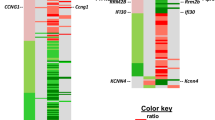
Abstract
The XRCC1 (X-Ray Repair Cross C omplementing) gene was described to play a role for the sensitivity of mammalian cell lines toward ionizing irradiation. Cells with a mutation of this gene present with decreased single strand break repair and reduced recombination repair, they show increased double strand breaks, and sister chromatid exchange is increased up to 10-fold. The goal of our study was to investigate the transcription of this gene in the heart after ionizing irradiation in the mouse. Furthermore, we intended to examine whether radiation-sensitive mice would show a transcriptional pattern different from radiation-resistant mice. Radiation-sensitive BALB/c/J Him mice and radiation-resistant C3H He/Him mice were whole body irradiated with x-ray at 2, 4, and 6 Gy and killed 5, 15, and 30 min after irradiation. mRNA was isolated from the heart and hybridized with probes for XRCC1 and β-actin as a housekeeping gene control. Irradiation at 2 Gy showed increased transcription of XRCC1 at 5 min in the C3H He/Him group, approached XRCC1 transcription of BALB/c J/Him mice at 15 min, and was lower in the latter at 30 min after irradiation. Irradiation at 4 Gy showed double the transcription at 5 min and an about 3-fold rapid increase of mRNA XRCC1 in the radiation-resistant group at 15 min after irradiation, returning to the transcriptional level of sensitive animals at 30 min. Irradiation at 6 Gy seemed to overwhelm the system in both groups, but resistant mice still showed higher levels of XRCC1 transcription. We conclude that radiation-resistant mice show a higher transcription level for the XRCC1 gene in the heart early after x-ray whole body irradiation. This finding is the first in vivo study on XRCC1 of this kind and may in part explain the differences in the radiation sensitivity between the two strains studied.
Similar content being viewed by others
Main
The XRCC1 gene complements the deficiency of the RS CHO mutant EM9(1, 2). There is no detectable expression of the XRCC1 gene in this cell line(3). This mutant is not only more sensitive against ionizing irradiation(4, 5) but also against ethylmethansulfonate, alkylating agents(4) hydrogen peroxide, topoisomerase I-inhibitor camptothecin(6, 7), and UV light at 265 nm(8). The molecular basis for the increased susceptibility toward these noxes in the EM9 cell line is given by the decreased single strand break repair(9), reduced recombination repair(10), increased double strand breaks(11), and a 10-fold increase of sister chromatid exchange compared with the parental cell line AA8(12, 13). As recently as in 1994 the Xrcc1 gene was isolated in the mouse system. It is 92% homologous to the human XRCC1 gene(14). This discovery enabled us to study the differences in radiation sensitivity between two mouse strains, a RS (BALB/c J/Him) and a RR strain (C3H He/Him).
We decided to study transcriptional patterns in total ventricular tissue of these animals and selected the heart as a whole organ as any cultivation or handling of individual cells could have had confounding influences on the outcome at the transcriptional level. A transcriptional pattern of XRCC1 consistent with the phenotypes “resistant” or“sensitive” could be revealed in this first report on an in vivo experiment.
METHODS
Animals and irradiation protocol. Sixty female BALB/c/JHim (RS) and 60 female C3H/HeHim (RR), mean age 90 ± 3 d, were used in the experiments. Their differences in response to ionizing irradiation is well known and documented(15). All experiments were carried out with the permission of the German Committee on Animal Experiments (Bonn). Animals were divided into a RS and RS control group, and the other six (three RS and three RR) groups were irradiated with 2, 4, and 6 Gy. Mice were killed by neck dislocation after 5, 15, and 30 min. Each group consisted of six animals. Whole body irradiation was performed with a x-ray tube 200 RT (C.H.F. Mueller, Germany). 200 kV, 20 mA, 0.1-mm Cu filters, and a dose rate of 2 Gy/min was applied.
mRNA isolation from mouse heart/Northern blots and slot blots. The organs were obtained at autopsy and taken immediately into liquid nitrogen. Frozen heart samples were ground, and mRNA extraction was performed using the Quick Prep Micro mRNA Purification kit (Pharmacia Biotech Inc.). Subsequently, mRNA was applied onto a 1.4% agarose gel after denaturation with glyoxal and DMSO according to the method by McMaster and Carmichael(16) and electrophoresed at 3-4 V/cm for 2.5 h in circulating 0.01 M phosphate buffer, pH 7.0. RNA was then transferred to a positively charged nylon membrane (Hybond N+, Dupont, NEF 986) by capillary blotting(17) and fixed with 0.05 N sodium hydroxide for 5 min at room temperature and finally equilibrated at pH 7.0 with three washes in 2 × SSC.
Probes for human β-actin (ATCC 9800) and XRCC1 (plasmid pCD2EX with cloned human XRCC1 cDNA was generously supplied by Dr. L. H. Thompson, Lawrence Livermore National Laboratory, Canada) were used for Northern and slot blots.
For transfection, an aliquot of frozen competent cells (Escherichia coli HB 101) was thawed to 0°C, and 5 μL of the plasmid (5 μg/100μL) were incubated with the competent cells on ice, followed by an incubation step at 90 s at 42°C and returning the tubes to the ice bath for 2 min. Subsequently, 1 mL of prewarmed (37°C) SOC medium was added and incubated for 60 min on a shaker. Then tubes were centrifuged for 10 min at 4500 × g, and the pellet was resuspended in LB medium at three different dilutions and plated in LB agar medium plus ampicillin (100μg/mL) where they were allowed to grow overnight at 37°C.
A single bacterial colony was transferred to 4 mL of LB medium containing ampicillin, and the culture was incubated overnight at 37°C with vigorous shaking. One milliliter of the tube was inoculated in 500 mL of LB medium containing ampicillin prewarmed to 37°C in a 2-L flask; this culture was incubated under vigorous shaking until the OD at 600 nm was 0.4.
For the isolation of the pcD2EX plasmid the plasmid Maxi kit (Omega 12145), based upon a modified alkaline lysis procedure, was used followed by the binding of plasmid DNA to an anion-exchange resin under appropriate low salt and pH conditions. Plasmid DNA is eluted by this principle by a high salt buffer and concentrated by isopropanol precipitation.
For the digestion with restriction enzymes, 3 μg of plasmid, 2 μL of SuRECut buffers for restriction endonucleases (Boehringer Mannheim), 2 μL of BamHI, 2 μL of EcoRI (Boehringer Mannheim), and 9μL of water were incubated for 3 h in a thermoblock at 37°C. After the reaction was stopped, the solution was electrophoresed on 1% agarose gel in 0.15 M Tris borate buffer, pH 8.0, to show the length of fragments.
Using QUIAquick gel extraction (QUIAGEN 28704), a method with selective DNA binding properties of a silica gel membrane, the DNA probe was isolated and quantified at 260 nm. The probe was denatured before labeling by boiling for 5 min and subsequent cooling on ice, and labeled with fluorescein-12 dUTP using the Renaissance Random Primer Fluorescein-12 dUTP labeling kit (Dupont, NEL 203).
After fixation of bound RNA, the nylon membrane was incubated in prehybridization solution [0.25 M phosphate buffer, pH 7.2, containing 5% SDS wt/vol, 1 mM EDTA, and 0.5% blocking reagent (from Dupont NEL 203)] for 12 h at 65°C in a hybridization oven. The blots were hybridized overnight at 65°C with the labeled probes each (50 ng/mL of prehybridization buffer).
After hybridization, nonspecifically bound material was removed by posthybridization washes with 0.5 × and 0.1 × prehybridization buffer 2 × 10 min each at 65°C. The 0.5 × and 0.1 × prehybridization buffer was brought up to 65°C before use and the second wash was performed at room temperature. Hybridized blots were blocked with 0.5% blocking reagent in 0.1 M Tris-HCl, pH 7.2, and 0.15 M NaCl for 1 h at room temperature. Membranes were then incubated with anti-fluorescein horseradish peroxidase antibody (Dupont NEL 203) at a 1:1000 dilution in the solution given above for 1 h under constant shaking.
Membranes were washed four times for 5 min each in the solution given above. The nucleic acid chemiluminescence reagent (Dupont NEL 201) was added to the membranes and incubated for 1 min. Excess detection reagent was removed by the used of filter paper, and the membrane was placed in Saranwrap and exposed to autoradiography Reflection films (Dupont NEF 496) for 15 min at room temperature.
Slot blots were performed according to the method of White and Bancroft(18). This procedure consisted of placing 2 μg of total RNA dissolved in 10 μL of double-distilled water mixed with 500 μL of 100% formamide, 162 μL of 37% formaldehyde, and 100 μL of 10 × 4-morpholinepropanesulfonic acid buffer. This mixture was incubated for 10 min at 65°C and cooled subsequently on ice. Samples were placed onto the membrane by the Manifolds filtration equipment (slot blot apparatus Bio slot, Bio-Rad), and hybridization was performed as described above. Denistometry of films was performed using the Hirschmann elscript 400 densitometer(Germany).
Statistical methods. The analysis of variation with subsequent Kruskal Wallis test and t test was applied. The level of significance was at p < 0.05).
RESULTS
The results are demonstrated in Figure 1 and are listed numerically in Table 1. No differences for the transcription of mRNA XRCC1/mRNA β-actin (normalization by a house-keeping gene subsequently described as XRCC transcription) could be found between the unirradiated mouse strains.
At low dose (2 Gy) XRCC transcription in the RR mice was significantly higher as early as after 5 min after irradiation. The levels in RS mice approached those of RR mice at 15 min but were significantly lower at 30 min.
At 4 Gy, RR mice significantly doubled the XRCC transcription with significantly depressed XRCC transcription in the RS mice as observed 5 min after irradiation. At 15 min a comparable transcription to the 5-min pattern was found, and again at 30 min, XRCC transcription of both strains were convergent. At 6 Gy, RR mice showed XRCC transcription not statistically different from that in unirradiated mice in contrast to RS mice, which showed significantly depressed XRCC1 transcription, which persisted until 30 min after irradiation. XRCC1 transcription in RR mice was also overwhelmed by high dose irradiation at 30 min after irradiation.
The Northern blot (Fig. 2A) revealed a single band each for XRCC1 at 2.2 kb and at 1.9 kb for β-actin, demonstrating the specific probe and mRNA hybridization product given in the slot blots (Fig. 2B).
DISCUSSION
The finding of higher XRCC1 transcription in hearts of RR mice than in RS animals even in the absence of irradiation corresponded with the biologic susceptibility against ionizing irradiation of these two strains used in the experiments. This fact may be biologically important as any delayed transcriptional response of this repair system could have deleterious effects. It was intriguing to note the clear differences of early (5 min) transcription in the RR mice after even low dose irradiation (2 Gy). At 4 Gy the RR mice significantly increased XRCC transcription, doubling it at 5 and increasing it 3-fold at 15 min. The clear-cut different XRCC transcription in the RR was also reflected in the 6-Gy irradiation experiments when RS mice rapidly reduced XRCC transcription in contrast to RR, which decreased it at a later time point (30 min).
We, therefore, could link the phenotype of radiation sensitivity to the transcription of the XRCC1 gene. The very few x-ray studies in the mammalian system could not find this association, however. Shung and co-workers(19) studied the effect of x-rays on the transcription of the XRCC1 gene in human cell lines. They found little radiation-induced effects on XRCC1 transcription in normal cells but a significant response in ataxia teleangiectasia cells, an effect they assigned to the“hyperradiosensitivity” of these cells. Their observation is not contradictory to our findings, as our in vivo irradiation studies are not comparable to their in vitro irradiation procedures, and organ- and species-specific differences must be taken into accounts as well. We preferred in vivo studies using whole body irradiation and isolating mRNA from intact tissues rather than cells, as handling and culturing cells in artificial media may be confounding factors.
Yoo and co-workers(20) found similar levels of steady-state mRNA XRCC1 in normal cells, Bloom's syndrome cells with altered sister chromatid exchange, and in squamous carcinoma cells with different x-ray sensitivity. In their study mRNA of XRCC1 decreased to 20-40% after treatment of cells with a DNA-damaging agent, which may reflect decreased mRNA of XRCC1 in our high dose irradiation protocol at 6 Gy (Fig. 1).
Dunphy and co-workers(21) studying the expression of the polymorphic human DNA repair gene XRCC1 in cells of human head and neck tumor cell lines could not find any correlation between radiation sensitivity and mRNA of XRCC1. Transcription of this gene also failed to correlate with radiobiologic parameters, clinical outcome, or the DNA restriction fragment length polymorphisms(21).
In our opinion, any of the studies cited above, however important they are, deal with a too complex biologic situation, ranging from tumors to genetic disease. We decided to start with investigations into two entirely healthy, genetically well defined animal systems differing in their (known) susceptibility toward x-rays only(15).
In biochemical terms, the meaning of our findings is that RR mice have a more effective DNA repair. As ionizing irradiation produces a broad spectrum of DNA damage, the subsequent DNA repair cannot be assigned to an individual repair mechanism. A series of studies point to the involvement of the XRCC1 gene in step(s) of DNA repair common to these different repair mechanisms(5). It could be shown that a deficient XRCC1 gene leads to an impaired DNA ligase III activity(22); XRCC1 protein forms a complex with this ligase, thus activating this enzyme(23). And indeed, DNA ligase III activity in contrast to other ligases is significantly reduced in the XRCC1-deficient RS mutant EM9(23). In addition, significant homology between a mouse Xrcc1 gene 93-bp fragment, a yeast radiation damage repair gene rad4+(24), and a recombination activator protein, rag2 from mouse, chicken, and rabbit, exists(25).
The animal model of in vivo radiation sensitivity/resistance was shown to correlate with the transcriptional pattern of the XRCC1 gene, both before and after irradiation at distinct doses; at higher doses the system becomes overwhelmed. Our findings form the basis for further work on in vivo irradiation studies on animals, a biologic system which should be used as it is nonsophisticated and not confounded by the many uncertainties of in vitro tests. Our results could furthermore serve as an explanation for the different degrees of cardiac responses after ionizing irradiation(26).
Abbreviations
- XRCC1:
-
x-ray repair cross complementing
- RS:
-
radiation sensitive
- RR:
-
radiation resistant
- CHO:
-
Chinese hamster ovary
References
Jeggo PA, Tesmer J, Chen DJ 1991 Genetic analysis of ionising radiation sensitive mutants of cultured mummalian cell lines. Mutat Res 254: 125–132
Collins AR 1993 Mutant rodent cell lines sensitive to ultraviolet light, ionizing radiation and cross-linking agents: a comprehensive survey of genetic and biochemical characteristics. Mutat Res 293: 99–106
Barrows LR, Paxton MB, Kennedy KA, Thompson LH 1993 Characterization of the CHO EM9 mutant arising during DNA transfection. Carcinogenesis 12: 805–811
Thompson LH, Brookman KW, Dillehay LE, Carrano AV, Maszrimas JA, Mooney CL, Minkler JL 1982 A CHO-cell strain having hypersensitivity to mutagens, a defect in strand break repair, and an extraordinary baseline frequency of sister chromatid exchange. Mutat Res 95: 427–435
Thompson LH, Brookman KW, Jones NJ, Allen SA, Carrano AV 1990 Molecular cloning of the human XRCC1 gene, which corrects defective DNA strand repair and sister chromatid exchange. Mol Cell Biol 10: 6160–6171
Caldecott KW, Jeggo P 1991 Cross sensitivity of gamma ray sensitive hamster mutants to cross linking agents. Mutat Res 255: 111–119
Palitti F, Cortes F, Bassi L, Di Chiara D, Fiore M, Pinero J 1993 Higher G2 sensitivity to the induction of chromosomal damage in the CHO mutant EM9 than in its parental line AA8 by camptothecin, an inhibitor of DNA topoisomerase I. Mutat Res 285: 281–295
Churchill ME, Peak JG, Peak MJ 1991 Correlation between cell survival and DNA single strand break repair proficiency in the Chinese hamster ovary cell lines AA8 and EM9 irradiated with 365 nm ultraviolet A radiation. Photochem Photobiol 53: 229–238
Van Ankeren SC, Murray D, Stafford PM, Meyn RE 1988 Cell survival and recovery processes in Chinese hamster AA8 cells and in two radiosensitive clones. Radiat Res 116: 223–238
Hoy CA, Fuscoe JC, Thompson LH 1987 Recombination and ligation of transfected DNA in CHO mutant EM9, which has high levels of sister chromatid exchange. Mol Cell Biol 7: 2007–2017
Green A, Prager A, Stoudt PM, Murray D 1992 Relationships between DNA damage and the survival of radiosensitive mutant Chinese hamster cell lines exposed to gamma radiation. Int J Radiat Biol 61: 465–479
Dillehay LE, Thompson LH, Minkler JL, Carrano AV 1983 The relationship between sister chromatid exchange and perturbations in DNA replication in mutant EM9 and normal CHO cells. Mutat Res 109: 283–291
Carrano AV, Minkler JL, Dillehay LE, Thompson LH 1986 Incorporated bromodeoxyuridine enhances the sister chromatid exchange and chromosomal aberration frequencies in an EMS sensitive Chinese hamster cell line. Mutat Res 162: 223–241
Brookman KW, Tebbs RS, Allen SA, Tucker JD, Swiger RR, Lamerdin JE, Carrano AV, Thompson LH 1994 Isolation and characterization of mouse Xrcc-1, a DNA repair gene affecting ligation. Genomics 22: 180–189
Roderick TH 1963 The response of twenty seven inbred strains of mice to daily doses of whole body X-irradiation. Radiat Res 20: 631–639
McMaster GK, Carmichael GG 1977 Analysis by single and double stranded nucleic acids on polyacrylamide and agarose gels by using glyoxal and acridine orange. Proc Natl Acad Sci USA 74: 4835–4841
Southern EM 1975 Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol 98: 503–511
White BA, Bancroft FC 1982 Cytoplasmic dot hybridization. Simple analysis of mRNA levels in multiple small cell or tissue samples. J Biol Chem 257: 8569–8575
Shung B, Miyakoshi J, Takebe H 1994 X-ray induced transcriptional activation of c-myc and XRCC1 genes in ataxia teleangiectasia cells. Mutat Res 307: 43–51
Yoo H, Li L, Sacks PG, Thompson LH, Becker FF, Chan JY( ( 1992) Alterations in expression and structure of the DNA repair gene XRCC1. Biochem Biophys Res Commun 186: 900–910
Dunphy EJ, Beckett MA, Thompson LH, Weichselbaum RR( ( 1992) Expression of the polymorphic human DNA repair gene XRCC1 does not correlate with radiosensitivity in the cells of human head and neck tumor cell lines. Radiat Res 130: 166–170
Wei YF, Robins P, Carter K, Caldecott K, Pappin DJ, Yu GL, Wang RP, Shell BK, Nash RA, Schar P 1995 Molecular cloning and expression of human cDNAs encoding a novel DNA ligase IV and DNA ligase III, an enzyme activity in DNA repair and recombination. Mol Cell Biol 15: 3206–3216
Caldecott KW, Tucker JD, Stanker LH, Thompson LH 1995 Characterization of the XRCC!-DNA ligase III complex in vitro and its absence from mutant hamster cells. Nucleic Acids Res 23: 4836–4843
Lehmann AR 1993 Duplicated region of sequence similarity to the human XRCC1 DNA repair gene in theSchizosaccharomyces pombe rad4/cut 5 gene. Nucleic Acids Res 21: 5274–5283
Cuomo C Oettinger MA 1994 Analysis of regions of RAG-2 important for V(D)J recombination. Nucleic Acids Res 22: 1810–1814
Schultz-Hector S 1992 Radiation induced heart disease: review of experimental data on dose response and pathogenesis. Int J Radiat Biol 61: 149–16
Author information
Authors and Affiliations
Additional information
Supported by the Red Bull Company, Salzburg, Austria.
Rights and permissions
About this article
Cite this article
Labudova, O., Hardmeier, R., Rink, H. et al. The Transcription of the XRCC1 Gene in the Heart of Radiation-Resistant and Radiation-Sensitive Mice after Ionizing Irradiation. Pediatr Res 41, 435–439 (1997). https://doi.org/10.1203/00006450-199703000-00022
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199703000-00022