Abstract
The development of new semiconducting polymers is one of the most important issues to address in improving the performance of organic electronic devices. Donor–acceptor semiconducting polymers, in which the backbone consists of alternating electron-rich (donor) and electron-deficient (acceptor) heterocyclic π-building units, are emerging materials that promote intermolecular interactions and offer desirable electronic structures, in turn leading to high charge carrier transport. This review focuses on the development of donor–acceptor semiconducting polymers using electron-deficient π-building units, such as thiazolo[5,4-d]thiazole, benzo[1,2-d:4,5-d′]bisthiazole, naphtho[1,2-c:5,6-c′]bis[1,2,5]thiadiazole and thieno[3,2-b]thiophene-2,5-dione. All of the polymers form crystalline structures in thin films and possess deep highest occupied molecular orbital energy levels; consequently, the polymers demonstrate high charge carrier mobilities with high air stability. Based on these studies, several key parameters were identified that must be taken into account when designing high-performance polymers: the symmetry of the building units, backbone shape and delocalization of the π-electrons along the backbone. Notably, many of the polymers synthesized in the studies exhibit high photovoltaic performance, indicating that such crystalline polymers are also useful for solar cell applications. Thus, this review affords an important guideline for the development of high-performance semiconducting polymers.
Similar content being viewed by others
Introduction
π-Conjugated polymers, often called semiconducting polymers, are an important class of materials that allow for solution-based fabrication and thus enable the development of low-cost, light-weight, flexible, and large-area electronic devices, such as organic field-effect transistors (OFETs) and organic photovoltaics.1, 2, 3, 4 Over the past decades, a number of semiconducting polymers have been designed and synthesized, and their performance in these devices has been markedly improved.5, 6, 7 In both types of devices, an important property required for semiconducting polymers is charge carrier mobility. As revealed by numerous studies of regioregular poly(3-hexylthiophene) (rrP3HT),8,9 improvement of the charge carrier transport through π–π overlaps between the face-to-face π-stacked polymer chains is crucial for overall charge transport in polymer films;10,11 hence, a coplanar backbone with strong intermolecular interaction that can lead to highly ordered crystalline structures is necessary.
A promising molecular design strategy for producing a coplanar backbone in semiconducting polymers is the introduction of π-extended fused heteroaromatic rings.12 The introduction of a ‘donor–acceptor’ (D–A) motif into the backbone, in which electron-rich (donor) and electron-poor (acceptor) units are copolymerized, is also a useful design strategy because the D–A motif can facilitate intermolecular interaction because of dipole–dipole interactions.13 Indeed, most of the high-performance polymers that have been reported recently are synthesized in accordance with these strategies. In D–A polymers, it is apparent that the acceptor unit often plays a key role in determining the electronic structure of the final material. The polymers are desired to have a narrow bandgap, which qualitatively represents a π-electron system, and a deep highest occupied molecular orbital (HOMO) energy level, which ensures oxidative stability. Therefore, a relatively deep lowest unoccupied molecular orbital (LUMO) energy level is required, and thus the acceptor unit, which significantly affects the LUMO level, is a key building unit. It is also noted that because deep LUMO levels near −4 eV or below are required for electron transport in air,14, 15, 16 the exploration of strong acceptors is important for the development of n-channel semiconducting polymers.
This Focus Review will summarize our recent work on the development of semiconducting polymers with a D–A backbone system for OFETs, which was conducted through the exploration of acceptor units including thiazolo[5,4-d]thiazole (TzTz), benzo[1,2-d:4,5-d′]bisthiazole (BBTz), naphtho[1,2-c:5,6-c′]bis[1,2,5]thiadiazole (NTz) and thieno[3,2-b]thiophene-2,5-dione (TTD). The electronic structure, ordering structure in thin films, OFET performance and the correlation of these properties with the polymers’ structure will be discussed.
Thiazolo[5,4-d]thiazole-based polymers
TzTz was first introduced into organic semiconductors by Yamashita and co-workers. The researchers synthesized a series of small molecules for both p-channel and n-channel OFETs, which showed high mobilities.17, 18, 19 Figure 1 displays the chemical structures of TzTz-based polymers reported as semiconducting polymers for OFETs.20, 21, 22 We initially introduced TzTz into a polythiophene backbone in a regiosymmetric fashion, in which the polymer was composed of two n-dodecylthiophenes and one TzTz (PTzBT-12). However, PTzBT-12 was insoluble in any solvent, most likely due to its rigidity and strong intermolecular interactions. We then designed PTzQTs with four alkylthiophenes to impart sufficient solubility.20 Indeed, the polymers were soluble in chloroform, toluene, and chlorinated benzenes. PTzQTs are observed to have absorption maxima (λmax) at ~580 and 630 nm both in solution and in films, which are red-shifted from those of rrP3HT (550 nm) owing to the D–A backbone. The bandgap (Eg) determined from the absorption onset was ~1.80 eV. The HOMO and LUMO energy levels of the polymers determined by cyclic voltammetry were −5.1 and −3.3 eV, respectively.23 The HOMO energy level was 0.2 eV lower than that of rrP3HT.
The field-effect mobility measured for annealed thin films of PTzQTs was as high as 0.3 cm2 V−1 s−1 (Figure 2a), greater than that of rrP3HT fabricated under the same conditions (~0.1 cm2 V−1 s−1). The current on to off ratio (Ion/Ioff) on the order of up to 107 was much higher than that of rrP3HT (~104). The relatively high mobility in this polymer system was attributed to the high crystalline nature of the thin film. A grazing incidence X-ray diffraction (GIXD) study revealed that the polymers form a well-ordered lamellar structure with an edge-on orientation and have a short π–π stacking distance of 3.5 Å (Figure 2b). This distance is 0.2–0.3 Å shorter than that of polythiophene-based semiconducting polymers, including rrP3HT, or thienothiophene-based polymers.12 The high crystallinity of PTzQTs is likely achieved not with the side-chain interdigitation that enhances crystallinity but with amorphous-like disordered side chains,21 which implies that the intermolecular interaction through the thiophene–TzTz backbones is significantly strong.
The environmental stability of the PTzQT devices was significantly higher than that of the rrP3HT devices because of the deeper HOMO level of the former. However, the mobility of the PTzQT-14-based devices still decreased by ~20% after being stored in air for 50 days (Figure 2a). To improve the stability of the TzTz polymer system, several thiophene–TzTz polymers were designed and synthesized (PTz3T, PTz4T and PTzBT-14HD).22 With lower alkyl side chain densities than PTzQTs, these polymers exhibited deeper HOMO levels of approximately −5.2 eV. Consequently, the OFET performance of these polymers remained nearly unchanged even after 50 days storage in air (Figure 2c for PTzBT-14HD), although the mobility was slightly reduced (~0.1 cm2 V–1 s–1) compared with that of PTzQT. The lower mobility in these polymers is likely attributed to the misorientation of the polymer crystallite, as indicated by the diffraction arcs illustrated by two-dimensional GIXD (Figure 2d). However, by increasing its molecular weight, PTzBT-14HD formed a complete edge-on orientation and exhibited a mobility as high as 0.42 cm2 V–1 s–1.24 In addition, by further investigating the PTzBT system, the polymers were observed to show high power conversion efficiencies of up to 7.5% when used in bulk-heterojunction solar cells.24,25
Benzo[1,2-d:4,5-d′]bisthiazole-based polymers
BBTz has been known as a good building unit for electroactive materials and thermally stable polymer fibers.26, 27, 28 In BBTz, benzene is fused between two thiazoles formally π-extending the thiazolothiazole system and thus can forge even more efficient π–π interactions than TzTz.29,30 We thus introduced BBTz into the polythiophene backbone in a fashion similar to that adopted for PTz3T and PTz4T (PBBTz3T and PBBTz4T, Figure 3).31 These polymers showed improved mobilities as high as 0.3 cm2 V−1 s−1, even with the misorientation of the edge-on crystallite. More interestingly, the OFET devices fabricated using these polymers demonstrated significantly high stability in high-humidity air; the change in the transfer curve was very small when the devices were stored under a relative humidity of ~75% (Figure 3).
The chemical structure of BBTz-based polymers and the change in the transfer characteristics of the OFET devices under a relative humidity of ~75%. (a) PBBTz3T and (b) PBBTz4T. Reproduced with permission from ref. 35. Copyright 2010 Wiley. A full color version of this figure is available at Polymer Journal online.
Naphtho[1,2-c:5,6-c′]bis[1,2,5]thiadiazole-based polymers
NTz (Figure 4a upper) is a doubly benzothiadiazole (BTz)-fused heteroaromatic ring32 and has a strong electron-accepting nature as well as high rigidity and planarity. Thus, the incorporation of NTz into the polythiophene backbone could lead to a deep HOMO energy level and a small bandgap. It is also expected that intermolecular interactions will be enhanced and that a strong π–π stacking structure will thereby be promoted, in turn leading to high mobilities. Cao and co-workers reported on a NTz-based polymer with a benzodithiophene unit and demonstrated that the polymer showed high photovoltaic properties with power conversion efficiencies as high as 6% in a bulk-heterojunction solar cell in combination with [6,6]-phenyl-C71-butyric acid methyl ester (PC71BM).33 We independently synthesized an NTz-based polymer (PNTz4T) and a BTz-based polymer (PBTz4T) with quaterthiophene, featuring a 2-decyltetradecyl long branched alkyl side chain, as the D unit (Figure 4a).34
The λmax for PNTz4T appeared at 725 nm in a thin film, which was significantly red-shifted compared with that for PBTz4T (662 nm). The value of Eg was estimated to be 1.54 eV for PNTz4T and 1.65 eV for PBTz4T. Whereas the HOMO and LUMO energy levels for PNTz4T determined by cyclic voltammetry to be −5.15 and −3.60 eV, those for PBTz4T were −5.07 and −3.42 eV, respectively, indicating that NTz is indeed a strong acceptor unit.
The field-effect mobilities of PNTz4T were as high as 0.56 cm2 V−1 s−1, with the Ion/Ioff ratios on the order of 107 (Figure 4b). The mobility of PNTz4T was one order of magnitude higher than that of PBTz4T, typically 0.05 cm2 V–1 s–1, indicating the high potential of the NTz unit. In the two-dimensional GIXD pattern of the PNTz4T thin film, diffractions attributable to lamellar (qz≈0.25 Å−1) and the π–π stacking structures (qxy≈1.7 Å−1) appeared on the qz and qxy axes, respectively; these findings indicated a predominant edge-on orientation on the substrate surface, although there were some misoriented fractions, most likely as a result of the introduction of long branched alkyl groups in the side chain. The π–π stacking distance was 3.5 Å. In contrast, PBTz4T showed large arcing of the lamellar diffraction, indicating that there is no preferential orientation. PBTz4T did not show clear π–π stacking diffraction, indicative of the much lower crystalline nature of PBTz4T in the thin film. The difference in ordering structure between PNTz4T and PBTz4T is in good agreement with the fact that the mobility was much higher for PNTz4T than for PBTz4T. This high crystalline nature of PNTz4T is also beneficial for the use of the polymer in bulk-heterojunction solar cells. A solar cell using PNTz4T with [6,6]-phenyl-C61-butyric acid methyl ester (PC61BM) exhibited a power conversion efficiency of 6.3%.34
To gain deeper insight into the large difference in ordering structure between PNTz4T and PBTz4T, we conducted a single-crystal X-ray analysis of the model compound of these polymers, namely NTz2T and BTz2T. Whereas NTz2T affords an anti arrangement of the thiophene rings that sandwich NTz because NTz has C2h symmetry, BTz2T affords a syn arrangement of the neighboring thiophenes because BTz has C2v symmetry (Figure 5a). Taking these arrangements into account, PNTz4T provides a more linear-shaped backbone compared with PBTz4T, which shows a ‘wavy’ shape (Figure 5b). In addition, whereas the alkyl side chains are placed in an anti arrangement for PNTz4T, they are alternately placed in an anti and a syn arrangement for PBTz4T. We believe that this relatively linear backbone shape, together with the rigidity of NTz, and the more regularly arranged alkyl side chains largely contribute to the highly ordered packing structure of PNTz4T.
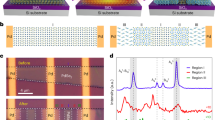
(a) Two-dimensional GIXD pattern of the PNTz4T film. (b) Two-dimensional GIXD pattern of the PBTz4T film. (c) The molecular structure of the model compounds, NTz2T (left) and BTz2T (right), elucidated by single-crystal X-ray analysis. (d) The optimized backbone structure of PNTz4T (upper) and PBTz4T (below). Reproduced with permission from ref. 34. Copyright 2012 American Chemical Society.
We have also reported on other NTz-based polymers (Figure 6).35, 36, 37 An isomeric polymer of PNTz4T, that is, PNTz4Ti, that has a different alkyl side-chain placement showed relatively red-shifted absorption spectra relative to the spectrum of PNTz4T.35 This findings indicates that this isomer has a somewhat more ordered structure, as supported by the results of GIXD. Another sample polymer system consists of naphthodithiophene as the co-unit (PNNTs, Figure 6).36,37 Although these polymers also showed high field-effect mobility, they operated rather favorably in solar cells. In particular, PNNT-12HD with additional alkyl side chains on the naphthodithiophene unit showed very high efficiencies exceeding 8% in combination with PC71BM.37
Thieno[3,2-b]thiophene-2,5-dione-based polymers
The quinoid structure has been introduced into π-conjugated polymer backbones to develop small-bandgap polymers and thus inherently conductive polymers.38 Quinoid compounds with electron-withdrawing substituents such as a cyano group have also been used as electron acceptors.39, 40, 41 In particular, quinoid compounds based on thiophene-fused rings or oligothiophenes, namely thienoquinoids, have been used as n-type organic semiconductors, which have demonstrated high electron mobilities.42, 43, 44 TTD is also a class of electron-withdrawing thienoquinoid with ketone terminal groups.45 With the less bulky ketone group, compared with the dicyanomethylene group, TTD offers a coplanar structure when incorporated into conjugated molecules.46 Recently, we introduced TTD into a π-conjugated backbone, forming a structure referred to as PTTD4T.47 By linking with neighboring thiophenes at the β-position of the TTD unit, the backbone allows for a fully π-conjugated system in which TTD can be viewed as a trans-butadiene moiety that is bridged to form thiolactones.
The HOMO and LUMO energy levels of PTTD4Ts determined by the cyclic voltammetry were −5.1 and −3.8 eV, respectively. Interestingly, the λmax of PTTD4T was located at a very long wavelength of 835 nm, and the absorption edge was 1030 nm, which corresponds to the very small Eg of 1.20 eV. We also observed that the electronic structure of PTTD4Ts is fairly unique. Whereas the shift of λmax from TTD2T (the monomer unit), in which TTD is sandwiched by alkythiophenes, to PTTD4T was 342 nm, in the case of PNTz4T, the shift of λmax from the monomer compound (NTz2T) to the corresponding polymer (PNTz4T) was 216 nm, even though the λmax values of NTz2T and TTD2T were largely the same (Figure 7b). This marked red-shift in the absorption spectra from the monomer unit to the polymer indicates that the polymer backbone possesses a quite effectively developed π-conjugation. The significantly elongated effective π-conjugation length is related to the elevation of the HOMO energy level and HOMO delocalization.
(a) Chemical structure of PTTD4T. (b) UV–vis absorption spectra of the monomer unit (NTz2T and TTD2T) and the corresponding polymer (PNTz4T and PTTD4T) for NTz and TTD-based polymers. (c) Transfer characteristics of PTTD4T-DT in the bottom-gate top-contact device. (d) Transfer characteristics of PTTD4Ts in the top-gate bottom-contact device. Reproduced with permission from ref. 47. Copyright 2014 Royal Society of Chemistry.
OFET devices with bottom-gate top-contact and top-gate bottom-contact configurations were tested. In both cases, PTTD4T-DT, with the longer branched alkyl side chain, exhibited higher mobilities than PTTD4T-OD did. In the bottom-gate top-contact device, in which the SiO2 substrate was treated with 1H,1H,2H,2H-perfluorodecyltriethoxysilane, the PTTD4Ts showed only p-channel behavior (Figure 7c), although they possessed a LUMO energy level of approximately 4 eV, which is close to the threshold for ambient n-channel operation. Although the hole mobilities were moderately high, ~0.2–0.3 cm2 V−1 s−1, when the films were annealed at 150 °C, the values significantly improved to 1.38 cm2 V−1 s−1, with an average of 0.66 cm2 V−1 s−1, when the films were annealed at 300 °C. The on/off ratios typically ranged from 105–107. Interestingly, on the other hand, in the top-gate bottom-contact devices PTTD4Ts showed ambipolar characteristics (Figure 7d), possibly as a result of the passivation by the gate insulator on top of the polymer layer, preventing electron trapping by atmospheric moisture. The hole and electron mobilities were as high as 0.12 and 0.20 cm2 V−1 s−1, respectively.
Based on the results of GIXD, the PTTD4Ts in the thin films were observed to form a highly ordered lamellar structure with an edge-on orientation and to have a π–π stacking distance of ~3.6 Å. This finding partially rationalizes the high mobility. Furthermore, we assume that the highly developed backbone π-conjugation, in which the HOMO is highly delocalized, as described above would facilitate both intramolecular and intermolecular electronic coupling and would thus play an important role in charge transport.
Summary
In this Focus Review, the development of D–A semiconducting polymers using particular acceptor units that exhibit high carrier mobility and stability is discussed. By incorporating increasingly large and strong electron-deficient π-building units, high carrier mobilities were achieved. It is also noted that ambipolar behavior was observed when using new electron-deficient units. In addition to OFET applications, many of the polymers described in this review can be used in solar cells, some of which demonstrate high power conversion efficiencies greater than 8%. Based on the studies correlating the electronic and thin-film structures of these polymers to device performance, several key factors in designing high-performance semiconducting polymers were identified: symmetry of the building unit, backbone shape and delocalization of the π-electrons along the backbone. Although this review specifically discussed the effect of building units, it is also important to design the side chains appropriately, which also play important roles in determining electronic structure, crystallinity, and backbone orientation, when in turn critically affect charge transport properties.48,49 By incorporating all of these aspects into molecular design, further high-performance semiconducting polymers should certainly be developed in the near future.
References
Noriega, R ., Salleo, A. & Klauk, H . Organic Electronics II: More Materials and Applications (Wiley-VCH, Weinheim, Germany, 2011).
Bao, Z. & Locklin, J. Organic Field-Effect Transistors (CRC Press, Boca Raton, FL, USA, 2007).
Brabec, C ., Dyakonov, V. & Scherf, U. Organic Photovoltaics: Materials, Device Physics, and Manufacturing Technologies (Wiley-VCH, Weinheim, Germany, 2008).
Hadziioannou, G. & Malliaras, G. G. Semiconducting Polymers, Chemistry, Physics and Engineering (Wiley-VCH, Weinheim, Germany, 2007).
Leclerc, M. & Morin, J.-F. Design and Synthesis of Conjugated Polymers (Wiley VCH, Weinheim, Germany, 2010).
Facchetti, A. π-Conjugated polymers for organic electronics and photovoltaic cell applications. Chem. Mater. 23, 733–758 (2011).
Beaujuge, P. M. & Fréchet, J. M. J. Molecular design and ordering effects in π-functional materials for transistor and solar cell applications. J. Am. Chem. Soc. 133, 20009–20029 (2011).
McCullough, R. The Chemistry of Conducting Polythiophenes. Adv. Mater. 10, 93–116 (1998).
Osaka, I. & McCullough, R. D. Advances in molecular design and synthesis of regioregular polythiophenes. Acc. Chem. Res. 41, 1202–1214 (2008).
Bao, Z ., Dodabalapur, A. & Lovinger, A. Soluble and processable regioregular poly (3-hexylthiophene) for thin film field-effect transistor applications with high mobility. Appl. Phys. Lett. 69, 4108–4110 (1996).
Sirringhaus, H ., Brown, P ., Friend, R. & Nielsen, M. Two-dimensional charge transport in self-organized, high-mobility conjugated polymers. Nature 401, 685–688 (1999).
McCulloch, I ., Heeney, M ., Chabinyc, M. L ., Delongchamp, D ., Kline, R. J ., Cölle, M ., Duffy, W ., Fischer, D ., Gundlach, D ., Hamadani, B ., Hamilton, R ., Richter, L ., Salleo, A ., Shkunov, M ., Sparrowe, D ., Tierney, S. & Zhang, W. Semiconducting thienothiophene copolymers: design, synthesis, Morphology, and Performance in Thin-Film Organic Transistors. Adv. Mater. 21, 1091–1109 (2009).
Sirringhaus, H ., Bird, M ., Richards, T. & Zhao, N. Charge transport physics of conjugated polymer field-effect transistors. Adv. Mater. 22, 3893–3898 (2010).
Jones, B. A ., Facchetti, A ., Wasielewski, M. R. & Marks, T. J. Tuning Orbital Energetics in Arylene Diimide Semiconductors. Materials Design for Ambient Stability of n-Type Charge Transport. J. Am. Chem. Soc. 129, 15259–15278 (2007).
De Leeuw, D. M ., Simenon, M. M. J ., Brown, A. R. & Einerhand, R. E. F. Stability of n-type doped conducting polymers and consequences for polymeric microelectronic devices. Synth. Met. 87, 53–59 (1997).
Anthopoulos, T. D ., Anyfantis, G. C ., Papavassiliou, G. C. & de Leeuw, D. M. Air-stable ambipolar organic transistors. Appl. Phys. Lett. 90, 122105 (2007).
Ando, S ., Nishida, J.-I ., Inoue, Y ., Tokito, S. & Yamashita, Y. Synthesis, physical properties, and field-effect transistors of novel thiophene/thiazolothiazole co-oligomers. J. Mater. Chem. 14, 1787–1790 (2004).
Ando, S ., Nishida, J ., Fujiwara, E ., Tada, H ., Inoue, Y. & Zhu, S. Characterization and field-effect transistor performance of heterocyclic oligomer containing a thiazolothiazole unit. Chem. Lett. 33, 1170–1171 (2004).
Ando, S ., Nishida, J ., Tada, H ., Inoue, Y ., Tokito, S. & Iso, Y. High performance n-type organic field-effect transistors based on pi-electronic systems with trifluoromethylphenyl groups. J. Am. Chem. Soc. 127, 5336–5337 (2005).
Osaka, I ., Sauvé, G ., Zhang, R ., Kowalewski, T. & McCullough, R. D. Novel thiophene-thiazolothiazole copolymers for organic field-effect transistors. Adv. Mater. 19, 4160–4165 (2007).
Osaka, I ., Zhang, R ., Sauvé, G ., Smilgies, D. M ., Kowalewski, T. & McCullough, R. D. High-lamellarordering and amorphous-like π-network inn short-chaint hiazolothiazole−thiophene copolymers lead to high mobilities. J. Am. Chem. Soc. 131, 2521–2529 (2009).
Osaka, I ., Zhang, R ., Liu, J ., Smilgies, D.-M ., Kowalewski, T. & McCullough, R. D. Highly stable semiconducting polymers based on thiazolothiazole. Chem. Mater. 22, 4191–4196 (2010).
Takimiya, K ., Osaka, I. & Nakano, M. π-Building blocks for organic electronics: revaluation of “inductive” and “resonance” effects of π-electron deficient units. Chemi. Mater 26, 587–593 (2013).
Osaka, I ., Saito, M ., Mori, H ., Koganezawa, T. & Takimiya, K. Drastic change of molecular orientation in a thiazolothiazole copolymer by molecular-weight control and blending with PC61BM leads to high efficiencies in solar cells. Adv. Mater. 24, 425–430 (2012).
Osaka, I ., Saito, M ., Koganezawa, T. & Takimiya, K. Thiophene–thiazolothiazole copolymers: significant impact of side chain composition on backbone orientation and solar cell performances. Adv. Mater. 26, 331–338 (2014).
Wolfe, J. F ., Loo, B. H. & Arnold, F. E. Rigid-rod polymers. 2. Synthesis and thermal properties of para-aromatic polymers with 2,6-benzobisthiazole units in the main chain. Macromolecules 14, 915–920 (1981).
Osaheni, J. A. & Jenekhe, S. A. Synthesis and processing of heterocyclic polymers as electronic, optoelectronic, and nonlinear optical materials. 1. New conjugated rigid-rod benzobisthiazole polymers. Chem. Mater. 4, 1282–1290 (1992).
Osaheni, J. A. & Jenekhe, S. A. Electroactive and photoactive rod-coil copolymers: design, synthesis, and supramolecular regulation of photophysical properties. J. Am. Chem. Soc. 117, 7389–7398 (1995).
Mike, J. F ., Inteman, J. J ., Ellern, A. & Jeffries-El, M. Facile synthesis of 2,6-disubstituted benzobisthiazoles: functional monomers for the design of organic semiconductors. J. Org. Chem. 75, 495–497 (2009).
Ahmed, E ., Kim, F. S ., Xin, H. & Jenekhe, S. A. Benzobisthiazole−thiophene copolymer semiconductors: synthesis, enhanced stability, field-effect transistors, and efficient solar cells. Macromolecules 42, 8615–8618 (2009).
Osaka, I ., Takimiya, K. & McCullough, R. D. Benzobisthiazole-Based Semiconducting Copolymers Showing Excellent Environmental Stability in High-Humidity Air. Adv. Mater. 22, 4993–4997 (2010).
Mataka, S ., Takahashi, K ., Ikezaki, Y ., Hatta, T ., Tori-i, A. & Tashiro, M. Sulfur nitride in organic chemsitry. XIV, selective formation of benzo-and benzobis [1, 2, 5] thiadiazole skeleton in the reaction of tetrasulfur tetranitride with naphthalenols and related compounds. Bull. Chem. Soc. Jpn 64, 68–73 (1991).
Wang, M ., Hu, X ., Liu, P ., Li, W ., Gong, X ., Huang, F. & Cao, Y. A donor-acceptor conjugated polymer based on naphtho [1, 2-c: 5, 6-c] bis [1, 2, 5] thiadiazole for high performance polymer solar cells. J. Am. Chem. Soc. 133, 9638–9641 (2011).
Osaka, I ., Shimawaki, M ., Mori, H ., Doi, I ., Miyazaki, E ., Koganezawa, T. & Takimiya, K. Synthesis, characterization, and transistor and solar cell applications of a naphthobisthiadiazole-based semiconducting polymer. J. Am. Chem. Soc. 134, 3498–3507 (2012).
Kawashima, K ., Miyazaki, E ., Shimawaki, M ., Inoue, Y ., Mori, H ., Takemura, N ., Osaka, I. & Takimiya, K. 5,10-Diborylated naphtho[1,2-c:5,6-c']bis[1,2,5]thiadiazole: a ready-to-use precursor for the synthesis of high-performance semiconducting polymers. Polym. Chem 4, 5224–5227 (2013).
Osaka, I ., Abe, T ., Shimawaki, M ., Koganezawa, T. & Takimiya, K. Naphthodithiophene-based donor–acceptor polymers: versatile semiconductors for OFETs and OPVs. ACS Macro Lett. 1, 437–440 (2012).
Osaka, I ., Kakara, T ., Takemura, N ., Koganezawa, T. & Takimiya, K. Naphthodithiophene–naphthobisthiadiazole copolymers for solar cells: alkylation drives the polymer backbone flat and promotes efficiency. J. Am. Chem. Soc. 135, 8834–8837 (2013).
Skotheim, T. S. Handbook of Conducting Polymers Second Edition CRC Press, (1997).
Ferraris, J ., Cowan, D. O ., Walatka, V. & Perlstein, J. H. Electron transfer in a new highly conducting donor–acceptor complex. J. Am. Chem. Soc. 95, 948–949 (1973).
Martin, N ., Segura, J. L. & Seoane, C. Design and synthesis of TCNQ and DCNQI type electron acceptor molecules as precursors for 'organic metals’. J. Mater. Chem. 7, 1661–1676 (1997).
Yui, K ., Aso, Y ., Otsubo, T. & Ogura, F. New electron acceptors for organic metals: extensively conjugated homologues of thiophene-7,7,8,8-tetracyanoquinodimethane (TCNQ). J. Chem. Soc. Chem.Commun. 1816–1817 (1987).
Kunugi, Y ., Takimiya, K ., Toyoshima, Y ., Yamashita, K ., Aso, Y. & Otsubo, T. Vapour deposited films of quinoidal biselenophene and bithiophene derivatives as active layers of n-channel organic field-effect transistors. J. Mater. Chem. 14, 1367–1369 (2004).
Pappenfus, T. M ., Chesterfield, R. J ., Frisbie, C. D ., Mann, K. R ., Casado, J ., Raff, J. D. & Miller, L. L. A π-stacking terthiophene-based quinodimethane Is an n-channel conductor in a thin film transistor. J. Am. Chem. Soc. 124, 4184–4185 (2002).
Chesterfield, R. J ., Newman, C. R ., Pappenfus, T. M ., Ewbank, P. C ., Haukaas, M. H ., Mann, K. R ., Miller, L. L. & Frisbie, C. D. High electron mobility and ambipolar transport in organic thin-film transistors based on a &pgr;-stacking quinoidal terthiophene. Adv. Mater. 15, 1278–1282 (2003).
Testaferri, L ., Tiecco, M ., Zanirato, P. & Martelli, G. Preparation and tautomeric structures of some potential 2,5-dihydroxythieno[3,2-b]thiophenes. J. Org. Chem. 43, 2197–2200 (1978).
Chen, S ., Bolag, A ., Nishida, J.-I. & Yamashita, Y. n-Type feld-effect transistors based on Thieno[3,2-b]thiophene-2,5-dione and the Bis(dicyanomethylene) derivatives. Chem. Lett. 40, 998–1000 (2011).
Osaka, I ., Abe, T ., Mori, H ., Saito, M ., Takemura, N ., Koganezawa, T. & Takimiya, K. Small band gap polymers incorporating a strong acceptor, thieno[3,2-b]thiophene-2,5-dione, with p-channel and ambipolar charge transport characteristics. J. Mater. Chem. C 2, 2307–2312 (2014).
Lei, T ., Wang, J.-Y. & Pei, J. Roles of flexible chains in organic semiconducting Materials. Chem. Mater. 26, 594–603 (2013).
Mei, J. & Bao, Z. Side chain engineering in solution-processable conjugated polymers. Chem. Mater. 26, 604–615 (2013).
Acknowledgements
The author thanks Prof Richard D. McCullough (Harvard University) and Prof Kazuo Takimiya (RIKEN) for all of their support as research supervisors. The author also thanks all of his co-workers and collaborators for their great contributions to the studies covered in this review. The author is grateful for the financial support from Grant-in-Aid for Scientific Research (Nos. 2035008800 and 24685030; Ministry of Education, Culture, Sports, Science and Technology), Precursory Research for Embryonic Science and Technology (Japan Science and Technology Agency) and the Sumitomo Foundation. Portions of the work were also supported by Strategic Promotion of Innovative Research and Development (Japan Science and Technology Agency) and the Sumitomo Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Osaka, I. Semiconducting polymers based on electron-deficient π-building units. Polym J 47, 18–25 (2015). https://doi.org/10.1038/pj.2014.90
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pj.2014.90
This article is cited by
-
Development of semiconducting polymers based on a novel heteropolycyclic aromatic framework
Polymer Journal (2021)
-
Evaluation-oriented exploration of photo energy conversion systems: from fundamental optoelectronics and material screening to the combination with data science
Polymer Journal (2020)
-
Low-bandgap semiconducting polymers based on sulfur-containing phenacene-type molecules for transistor and solar cell applications
Polymer Journal (2018)
-
Effects of branching position of alkyl side chains on ordering structure and charge transport property in thienothiophenedione- and quinacridone-based semiconducting polymers
Polymer Journal (2017)