Abstract
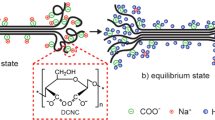
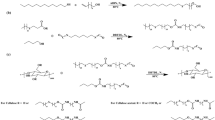
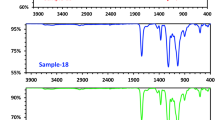
Formation of a polyelectrolyte complex was investigated as a function of pH by using carboxymethyl cellulose and poly(ethylenimine) as polyanion and polycation components, respectively. Experimental data on turbidity and conductometric and potentiometric titrations led to the conclusion that the formation of the polyelectrolyte complex did not obey stoichiometry. Such a result may be attributed to the less flexible nature of the polyanion molecular chains and to the distribution of ionizable groups on the chains of both components.
Similar content being viewed by others
Article PDF
References
A. Nakajima and H. Sato, Biopolymers, 11, 1345 (1972).
H. Terayama, Kagaku no Kenkyu, 1, 75 (1948).
H. Terayama, J. Polym. Sci., 8, 243 (1952).
A. S. Michaels, L. Mir, and N. S. Schneider, J. Polym. Sci., 69, 1447 (1965).
M. Hosono, S. Sugii, O. Kusudo, and W. Tsuji, Report of the Poval Committee, Kyoto, 61, 79 (1972).
R. M. Fuoss and H. Sadek, Science, 110, 552 (1949).
M. Hosono, O. Kusudo, and W. Tsuji, Report of the Poval Committee, Kyoto, 57, 99 (1970).
G. D. Jones A. Langsjoen, M. M. C. Neumann, and J. Zomlefer, J. Org. Chem., 9, 125 (1944).
H. G. Bungenberg de Jong, “Colloid Science,” Vol. II, H. R. Kruyt, Ed., Elsevier, Amsterdam, 1949.
E. S. Wajnerman, W. Ja. Grinberg, and W. B. Tolstogusow, Kolloid Z. Z. Polym., 250, 945 (1972).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sato, H., Nakajima, A. Formation of a Polyelectrolyte Complex from Carboxymethyl Cellulose and Poly(ethylenimine). Polym J 7, 241–247 (1975). https://doi.org/10.1295/polymj.7.241
Issue Date:
DOI: https://doi.org/10.1295/polymj.7.241