Abstract
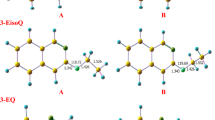
Conformational analyses have been carried out for ethylene oxide (EO) and ethylene imine (EI) oligomer models (single chain) by quantum chemical calculations (RHF/6-31+G(d,p)). The conformations repeated for a unit of X-C, C-C and C-X bonds (X: O or N) were examined. For optimizations of the EI oligomers, some designation systems with trans values were used, taking account of pseudoasymmetry. For the EO oligomers (EO-x, x = 2- to 8-mers), (ttt)x and (tg+t)x conformers were first and second stable (ΔE = 1.17–1.26 kcal/m.u.), respectively. On the contrary, for the EI oligomers (EI-x, x = 2- to 11-mers), (tg+t)x and (ttt)x were first and second stable (ΔE = 1.32–1.42 kcal/m.u.), respectively. Therefore, the gauche preference of C-C bond in EI-x is stronger than that in EO-x. The calculated structures of (ttt)x and (tg+t)x in EO-x agreed with those observed by XRD analysis for PEO crystals in stretched (planar zigzag) and original (7/2 helix) states, respectively. The calculated structures of (tg+t)x and (ttt)x in EI-x also agreed with those observed for PEI crystals in dehydrate (7/2 helix) and hydrate (planar zigzag) states, respectively. However, the dihedral angles (EO-8) and the 5 mol length (EI-11) in both helices did not agree with each of the observed values for the polymers. Each difference between the oligomers (single chain) and polymers was estimated by the effect of intermolecular interactions in PEO or PEI. For (tg+t)x of EI-11, the existence of metastable skewed helical structures (reversed and/or kinked) were estimated in addition to the helix.
Similar content being viewed by others
Article PDF
References
Y. Takahashi and H. Tadokoro, Macromolecules, 6, 672 (1973).
Y. Takahashi, I. Sumita, and H. Tadokoro, J. Polym. Sci., Polym. Phys. Ed., 11, 2113 (1973).
Y. Chatani, H. Tadokoro, T. Saegusa, and H. Ikeda, Macromolecules, 14, 315 (1981).
Y. Chatani, T. Kobatake, H. Tadokoro, and R. Tanaka, Macromolecules, 15, 170 (1982).
Y. Chatani, T. Kobatake, and H. Tadokoro, Macromolecules, 16, 199 (1983).
T. Hashida, K. Tashiro, S. Aoshima, and Y. Inaki, Macromolecules, 35, 4330 (2002).
C. S. Harris, M. A. Ratner, and D. F. Shriver, Macromolecules, 20, 1778 (1987).
A. Akinc, D. M. Lynn, D. G. Anderson, and R. Langer, J. Am. Chem. Soc., 125, 5316 (2003).
J. E. Mark and P. J. Flory, J. Am. Chem. Soc., 87, 1415 (1965).
J. E. Mark and P. J. Flory, J. Am. Chem. Soc., 87, 3702 (1966).
G. Geun and J. Breitkreutz, Pharmazie, 49, 562 (1994).
H. Dong, J. K. Hyun, C. Durham, and R. A. Wheeler, Polymer, 42, 7809 (2001).
S. Wang, L. DeBolt, and J. E. Mark, Polym. Prepr., 34, 478 (1993).
S. Tsuzuki, T. Uchimaru, K. Tanabe, and T. Hirano, J. Phys. Chem., 97, 1346 (1993).
R. L. Jaffe, G. D. Smith, and D. Y. Yoon, J. Phys. Chem., 97, 12745 (1993).
G. D. Smith, D. Y. Yoon, and R. L. Jaffe, Macromolecules, 26, 5213 (1993).
Y. Sasanuma, H. Ohta, I. Touma, H. Matoba, Y. Hayashi, and A. Kaito, Macromolecules, 35, 3748 (2002).
Y. Sasanuma and K. Sugita, Polym. J., 38, 983 (2006).
S. E. Boesch, S. S. York, R. Frech, and R. A. Wheeler, Phys. Chem. Commun., 4, 1 (2001).
Y. Sasanuma, S. Hattori, S. Imazu, S. Ikeda, T. Kaizuka, T. Iijima, M. Sawanobori, M. A. Azam, R. V. Law, and J. H. G. Steinke, Macromolecules, 37, 9169 (2004).
M. Kobayashi and H. Sato, Kobunshi Ronbunshu, 64, 119 (2007).
“Gaussian 03 User’s Reference,” Gaussian Inc., Carnegie, PA, 2003.
J. Rigaudy and S. P. Klesney, “Nomenclature of Organic Chemistry,” Section E, Pergamon Press, Oxford, 1979, p 483.
P. J. Flory, J. Am. Chem. Soc., 89, 1798 (1967).
H. Kusanagi and A. Ishimoto, Polym. Prepr. Jpn., 38, 3245 (1989).
H. Kusanagi, Polym. J., 25, 362 (1996).
H. Kusanagi, Polym. Prepr. Jpn., 53, 3610 (2004).
K. Ute, T. Nishimura, Y. Matsuura, and K. Hatada, Polym. J., 21, 231 (1989).
K. Ute, Y. Yamasaki, M. Naito, N. Miyatake, and K. Hatada, Polym. J., 27, 951 (1995).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kobayashi, M., Sato, H. Conformational Analysis of Ethylene Oxide and Ethylene Imine Oligomers by Quantum Chemical Calculation. Polym J 40, 343–349 (2008). https://doi.org/10.1295/polymj.PJ2007140
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.PJ2007140
Keywords
This article is cited by
-
Conformational analyses of an alanine oligomer during chain propagation using quantum chemical calculations
Polymer Journal (2015)
-
Conformational analysis for hydrated ethylene oxide oligomer models by quantum chemical calculations
Polymer Bulletin (2009)
-
Conformational analysis of ethylene oxide and ethylene imine oligomers by quantum chemical calculations: solvent effects
Polymer Bulletin (2008)