Abstract
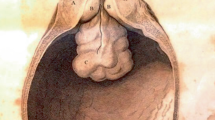
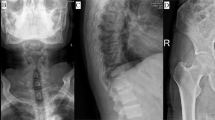
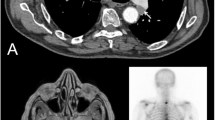
A 64-year-old male patient with a serum PSA value of 51.91 ng ml−1 and an magnetic resonance imaging scan suggestive of prostate cancer was admitted with bilateral hydronephrosis and anemia. A suprapubic cystostomy had been inserted 6 months earlier, owing to voiding difficulties. The patient was found to have huge multiple bladder stones and a massively thickened bladder wall. A sectio alta with bladder stone removal and bilateral ureteral stenting was performed. Histopathological examination revealed the growth of prostate cancer cells in the cystostomy tube needle tract.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Bernhardt J, Letzkus C, Kind M, Reith HB, Pfitzenmaier N . Metastasis of prostate carcinoma to the lamina submucosa of the distal rectum in ulcerative colitis 2 years after transrectal prostate biopsy and radical prostatovesiculectomy. Urologe A 2005; 44: 64–67.
Blight Jr EM . Seeding of prostate adenocarcinoma following transrectal needle biopsy. Urology 1992; 39: 297–298.
Greenstein A, Merimsky E, Baratz M, Braf Z . Late appearance of perineal implantation of prostatic carcinoma after perineal needle biopsy. Urology 1989; 33: 59–60.
Haddad FS, Somsin AA . Seeding and perineal implantation of prostatic cancer in the track of the biopsy needle: three case reports and a review of the literature. J Surg Oncol 1987; 35: 184–191.
Haddad FS . Delayed implantation of prostatic cancer after perineal needle biopsy. Urology 1989; 33: 351.
Ryan PG, Peeling WB . Perineal prostatic tumour seedling after ‘Tru-Cut’ needle biopsy: case report and review of the literature. Eur Urol 1990; 17: 189–192.
Vaghefi H, Magi-Galluzzi C, Klein EA . Local recurrence of prostate cancer in rectal submucosa after transrectal needle biopsy and radical prostatectomy. Urology 2005; 66: 881.
Zablow AI . Implantation of prostatic cancer after perineal needle biopsy. Urology 1989; 33: 449.
Bangma CH, Kirkels WJ, Chadha S, Schröder FH . Cutaneous metastasis following laparoscopic pelvic lymphadenectomy for prostatic carcinoma. J Urol 1995; 153: 1635–1636.
Boreham P . The surgical spread of cancer in urology. Br J Urol 1956; 28: 163.
Breul J, Block T, Breidenbach H, Hartung R . Implantation metastasis after a suprapubic catheter in a case of bladder cancer. Eur Urol 1992; 22: 86.
Orihuela E, Smith AD . Percutaneous treatment of transitional cell carcinoma of the upper urinary tract. Urol Clin North Am 1988; 15: 425.
Bastacky SS, Walsh PC, Epstein JI . Needle biopsy associated tumor tracking of adenocarcinoma of the prostate. J Urol 1991; 145: 1003–1007.
Moul JW, Miles BJ, Skoog SJ, Mc Leod DG . Risk factors for perineal seeding of prostate cancer after needle biopsy. J Urol 1989; 142: 86–88.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Horn, T., Seidl, S., Gschwend, J. et al. Implantation metastasis of prostate cancer in a suprapubic cystostomy tube needle tract. Prostate Cancer Prostatic Dis 13, 292–294 (2010). https://doi.org/10.1038/pcan.2009.52
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pcan.2009.52
Keywords
This article is cited by
-
Malignancies of suprapubic catheter (SPC) tracts in spinal cord injury patients: a case series and review of literature
Spinal Cord Series and Cases (2019)
-
Breast cancer neoplastic seeding in the setting of image-guided needle biopsies of the breast
Breast Cancer Research and Treatment (2017)