cm bar = 50 μm. (C) Porcine ileal mucosa from Thiry-Vella loop, subjected to 2 h of ischemia, and recovered for 6 h after treatment with Ringer's solution containing L-glutamine (2%) and TGF-α(60 μg/L). Mucosa appears similar to that treated with Ringer's solution containing glutamine; 1-cm bar = 50 μm. (D) Ileal mucosa from a Thiry-Vella loop subjected to 2 h of ischemia and recovered for 72 h after daily treatment with Ringer's solution containing glucose (2.5%). Although villi are completely covered in columnar epithelium, villi are markedly stunted, and crypt hyperplasia is seen; 1-cm bar = 100 μm. (E) Histologic appearance of porcine ileal mucosa from a Thiry-Vella loop, subjected to 2 h of ischemia, and recovered for 72 h after daily treatment with Ringer's solution containing glutamine (2%) and TGF-α (60 μg/L). Villi are taller and more distinct than in similar mucosa treated with glucose; 1-cm bar = 100 μm.
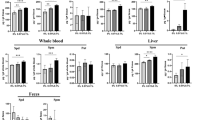
Regional distribution of MGAT and DGAT activities in piglet intestine. MGAT and DGAT activities in normal duodenum, jejunum, and ileum from 7-wk-old pigs fed standard diet were approximately 3.8- and 2.8-fold greater, respectively, in the proximal small intestine compared with the distal small intestine (Fig. 2). Both acyltransferase activities in the distal small intestine were significantly lower than those in the proximal small intestine (p < 0.05), suggesting that the majority of TAG biosynthesis occurs proximally but that TAG biosynthesis can occur in the ileum.
MGAT and DGAT activities in pig duodenum, jejunum, and ileum. KCl-washed microsomes were prepared from duodenum, jejunum, or ileum from the same experimental 7-wk-old pigs undergoing surgery. MGAT and DGAT activities expressed as mean ± SE. *For MGAT and DGAT, significantly different from jejunum and duodenum, p < 0.05. The number of samples per group is shown at the bottom of each bar.
Effects of ischemic damage and substrate availability on the activities of MGAT and DGAT. The specific activity in the nonischemic(NI) and ischemic (I) portions of the loop showed inter-animal variability (Figs. 3 and 4). As a group, the enzyme activities in nonischemic Thiry-Vella loop mucosa were lower than in the ileum-in-continuity. This difference in activity approached statistical significance (p < 0.06). To determine the effect of ischemia on the activities of MGAT and DGAT in ileal loops, the local mesenteric arteries were occluded for 2 h and then allowed to recover for 6 or 72 h. As a group at 6 h, MGAT and DGAT activities decreased in ischemic ileum by approximately 50% compared with activities in nonischemic ileum (Figs. 3 and 4). There were no statistically significant differences in MGAT and DGAT activity between treatment groups, 6 or 72 h. At 72 h postischemia, intestine exposed to glucose plus TGF-α, Gln, or Gln plus TGF-α showed enzyme activities that were equivalent to values in the nonischemic bowel (Figs. 3 and 4).
Effect of glucose, Gln, and TGF-α on the recovery of MGAT activity in control and ischemic intestine. Ileal loops were exposed to 2-h nonischemia (NI) or ischemia (I) and allowed to recover in glucose (2.5%) for 6 h (A), 72 h (B); in glucose (2.5%) and TGF-α (60 μg/L) for 72 h (C); in Gln(2%) for 6 h (D), or 72 h (E); or in Gln (2%) and TGF-α (60 μg/L) for 6 h (F), or 72 h (G). Each line segment represents a comparison, for the same pig, of MGAT activity between nonischemic and ischemic intestinal mucosa. MGAT activity in normal control ileum, measured in mucosal samples from the same experimental pigs, is expressed as mean ± SE; n = 6.
Effect of glucose, Gln, and TGF-α on the recovery of DGAT activity in control and ischemic intestine. Ileum loops were exposed to 2-h nonischemia (NI) or ischemia (I) and allowed to recover under the same experimental condition as described in Figure 3. Each line segment represents a comparison, for the same pig, of DGAT activity between nonischemic and ischemic intestinal mucosa. DGAT in normal control ileum, measured in mucosal samples from the same experimental pigs, is expressed as mean ± SE; n = 7.
Recovery of MGAT and DGAT activity between 6 and 72 h. For both MGAT and DGAT activities, the levels at 72 h in the groups treated with Gln plus TGF-α were significantly different from their corresponding 6-h peak damage levels (p < 0.05) (Figs. 5 and 6). Differences for the other treatments did not reach statistical significance. MGAT and DGAT activity increased between 6 and 72 h, with 4- and 3.6-fold increases, respectively, after Gln plus TGF-α treatment. Increases in MGAT and DGAT recovery, in tissues treated with Gln alone, were 2.4- and 2.3-fold, respectively (NS).
Recovery of MGAT activity in ischemic ileum loops treated with glucose, Gln, or TGF-α as a percent of the mean nonischemic control loop. Ileum loops were exposed to 2-h nonischemia or ischemia and allowed to recover for 6 or 72 h in the indicated treatment solutions. The bars show MGAT activity in the ischemic loop divided by mean MGAT activity in nonischemic loops multiplied by 100. Treatment and recovery time designations are similar to those described in Figure 3. No 6-h glucose plus TGF-α group was designed, therefore, no comparison was made between a 6- and 72-h glucose plus TGF-α group. *Significantly different from the 6-h L-glutamine plus TGF-α group (p < 0.05). The percent enzyme activity is expressed as mean ± SE. The number of samples per group is shown at the bottom of each bar.
Recovery of DGAT activity in ischemic ileum loops treated with glucose, Gln, or TGF-α as a percent of the mean nonischemic control loop. Experimental procedures are identical to those described in Figure 5. *Significantly different from the 6-h Gln plus TGF-α group (p < 0.05). Percent enzyme activity is expressed as mean ± SE. The number of samples per group is shown at the bottom of each bar.
Differential effects of glucose, Gln, and TGF-α treatments on sucrase activity after 72-h recovery. Sucrase is a brush border enzyme present in differentiated cells on the villus but not in the undifferentiated crypt cells. Sucrase specific activity of mucosal homogenates was only 40% of normal in glucose-treated ileum, but was ≈75-100% of normal in loops exposed to Gln, glucose plus TGF-α, or Gln plus TGF-α. However, there were no statistically significant differences comparing sucrase activity among the different treatment groups (Fig. 7).
Differential effects of glucose, Gln, and TGF-α on sucrase, MGAT, and DGAT activities after 72-h postischemic recovery. Ileal loops were exposed to 2-h nonischemia or ischemia and allowed to recover for 72 h in the indicated treatment solutions. Mucosa was collected and assayed as described in “Methods.” The bars show sucrase, MGAT, and DGAT activity in the ischemic loop divided by enzyme activity in nonischemic loops multiplied by 100. Experimental procedures are the same as described in Figure 3 for the 72-h groups. Sucrase, MGAT, and DGAT activities are expressed as mean ± SE. n = 8 pigs in each of the treatment groups.
DISCUSSION
Once sn-2-monoacylglycerols and fatty acids enter the cytosol of enterocytes, these neutral lipid components move to the smooth endoplasmic reticulum where MGAT and DGAT catalyze the sequential acylation of sn-2-monoacylglycerol to sn-1,2-diacylglycerol to TAG(5, 22). This monoacylglycerol pathway constitutes the major route of TAG synthesis (75-85%) from luminally absorbed monoacylglycerol and fatty acids(23). It is widely accepted that most lipid absorption occurs in the duodenum and jejunum(5, 6, 22, 23). However, the ileum plays a pivotal role in lipid absorption(24–26). We detected MGAT and DGAT activity throughout the porcine small intestine, but MGAT and DGAT activity decreases from proximal to distal small intestine, a finding consistent with previous reports in rats(26–28).
Piglets were used in these experiments because of the similar physiology of porcine bowel to that of human bowel(9). A nonischemic control segment of the Thiry-Vella loop was used to control for changes in villus morphology and enzyme levels that may occur due to lack of substrate availability and possible mild tissue damage in an isolated Thiry-Vella loop. The exposure of the ileum to 2 h of ischemia led to the necrosis and subsequent sloughing of enterocytes found along the villus. Because these enzymes exist in differentiated enterocytes, proliferating cells were predicted to have lower enzyme activity.
To our knowledge, no studies in the literature have reported the effects of ischemia on intestinal lipid biosynthetic enzymes. A study by Fujimoto et al.(7) assessed the lipid output in lymph after a 10-min occlusion of the superior mesenteric artery of rat intestine. Their study suggested that, in rats exposed to brief ischemia, the structure of the intestinal mucosa was altered such that there was increased transport of radioactive TAGs via the portal route rather than the lymphatic route(7). A study by Kerner et al.(8) assessed the posttrauma hemorrhage lymph lipid output in rats. Their study suggested that the lipid absorptive capacity of enterocytes was significantly decreased after trauma-hemorrhage and resuscitation(8).
The roles of Gln and TGF-α as treatments were studied in the reversal of damage caused by intestinal ischemia. TGF-α, endogenously produced in the small intestine(29), and Gln play a role in increasing the rate of villus recovery(30). We used TGF-α because it is extensively produced by the small intestinal enterocytes and may be a more physiologic ligand for the epidermal growth factor receptor in the small intestine(29). Glutamine is necessary for transduction leading to proliferation in enterocytes and decreases intestinal ischemia/reperfusion-induced cell membrane lipid peroxidation(31–34). TGF-α enhances epithelial cell restitution in vitro and hastens intestinal epithelial morphologic (but not functional) recovery in piglets with rotavirus diarrhea, a disease associated with mucosal damage(14, 35).
The difference in enzyme activity between the ileum-in-continuity and the nonischemic samples suggests a trend toward diminished acyltransferase activity in uninjured Thiry-Vella loops. Mucosal atrophy within Thiry-Vella loops has been previously reported(36). Although the decrease in activity might have been caused by mild tissue damage induced by the systemic or local effects of the surgical procedure, the mucosa appeared normal histologically. The decrease in enzyme activity may have been caused by the lack of luminal nutrients, because none of the treatment preparations contained lipids.
Ileal loops exposed to 2 h of ischemia and subsequent 6- or 72-h recovery with glucose, Gln, TGF-α, or Gln plus TGF-α treatments showed increases in MGAT and DGAT activities as the villi were restored (Figs. 5 and 6). Looking at both MGAT and DGAT, only the 72-h group treated with Gln plus TGF-α had values significantly different from the corresponding 6-h “peak injury” group(p < 0.05). The difference between MGAT activity with glucose plus TGF-α and Gln plus TGF-α (versus glucose treated group) approached significance (p = 0.076) (Fig. 5). With DGAT, no significant differences in treatments were detected. Morphologic analysis of the cells covering the villi at 72 h of recovery indicated that the mucosal surface area had been restored to normal only in the Gln plus TGF-α group(37). The Gln and TGF-α treatment may have caused a greater increase in lipid-synthesizing enzyme activity by allowing the intestinal cells to proliferate and differentiate, thereby more rapidly regenerating the villi. It has been shown that differentiation is triggered by positional events along the villus-crypt axis, rather than enterocyte “lifespan”(38).
After 72 h, MGAT and DGAT expressed similar rates of recovery within each treatment group (Figs. 5 and 6). A similarity was also observed between MGAT and DGAT activities in different regions of the small intestine (Fig. 2). Because both enzymes are involved in the monoacylglycerol pathway for TAG synthesis, it is logical that both enzymes recover at similar rates and be expressed at similar levels. However, because antibodies and cDNA to purified MGAT and DGAT are unavailable, one cannot assess how each enzyme is affected by time, luminal treatment, or regional distribution.
Of the two “target” treatments, both have stimulatory effects on small intestinal cell proliferation and on ornithine decarboxylase, the first and rate-limiting enzyme in polyamine biosynthesis(31). Activity of ornithine decarboxylase in producing the polyamine putrescine, and subsequent generation of spermidine and spermine appears to be an essential step in bowel repair after ischemia/reperfusion injury(39). Two days after 15-min ischemia in the rat intestine, Fujimoto et al.(40) showed that lipid esterification to TAG and delivery into lymph returned to normal, and the mechanism was dependent on mucosal ornithine decarboxylase induction and production of histamine. In these studies, one may hypothesize that villus tips were rapidly replaced and, as in the current study, microsomal lipid reesterification enzymes rapidly returned to normal levels.
Our data clearly show that MGAT and DGAT activities recover as early or earlier than brush border sucrase, required for sugar hydrolysis and absorption (Fig. 7). This finding is in keeping with studies of infants with protracted diarrhea of infancy who gained weight more rapidly when treated with a semielemental diet with a 48%:41% fat:carbohydrate calorie ratio, compared with infants treated with the same formula with a 35%:54% fat:carbohydrate ratio(41). Thus, although our study cannot be applied to human infants, it does bring into question the practice of reducing dietary fat (e.g. by formula dilution) in infants with acute intestinal injury (for example, viral diarrhea).
One limitation of our study is that a 6-h glucose plus TGF-α treatment group was not studied; and therefore the increase from 6 to 72 h attributable to TGF-α alone could not be assessed. We did not anticipate that TGF-α would have had a significant effect, based on our previous studies, which showed that TGF-α treatment by itself stimulated proliferation, but not differentiation, after rotaviral damage to the small intestine(14). Because MGAT and DGAT activities increased to approximately 100% of normal in both groups that received TGF-α, an effect of TGF-α on villus regrowth (and not necessarily on subsequent differentiation) was suspected. A trend toward increased sucrase, MGAT, and DGAT activities in the Gln-treated but not in the glucose-treated group was also seen (Fig. 7). Another limitation of our study was that we did not determine the postischemic recovery of MGAT and DGAT activities in ileal loops exposed to a normal physiologic diet. Finally, we observed considerable variability in enzyme activities (Figs. 3 and 4), which may have reduced the power of our multiple comparison analysis. A previous analysis of rat intestinal MGAT and DGAT activities demonstrated similar variability(27).
We conclude that MGAT and DGAT activities recover rapidly during villus regeneration in piglet intestine subjected to severe injury. Dietary Gln with TGF-α has a greater potential to stimulate a rapid recovery of these lipid synthesizing enzymes in the intestine when compared with a standard glucose solution.
Abbreviations
- Gln:
-
L-glutamine
- TGF-α:
-
transforming growth factor-α
- MGAT:
-
monoacylglycerol acyltransferase
- DGAT:
-
diacylglycerol acyltransferase
- TAG:
-
triacylglycerol
References
Rhoads JM, Gomez GG, Goforth R, Argenzio RA, Neylan MJ, Gomez GG 1996 Can a “super” oral rehydration solution(“super ORS”) stimulate intestinal repair in acute viral enteritis?. J Diarrhoeal Dis Res 14: 175–181.
Fink MP 1991 Gastrointestinal mucosal injury in experimental models of shock, trauma, and sepsis. Crit Care Med 19: 627–641.
Bynum TE 1989 Vascular insufficiency of the bowel. In: Chopra S, May RJ (eds) Pathophysiology of Gastrointestinal Diseases. Little, Brown, Boston, pp 219–228.
Barnes GL 1996 Intestinal viral infections. In: Walker WA(ed) Pediatric Gastrointestinal Disease. Pathophysiology, Diagnosis, and Management. Decker, Philadelphia, pp 538–556.
Thomson ABR, Schoeller C, Keelan M, Smith L, Clandinin MT 1993 Lipid absorption: passing through the unstirred layers, brush-border membrane, and beyond. Can J Physiol Pharmacol 71: 531–555.
Tso P 1995 Gasrointesinal Physiology: motility, digestion, and absorption. In: Rhoads RA, Tanner GA (eds) Medical Physiology, 1st Ed. Little, Brown, Boston, pp 505–551.
Fujimoto K, Price VH, Granger DN, Specian R, Bergstedt S, Tso P 1991 Effect of ischemia-reperfusion on lipid digestion and absorption in rat intestine. Am J Physiol 260:G595–G602.
Kerner J, Wang P, Chaudry IH 1995 Impaired gut lipid absorptive capacity after trauma-hemorrhage and resuscitation. Am J Physiol 269: R869–R873.
Tumbelson ME 1986 Swine in Biomedical Research. Plenum Press, New York
Helton SW, Smith RJ, Wilmore DW 1991 Glutamine-supplemented nutrition following radiation and chemotherapy. In: Roche AF (ed) Role of Nutrients in Cancer Treatment, Report of the Ninth Ross Conference on Medical Research. Ross Laboratories, Columbus, OH, pp 95–99.
Fox AD, Kripke SA, DePaula J, Berman JM, Settle RG, Rombeau JL 1988 Effect of a glutamine-supplemented enteral diet on methotrexate induced enterocolitides. J Parenter Enteral Nutr 12: 325–331.
O'Dwyer ST, Smith RJ, Hwang TL, Wilmore DW 1989 Maintenance of small bowel mucosa with glutamine-enriched parenteral nutrition. J Parenter Enteral Nutr 13: 579–585.
Jacobs DO, Evans DA, Mealy K, O'Dwyer ST, Smith RJ, Wilmore DW 1988 Combined effects of glutamine and epidermal growth factor on the rat intestine. Surgery 104: 358–364.
Rhoads JM, Ulshen MH, Keku EO, Chen W, Kandil HM, Woodward JP, Liu SC, Fuller CR, Leary HL Jr, Lecce JG 1995 Oral transforming growth factor α enhances jejunal mucosal recovery and electrical resistance in piglet rotavirus enteritis. Pediatr Res 38: 173–781.
Rhoads JM, Argenzio RA, Chen W, Rippe RA, Westwick JK, Cox AD, Berschneider HM, Brenner DA 1997 L-Glutamine stimulates intestinal cell proliferation and activates mitogen-activated protein kinases. Am J Physiol 272:G943–G953.
Blikslager AT, Roberts MC, Rhoads JM, Argenzio RA 1997 Is reperfusion injury an important cause of mucosal damage after porcine intestinal ischemia?. Surgery 121: 526–534.
Merrill AH, Gidwitz S, Bell RM 1982 Facile enzymatic synthesis of fatty acylcoenzyme A thioesters. J Lipid Res 23: 1368–1373.
Coleman RA 1992 Diacylglycerol acyltransferase and monoacylglycerol acyltransferase from liver and intestine. Methods Enzymol 209: 98–104.
Lowry OH, Rosebrough NJ, Farr RA, Randall RJ 1951 Protein measurements with the Folin phenol reagent. J Biol Chem 193: 265–275.
Lineweaver H, Burke DJ 1934 The determination of enzyme dissociation constants. J Am Chem Soc 56: 658–666.
Dahlqvist A 1964 Method for assay of intestinal disaccharidases. Anal Biochem 7: 18–25.
Kutchai HC 1993 The gastrointestinal system: motility, digestion, and absorption. In: Berne RM, Levy MN (eds) Physiology, 3rd Ed. Mosby-Year Book, St Louis, MO, pp 615–685.
Johnston JM 1977 Gastrointestinal tissue. In: Snyder F(ed) Lipid Metabolism in Mammals, Vol. 1. Plenum Press, New York, pp 151–187.
Lin CH, Zhao XT, Wang L 1996 Fat absorption is not complete by midgut but is dependent on load of fat. Am J Physiol 271:G62–G67.
Kremen AJ, Linner JH, Nelson CH 1954 An experimental evaluation of the nutritional importance of proximal and distal small intestine. Ann Surg 140: 439–448.
Rodgers JB Jr, Bochenek W 1970 Localization of lipid reesterifying enzymes of the rat small intestine: effects of jejunal removal on ileal enzyme activities. Biochim Biophys Acta 202: 426–435.
Grigor MR, Bell RM 1982 Separate monoacylglycerol and diacylglycerol acyltransferases function in intestinal triacylglycerol synthesis. Biochim Biophys Acta 712: 464–472.
Jamdar SC, Cao WF 1995 Triacylglycerol biosynthetic enzymes in lean and obese Zuker rats. Biochim Biophys Acta 1255: 237–243.
Barnard JA, Beauchamp RD, Russell WE, Dubois RN, Coffey RJ 1995 Epidermal growth factor-related peptides and their relevance to gastrointestinal pathophysiology. Gastroenterology 108: 564–580.
Windmueller HG. 1982 Glutamine utilization by the small intestine. Adv Enzymol 53: 201–237.
Kandil HM, Argenzio RA, Chen W, Berschneider HM, Stiles AD, Westwick JK, Rippe RA, Brenner DA, Rhoads JM 1995 L-Glutamine and L-asparagine stimulate ODC activity and proliferation in a porcine jejunal enterocyte line. Am J Physiol 269:G591–G599.
Ko TC, Beauchamp RD, Townsend CM, Thompson JC 1993 Glutamine is essential for epidermal growth factor-stimulated intestinal cell proliferation. Surgery 114: 147–153.
Turowski GA, Rashid Z, Hong F, Madri JA, Basson MD 1994 Glutamine modulates phenotype and stimulates proliferation in human colon cancer cell lines. Cancer Res 54: 5974–5980.
Harward TR, Coe D, Souba WW, Klingman N, Seager JM 1994 Glutamine preserves gut glutathione levels during intestinal ischemia/perfusion. J Surg Res 56: 351–355.
Dignass AU, Podolsky DK 1993 Cytokine modulation of intestinal epithelial cell restitution: central role of transforming growth factor β. Gastroenterology 105: 1323–1332.
Echnauer R, Feyeraband G, Raffler H 1978 Analysis of the effects of food and digestive secretions on the small intestine of the rat. III. Mucosal mass, activity of brush border enzymes and in vivo absorption of galactose, sodium, and potassium. Gut 19: 707–714.
Blikslager AT, Bristol DG, Rhoads JM, Roberts MC, Argenzio RA 1996 Glutamine and transforming growth factor alpha enhance repair of ischemia/reperfusion injury. Gastroenterology 10:A313.
Hermiston ML, Wong MH, Gordon J I 1996 Forced expression of E-cadherin in the mouse intestinal epithelium slows cell migration and provides evidence for nonautonomous regulation of cell fate in a self-renewing system. Genes Dev 10: 985–996.
Fujimoto K, Granger DN, Price HV, Tso P 1991 Ornithine decarboxylase is involved in repair of small intestine after ischemia-reperfusion in rats. Am J Physiol 261:G523–G529.
Fujimoto K, Imamura I, Granger DN, Wada Hiroshi Sakata T, Tso P 1992 Histamine and histidine are correlated with mucosal repair in rat small intestine after ischemia-reperfusion. J Clin Invest 89: 126–133.
Jirapinyo P, Young C, Srimaruta N, Rossi TM, Cardano A, Lebenthal E 1990 High-fat semi-elemental diet in the treatment of protracted diarrhea of infancy. Pediatrics 86: 902–908.
Acknowledgements
The authors thank Ping Wang, Dr. R. Ariel Igal, Martha Armstrong, and Dr. John J.B. Anderson for their helpful advice.
Author information
Authors and Affiliations
Additional information
Supported by HD19068 from the National Institutes of Health (R.A.C.), 2-61990 from the North Carolina Institute of Nutrition (B.G.B.), 94-37204-0448 from the United States Department of Agriculture (R.A.A.), and by a pilot feasibility study from the Center for the Study of Gastrointestinal Biology and Disease 5-P30-DK34987 (A.T.B.).
Rights and permissions
About this article
Cite this article
Ahdieh, N., Blikslager, A., Bhat, B. et al. L-Glutamine and Transforming Growth Factor-α Enhance Recovery of Monoacylglycerol Acyltransferase and Diacylglycerol Acyltransferase Activity in Porcine Postischemic Ileum. Pediatr Res 43, 227–233 (1998). https://doi.org/10.1203/00006450-199802000-00012
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199802000-00012