Abstract
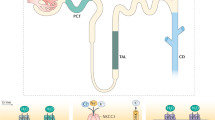
Chloride transport in the rabbit proximal convoluted tubule (PCT) has components of active, transcellular, and passive, paracellular transport. The preferential reabsorption of bicarbonate and organic solutes by the early proximal tubule leaves the luminal fluid with a higher chloride concentration than that in the peritubular capillaries. Previous studies have suggested that solute permeability of the paracellular pathway may be higher in the neonatal PCT and that the neonatal proximal tubule reabsorbs solutes by passive mechanisms to a greater extent than the adult segment. A higher chloride permeability would provide a mechanism for the greater rate of passive NaCl transport by the neonatal proximal tubule. The purpose of the presentin vitro microperfusion study was to directly examine the chloride permeability of neonatal and adult PCT. Superficial and juxtamedullary, neonatal and adult PCT were perfused with a high chloride perfusate without organic solutes, simulating late proximal tubular fluid, at 20°C, and bathed in a serum-like albumin solution. Chloride concentrations in the perfusate and the collected fluid were measured by electrometric titration. Neonatal juxtamedullary PCT chloride permeability (PCl) was significantly lower than adult juxtamedullary PCT PCl (0.15± 0.25 × 10-5 cm/s versus 5.23 ± 0.57× 10-5 cm/s, p < 0.001). The PCl of neonatal superficial PCT was not different from that of adult superficial PCT(0.81 ± 0.48 × 10-5 cm/s versus 0.05 ± 0.62 × 10-5 cm/s). Thus, there is a maturational increase in juxtamedullary PCT PCl, whereas superficial PCTPCl remains very low. The passive diffusion of chloride in neonatal PCT is extremely low and is not a mechanism to explain a higher rate of passive NaCl transport in this segment.
Similar content being viewed by others
Main
Chloride transport in the proximal tubule has three components(1, 2). The first component is active, transcellular transport that is mediated by specific apical and basolateral chloride transporters(3–6). The second component of chloride transport is solvent drag which is determined by the rate of volume absorption and chloride reflection coefficient. The third component is passive diffusion. The proximal tubule actively reabsorbs bicarbonate and organic solutes from the glomerular ultrafiltrate so that late proximal tubular fluid has a lower bicarbonate concentration, essentially no organic solutes and a higher chloride concentration than the peritubular fluid(7). The passive transport of chloride is then dependent on this chloride gradient and the transepithelial chloride permeability.
Proximal tubule solute permeability may be higher in neonates than adults. Experiments have assessed PCT mannitol permeability by microinjecting mannitol into the PCT of guinea pigs(8). The urinary recovery of mannitol increased from 92% in neonates to 100% in adults, indicating that PCT mannitol permeability is higher in the neonate. The permeability to chloride was not directly measured, but if the chloride permeability in neonatal PCT is higher than adults, the passive diffusion of chloride would be higher. The purpose of the present in vitro microperfusion study was to directly examine the chloride permeability of neonatal PCT to determine the contribution of passive diffusion to chloride transport.
METHODS
PCT from adult and neonatal (7-15 d old) New Zealand White rabbits were perfused in vitro as previously described(9, 10). Briefly, juxtamedullary and superficial PCT were dissected in a cooled (4°C) ultrafiltrate-like solution containing in mM: 115 NaCl, 25 NaHCO3, 4.0 Na2HPO4, 10 sodium acetate, 1.8 CaCl2, 1 MgSO4, 5 KCl, 8.3 glucose, and 5 alanine. Juxtamedullary PCT were obtained from the corticomedullary junction in both adults and neonates. Adult superficial PCT were obtained from nephrons that touched the surface or were just below the surface. Neonatal superficial PCT were obtained from nephrons that were below the surface. These appeared to be functioning nephrons because the tubule lumina were open. Tubules were then transferred to a 1.2-mL thermostatically controlled (20°C) bathing chamber and perfused with concentric glass pipettes. The bathing solution was similar to the dissection solution, but contained 7 g/dL of BSA (Sigma Chemical Co.), was bubbled with 95% O2 and 5% CO2 at 37°C, and had a pH of 7.4. The measured chloride concentration of the bathing solution was 100 mEq/L. The perfusion solution was a low bicarbonate-high chloride solution without organic solutes to mimic late proximal tubular fluid and contained in mM: 140 NaCl, 5 NaHCO3, 4 Na2HPO4, 5 KCl, 1 CaCl2, and 1 MgSO4. The osmolality of the perfusion and bathing solutions were adjusted to 295 by the addition of water or NaCl. The bathing solution was exchanged at a rate of 0.5 mL/min to keep the osmolality and pH constant. Tubules were perfused at 20°C to inhibit active transport(4, 12).
JV (in nL/min·mm) was measured as the difference between the perfusion and collection rates and normalized per mm of tubule length. Exhaustively dialyzed [methoxy-3H]inulin was added to the perfusate at a concentration of 50 μCi/mL so that the perfusion rate could be calculated. The collection rate was determined by timed collections using a constant volume pipette. The tubule length (L) was measured using an eyepiece micrometer. There were three determinations of volume absorption in each tubule.
Perfused and collected chloride concentrations were measured by the microtitration method of Ramsay(11). Chloride transport was then calculated by the following equation: whereVO and VL represent the perfusion and collection rates (in nL/min), respectively, and CO andCL represent the chloride concentration (in mEq/L) in the perfusate and collected fluid, respectively. There were three determinations of perfused and collected chloride concentrations for each tubule.
The transepithelial PD (in millivolts) was measured by using the perfusion pipette as the bridge into the tubular lumen. The recording and reference calomel half-cells were connected to the perfusion and bathing solutions via agarose bridges containing 3.6 M KCl/0.9 M KNO3. This arrangement avoided direct contact between the solution bathing the tubule and the KCl/KNO3 agarose bridge. The recording and reference calomel half-cells were then connected to the high and low impedance sides, respectively, of an electrometer (model 601; Keithley Instruments, Inc., Cleveland, OH). The liquid junction potential between the low bicarbonate-high chloride perfusion solution and the ultrafiltrate-like bathing solution was measured, and all recorded potential differences were corrected accordingly.
The transepithelial chloride permeability was calculated from the followingequation(2).
[horizontal bar over]C is the mean chloride concentration[horizontal bar over]C = (CB + [horizontal bar over]CL)/2, where CB is the bath chloride concentration and CL is the mean luminal chloride concentration [horizontal bar over]CL = (CO+ CL)/2. DFCl is the driving force for passive chloride transport and was calculated using the followingequation. where F is Faraday's constant,ΔV the transepithelial PD, R the gas constant, andT the absolute temperature. σCl is the chloride reflection coefficient so that (1 - σCl)[horizontal bar over]CJV represents the chloride flux due to solvent drag.Jact was zero because the studies were performed at 20°C(4, 12). The chloride permeability was calculated by solving for PCl and was normalized per tubule area using the tubule inner diameter. The tubule inner diameter was measured using an eyepiece micrometer.
Chloride permeability was also determined by adding 15 μCi/mL of36 Cl to the perfusate. PCl was then calculated by the following equation: where CO* andCL* are the counts/min of 36Cl in the perfusate and collected fluid, respectively, and A is the inner surface area determined from the inner diameter.
All data are represented as mean ± SEM. Statistical significance was determined by unpaired t test.
RESULTS
The first series of experiments examined the chloride permeabilities of neonatal and adult juxtamedullary PCT. The results are shown inTable 1 and Figure 1. The measured chloride concentration of the perfusate was 155.0 ± 2.9 mEq/L for the neonatal studies and 155.3 ± 2.9 mEq/L for the adult studies(p = NS). Passive chloride flux was much greater in the adult juxtamedullary PCT than in neonatal juxtamedullary PCT. ThePCl of the neonatal juxtamedullary PCT was more than 10-fold lower than the adult (0.15 ± 0.25 × 10-5 cm/sversus 5.23 ± 0.57 × 10-5 cm/s, p < 0.001). The chloride permeability of the neonatal juxtamedullary PCT was not significantly different from zero.
Chloride permeability of juxtamedullary PCT. PCT were perfused with a high chloride perfusate with no organic solutes to simulate late proximal tubular fluid and bathed with a serum-like albumin solution at 20°C. Perfusate and collected fluid chloride concentrations were measured by electrometric titration. Permeability was calculated from the measured chloride flux.
The second series of experiments examined the chloride permeabilities of neonatal and adult superficial PCT. These results are shown inTable 1 and Figure 2. The measured perfusate chloride concentration for these studies was 154.5 ± 0.9 mEq/L for the neonates and 152.1 ± 0.9 mEq/L for the adults(p = NS). There was no difference in PCl between neonatal and adult superficial PCT (0.81 ± 0.48 × 10-5 cm/s versus 0.05 ± 0.62 × 10-5 cm/s; p= NS). The chloride permeability of both neonatal and adult superficial PCT were not different from zero.
Chloride permeability of superficial PCT. PCT were perfused with a high chloride perfusate without organic solutes to mimic late proximal tubular fluid and bathed with a serum-like albumin solution at 20°C. Perfusate and collected fluid chloride concentrations were measured using electrometric titration. Permeability was calculated from the measured chloride flux.
The last series of experiments was designed to determine whether significant exchange diffusion of chloride occurred in the proximal tubule by measuring the permeability of chloride using 36Cl. The results of these experiments were qualitatively similar to the previous series of studies.PCl of neonatal juxtamedullary PCT was 1.35 ± 0.24× 10-5 cm/s which was significantly lower than the adult juxtamedullary PCT value of 7.10 ± 3.82 × 10-5 cm/s(p < 0.05). In the superficial PCT, there was no difference between the neonatal chloride permeability of 2.75 ± 0.71 × 10-5 cm/s and the adult value of 4.89 ± 2.35 × 10-5 cm/s. The permeability of chloride from the unidirectional flux studies was higher than that determined from the net flux studies indicating that there was significant exchange diffusion of chloride.
DISCUSSION
The present in vitro microperfusion study directly measured the chloride permeability of neonatal and adult PCT. The studies were performed at 20°C so that active transport was inhibited. The perfusate was similar to late proximal tubular fluid with a high chloride concentration so that there was a lumen to bath chloride gradient. Under these conditions, the only group of tubules with a significant chloride flux was the adult juxtamedullary PCT. Neonatal PCT, both juxtamedullary and superficial, had almost no chloride flux indicating that the PCl was close to zero. The adult superficial PCT also had very little chloride transport indicating that thePCl in this group was also near zero. Thus, there is a maturational increase in chloride permeability in juxtamedullary PCT, but not in superficial PCT.
Passive diffusion of chloride has been examined in adult rabbit PCT under similar conditions to the present study(12). The chloride permeability determined from the net flux was 4.50 ± 0.41× 10-5 cm/s for superficial PCT and 2.08 ± 0.11 × 10-5 cm/s for juxtamedullary PCT. The isotopically measuredPCl was slightly higher in each group indicating that there was some chloride exchange diffusion present. The reason for the differences in these findings compared with the present study are unclear, but may be related to the location of the tubule segment studied. The previous study specifically examined late PCT segments. The present study examined PCT that did not contain the early S1 segment, but may have been proximal to the area studied in the previous study. Another study of chloride permeability in adult PCT did not specify whether superficial or juxtamedullary PCT were used(13). The isotopically determined PCl was 3.2 ± 0.6 × 10-5 cm/s for sodium-selective tubules and 5.5 ± 0.6 × 10-5 cm/s for chloride-selective tubules. These values are comparable to the adult juxtamedullary PCTPCl determined in the present study.
Recently, the cellular transport mechanisms for chloride were examined in adult superficial and juxtamedullary PCT(6). One aspect of the study examined the transepithelial flux of chloride under conditions similar to the present experiment. It was found that JV in superficial PCT perfused at 37°C with a high chloride perfusate in the absence of formate was not different from zero. This suggested that thePCl for this segment was extremely low. TheJV in juxtamedullary PCT was greater than zero which suggested that the PCl in adult juxtamedullary PCT was higher than the superficial segment. The present study confirmed these results by showing that in the absence of active transport, there was no diffusion of chloride in superficial PCT, but there was significant diffusion of chloride in juxtamedullary PCT.
Several studies have indicated that the PCT paracellular pathway in the neonate is more permeable to solutes than in the adult PCT(8,14,15). This developmental difference was first suggested by Barnett et al.(14). In premature infants the clearance of mannitol was found to be significantly lower than the clearance of inulin. This indicated that some of the filtered mannitol must have been reabsorbed by the tubules. In adults, the clearance of mannitol is equal to that of inulin, suggesting that the paracellular permeability of mannitol is lower in the adult(15).
Kaskel et al.(8) examined the maturational changes in the paracellular pathway in guinea pigs by analyzing the urinary recovery of radiolabeled compounds that were microinjected into the proximal tubule. The recovery of inulin and sucrose was 100% during all stage of development. The recovery of creatinine was only 94%, but was also constant during maturation. The recovery of mannitol was 92% in the neonates and increased to 100% in the adults, suggesting that the PCT mannitol permeability was high in the neonate and decreased during development. Histologic measurements of the proximal tubule tight junctions were also made and showed that the length and width of the zonula occludens did not change, but the length of the paracellular pathway increased as the animals matured. Because of the maturational change of the mannitol permeability, it was concluded that passive transport of solute and fluid would be higher in the neonate.
The differences in our results and those of Kaskel et al.(8) are not clear but may be related to several factors. First, there are differences in the techniques used to assess permeability. The urinary recovery of microinjected mannitol will give a qualitative assessment of the permeability of the epithelium whereas in vitro microperfusion of PCT will yield a quantitative measure of the permeability. It is not clear what the change in the actual epithelial permeability to mannitol is in the pervious study. The microinjection technique also assesses the entire nephron so that it may not be clear which segment has a change in permeability. In vitro microperfusion allows for the study of specific segments of the nephron. Second, there may be differences in the species studied. Kaskel et al.(8) examined guinea pigs whereas the present study examined rabbits. Third, the solute studied may be important. Mannitol is an uncharged solute and chloride is negatively charged. It is quite possible that the pathway of diffusion for these solutes is different and could have quite different maturational changes.
Horster and Larsson(16) also examined the permeability of the paracellular pathway in neonatal rabbit PCT. They found that the hydraulic conductivity, measured by changes in volume absorption with changes in perfusion pressure, was higher in neonatal PCT. The integrity of the tight junctions was assessed by the ability of ferritin, microperoxidase and horse-radish peroxidase to penetrate from the lumen to the intercellular space. When the neonatal PCT was perfused under high pressure (20 cm water), microperoxidase was found to have penetrated the tight junction. Under all other conditions, the molecules remained in the lumen. This suggested that the neonatal PCT tight junction was not as intact as the adult tight junction and that passive transport of fluid could be higher in the neonatal PCT.
We have previously examined the maturational changes in bicarbonate permeability of the rabbit PCT to determine whether the low rate of bicarbonate transport in neonates could be due to a high backleak(10). Tubules were perfused with a bath to lumen bicarbonate gradient in the presence of acetazolamide to inhibit active transport of bicarbonate. Collected bicarbonate concentrations were measured by microcalorimetry(17). The bicarbonate permeability of the neonatal juxtamedullary PCT was found to be significantly lower than that of the adult segment. Thus, the lower rate of bicarbonate transport is due to lower active transport and not due to an increase on the backleak of bicarbonate.
The present study examined the chloride permeability of neonatal and adult juxtamedullary PCT and superficial PCT to determine the maturation of the passive diffusion component of chloride transport. Adult juxtamedullary PCT had the highest PCl with the adult superficial PCT and neonatal juxtamedullary PCT and superficial PCT having extremely smallPCl. Thus, there is a significant maturational increase ofPCl in juxtamedullary PCT, but not superficial PCT. The maturational increase in juxtamedullary PCT PCl may be due to an increase in the permeability of the pathway for diffusion or to an increase in the area for diffusion. The present study did not directly assess the area of diffusion. The component of passive chloride transport in rabbit PCT is extremely small except for adult juxtamedullary PCT. Thus, the contribution of passive chloride transport in the neonatal PCT is negligible.




Abbreviations
- PCT:
-
proximal convoluted tubule
- PCl:
-
chloride permeability
- PD:
-
potential difference
- JCl:
-
chloride transport rate
- Jv:
-
volume absorption rate
References
Kokko JP, Baum M 1992 Chloride transport. Handb Physiol 739–765
Alpern RJ, Howlin KJ, Preisig PA 1985 Active and passive components of chloride transport in the rat proximal convoluted tubule. J Clin Invest 76: 1360–1366
Baum M 1987 Evidence that parallel Na+-H+ and Cl--HCO-3(OH-) antiporters transport NaCl in the proximal tubule. Am J Physiol 252: F338–F345
Baum M, Berry CA 1985 Peritubular protein modulates neutral active NaCl absorption in the rabbit proximal convoluted tubule. Am J Physiol 248:F790–F795
Baum M, Berry CA 1986 Evidence for neutral transcellular NaCl transport and neutral basolateral chloride exit in the rabbit proximal convoluted tubule. J Clin Invest 74: 205–211
Sheu JN, Quigley R, Baum M 1995 Heterogeneity of chloride/base exchange in rabbit superficial and juxtamedullary proximal convoluted tubules. Am J Physiol ( in press)
Dubose Jr TD, Pucacco LR, Lucci MS, Carter NW 1979 Micropuncture determination of pH, PCO2 and total CO2 concentration in accessible structures of the rat renal cortex. J Clin Invest 64: 476–482
Kaskel FJ, Kumar AM, Lockhart EA, Evan A, Spitzer A 1987 Factors affecting proximal tubular reabsorption during development. Am J Physiol 252:F188–F197
Burg M, Grantham J, Abramow M, Orloff J 1966 Preparation and study of fragments of single rabbit nephrons. Am J Physiol 210: 1293–1298
Quigley R, Baum M 1990 Developmental changes in rabbit juxtamedullary proximal convoluted tubule bicarbonate permeability. Pediatr Res 28: 663–666
Ramsay JA, Brown RH, Croghan PC 1955 Electrometric titration of chloride in small volumes. J Exp Biol 32: 822–829
Holmberg C, Kokko JP, Jacobson HR 1981 Determination of chloride and bicarbonate permeabilities in proximal convoluted tubules. Am J Physiol 241:F386–F394
Warnock DG, Yee VJ 1982 Anion permeabilities of the isolated perfused rabbit tubule. Am J Physiol 242:F395–F405
Barnett HL, Hare WK, McNamara H, Hare RS 1948 Influence of postnatal age on kidney function in premature infants. Proc Soc Exp Biol Med 69: 55–57
Smith WW, Finkelstein N, Smith HW 1940 Renal excretion of hexitols (sorbitol, mannitol, and dulcitol) and their derivatives(sorbitan, isomannide, and sorbide) and of endogenous creatinine-like chromogen in dog and man. J Biol Chem 135: 231–250
Horster M, Larsson L 1976 Mechanisms of fluid absorption during proximal tubule development. Kidney Int 10: 348–363
Vurek GG, Warnock DG, Corsey R 1975 Measurement of picomole amounts of carbon dioxide by calorimetry. Anal Chem 47: 765–767
Acknowledgements
The authors are grateful for the technical assistance of Jay Dunn and the able secretarial assistance of Janell McQuinn.
Author information
Authors and Affiliations
Additional information
Supported by National Institute of Diabetes and Digestive and Kidney Diseases Grants K08-DK02232 (R.Q.) and DK41612 (M.B.).
Rights and permissions
About this article
Cite this article
Sheu, JN., Baum, M., Bajaj, G. et al. Maturation of Rabbit Proximal Convoluted Tubule Chloride Permeability. Pediatr Res 39, 308–312 (1996). https://doi.org/10.1203/00006450-199602000-00020
Issue Date:
DOI: https://doi.org/10.1203/00006450-199602000-00020
This article is cited by
-
Developmental changes in renal tubular transport—an overview
Pediatric Nephrology (2015)