Abstract
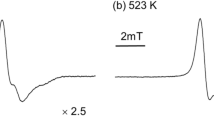
The cation radical of isoprene was first detected by means of ESR. It was produced by γ-irradiation in carbon tetrachloride, and has proton coupling constants of a1=13.0 G (2H), a2=4.9 G (3H), a3=3.2 G (1H), and a4=9.2 G (2H). In n-butyl chloride glass at 77°K a linear allyl-type radical was formed from isoprene. The initiation of polymerization by the isoprene cation radical was suggested in n-butyl chloride glass at 77°K during γ-irradiation. A cyclohexadienyl-type radical was produced from trans-1,3-pentadiene by γ-irradiation in carbon tetrachloride, whereas, a linear allyl-type radical was produced from cis-1,3-pentadiene. In a n-butyl chloride glass, a linear allyl-type radical was produced from both 1,3-pentadiene by γ-irradiation at 77°K. The cation radical were suggested to initiate radiation-induced polymerization in n-butyl chloride glass at 77°K.
Similar content being viewed by others
Article PDF
References
T. Shiga, A. Lund, and P. O. Kinell, Acta Chem. Scand., 29, 383 (1972).
T. Shiga and S. Okamura, Abstract, 15th Discussion Meeting of Japanese Society of Radiation Chemistry, Osaka, October 30, 1972, p 64.
T. Shiga, A. Lund, and P. O. Kinell, Int. J. Radiat. Phys. Chem., 3, 131 (1971).
T. Shiga, A. Lund, and P. O. Kinell, Acta Chem. Scand., 25, 1508 (1971).
J. Sohma, H. Kashiwabara, T. Komatsu, and T. Takahashi, Hokkaido Univ. Kogakubu Kenkyu Hohkoku, 35, 511 (1964).
L. C. Snyder and T. Amos, J. Chem. Phys., 42, 3670 (1965).
D. H. Levy, Mol. Phys., 9, 233 (1966).
S. Ohnishi, T. Tanei, and I. Nitta, J. Chem. Phys., 37, 2402 (1962).
C. Heller and H. M. McConnell, J. Chem. Phys., 32, 1535 (1960).
T. Yonezawa, Ed., “Ryoshi Kagaku Nyumon”, 3rd ed., Kagakudojin, Kyoto, 1969, p 218.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shiga, T., Okamura, S. ESR Studies on Primary Processes in Radiation-Induced Reactions. VI. Isoprene and 1,3-Pentadiene in Alkyl Halide Matrices. Polym J 6, 7–11 (1974). https://doi.org/10.1295/polymj.6.7
Issue Date:
DOI: https://doi.org/10.1295/polymj.6.7