Abstract
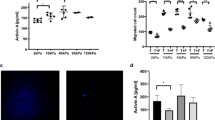
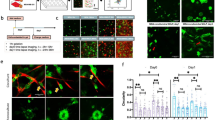
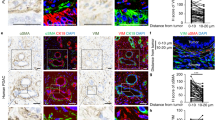
The transcription factor Twist1 is involved in the epithelial–mesenchymal transition and contributes to cancer metastasis through mostly unknown mechanisms. In colorectal cancer, Twist1 expression is mainly restricted to the tumor stroma. We found that human fibroblast cell lines stably transfected with Twist1 acquired characteristics of activated cancer-associated fibroblasts (CAFs), such as hyperproliferation, an increased ability to migrate and an alignment of the actin cytoskeleton. Further, Twist1-activated fibroblasts promoted increased matrix stiffness. Using quantitative proteomics, we identified palladin and collagen α1(VI) as two major mediators of the Twist1 effects in fibroblast cell lines. Co-immunoprecipitation studies indicated that palladin and Twist1 interact within the nucleus, suggesting that palladin could act as a transcription regulator. Palladin was found to be more relevant for the cellular biomechanical properties, orientation and polarity, and collagen α1(VI) for the migration and invasion capacity, of Twist1-activated fibroblasts. Both palladin and collagen α1(VI) were observed to be overexpressed in colorectal CAFs and to be associated with poor colorectal cancer patient survival and relapse prediction. Our results demonstrate that Twist1-expressing fibroblasts mimic the properties of CAFs present at the tumor invasive front, which likely explains the prometastatic activities of Twist1. Twist1 appears to require both palladin and collagen α1(VI) as downstream effectors for its prometastatic effects, which could be future therapeutic targets in cancer metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
De Craene B, Berx G . Regulatory networks defining EMT during cancer initiation and progression. Nat Rev Cancer 2013; 13: 97–110.
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008; 133: 704–715.
Polyak K, Weinberg RA . Transitions between epithelial and mesenchymal states: acquisition of malignant and stem cell traits. Nat Rev Cancer 2009; 9: 265–273.
Vermeulen L, De Sousa EMF, van der Heijden M, Cameron K, de Jong JH, Borovski T et al. Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat Cell Biol 2010; 12: 468–476.
Watson MA, Ylagan LR, Trinkaus KM, Gillanders WE, Naughton MJ, Weilbaecher KN et al. Isolation and molecular profiling of bone marrow micrometastases identifies TWIST1 as a marker of early tumor relapse in breast cancer patients. Clin Cancer Res 2007; 13: 5001–5009.
Eckert MA, Lwin TM, Chang AT, Kim J, Danis E, Ohno-Machado L et al. Twist1-induced invadopodia formation promotes tumor metastasis. Cancer Cell 2011; 19: 372–386.
Gomez I, Pena C, Herrera M, Munoz C, Larriba MJ, Garcia V et al. TWIST1 is expressed in colorectal carcinomas and predicts patient survival. PLoS One 2011; 6: e18023.
Ou DL, Chien HF, Chen CL, Lin TC, Lin LI . Role of Twist in head and neck carcinoma with lymph node metastasis. Anticancer Res 2008; 28: 1355–1359.
Xie F, Li K, Ouyang X . Twist, an independent prognostic marker for predicting distant metastasis and survival rates of esophageal squamous cell carcinoma patients. Clin Exp Metastasis 2009; 26: 1025–1032.
Tsai JH, Donaher JL, Murphy DA, Chau S, Yang J . Spatiotemporal regulation of epithelial-mesenchymal transition is essential for squamous cell carcinoma metastasis. Cancer Cell 2012; 22: 725–736.
Yang J, Mani SA, Donaher JL, Ramaswamy S, Itzykson RA, Come C et al. Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell 2004; 117: 927–939.
Sung CO, Lee KW, Han S, Kim SH . Twist1 is up-regulated in gastric cancer-associated fibroblasts with poor clinical outcomes. Am J Pathol 2011; 179: 1827–1838.
Valdes-Mora F, Gomez del Pulgar T, Bandres E, Cejas P, Ramirez de Molina A, Perez-Palacios R et al. TWIST1 overexpression is associated with nodal invasion and male sex in primary colorectal cancer. Ann Surg Oncol 2009; 16: 78–87.
Paszek MJ, Zahir N, Johnson KR, Lakins JN, Rozenberg GI, Gefen A et al. Tensional homeostasis and the malignant phenotype. Cancer Cell 2005; 8: 241–254.
Singh P, Carraher C, Schwarzbauer JE . Assembly of fibronectin extracellular matrix. Annu Rev Cell Dev Biol 2010; 26: 397–419.
Lee KW, Yeo SY, Sung CO, Kim SH . Twist1 is a key regulator of cancer-associated fibroblasts. Cancer Res 2015; 75: 73–85.
Amatangelo MD, Bassi DE, Klein-Szanto AJ, Cukierman E . Stroma-derived three-dimensional matrices are necessary and sufficient to promote desmoplastic differentiation of normal fibroblasts. Am J Pathol 2005; 167: 475–488.
Levental KR, Yu H, Kass L, Lakins JN, Egeblad M, Erler JT et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 2009; 139: 891–906.
Butcher DT, Alliston T, Weaver VM . A tense situation: forcing tumour progression. Nat Rev Cancer 2009; 9: 108–122.
Wei SC, Fattet L, Tsai JH, Guo Y, Pai VH, Majeski HE et al. Matrix stiffness drives epithelial-mesenchymal transition and tumour metastasis through a TWIST1-G3BP2 mechanotransduction pathway. Nat Cell Biol 2015; 17: 678–688.
Xu J, Lamouille S, Derynck R . TGF-beta-induced epithelial to mesenchymal transition. Cell Res 2009; 19: 156–172.
Wendt MK, Allington TM, Schiemann WP . Mechanisms of the epithelial-mesenchymal transition by TGF-beta. Future Oncol 2009; 5: 1145–1168.
Brentnall TA, Lai LA, Coleman J, Bronner MP, Pan S, Chen R . Arousal of cancer-associated stroma: overexpression of palladin activates fibroblasts to promote tumor invasion. PLoS One 2012; 7: e30219.
Chiu KH, Chang YH, Wu YS, Lee SH, Liao PC . Quantitative secretome analysis reveals that COL6A1 is a metastasis-associated protein using stacking gel-aided purification combined with iTRAQ labeling. J Proteome Res 2011; 10: 1110–1125.
Riaz M, Sieuwerts AM, Look MP, Timmermans MA, Smid M, Foekens JA et al. High TWIST1 mRNA expression is associated with poor prognosis in lymph node-negative and estrogen receptor-positive human breast cancer and is co-expressed with stromal as well as ECM related genes. Breast Cancer Res 2012; 14: R123.
Ohba K, Miyata Y, Matsuo T, Asai A, Mitsunari K, Shida Y et al. High expression of Twist is associated with tumor aggressiveness and poor prognosis in patients with renal cell carcinoma. Int J Clin Exp Pathol 2014; 7: 3158–3165.
Goicoechea SM, Bednarski B, Stack C, Cowan DW, Volmar K, Thorne L et al. Isoform-specific upregulation of palladin in human and murine pancreas tumors. PLoS One 2010; 5: e10347.
Gupta V, Bassi DE, Simons JD, Devarajan K, Al-Saleem T, Uzzo RG et al. Elevated expression of stromal palladin predicts poor clinical outcome in renal cell carcinoma. PLoS One 2011; 6: e21494.
Wan F, Wang H, Shen Y, Zhang H, Shi G, Zhu Y et al. Upregulation of COL6A1 is predictive of poor prognosis in clear cell renal cell carcinoma patients. Oncotarget 2015; 6: 27378–27387.
Calon A, Lonardo E, Berenguer-Llergo A, Espinet E, Hernando-Momblona X, Iglesias M et al. Stromal gene expression defines poor-prognosis subtypes in colorectal cancer. Nat Genet 2015; 47: 320–329.
Liu T, Yu H, Ding L, Wu Z, Gonzalez De Los Santos F, Liu J et al. Conditional knockout of telomerase reverse transcriptase in mesenchymal cells impairs mouse pulmonary fibrosis. PLoS One 2015; 10: e0142547.
Ronty MJ, Leivonen SK, Hinz B, Rachlin A, Otey CA, Kahari VM et al. Isoform-specific regulation of the actin-organizing protein palladin during TGF-beta1-induced myofibroblast differentiation. J Invest Dermatol 2006; 126: 2387–2396.
Goicoechea SM, Garcia-Mata R, Staub J, Valdivia A, Sharek L, McCulloch CG et al. Palladin promotes invasion of pancreatic cancer cells by enhancing invadopodia formation in cancer-associated fibroblasts. Oncogene 2014; 33: 1265–1273.
Endlich N, Schordan E, Cohen CD, Kretzler M, Lewko B, Welsch T et al. Palladin is a dynamic actin-associated protein in podocytes. Kidney Int 2009; 75: 214–226.
Liu XS, Luo HJ, Yang H, Wang L, Kong H, Jin YE et al. Palladin regulates cell and extracellular matrix interaction through maintaining normal actin cytoskeleton architecture and stabilizing beta1-integrin. J Cell Biochem 2007; 100: 1288–1300.
Jin L, Yoshida T, Ho R, Owens GK, Somlyo AV . The actin-associated protein Palladin is required for development of normal contractile properties of smooth muscle cells derived from embryoid bodies. J Biol Chem 2009; 284: 2121–2130.
Salaria SN, Illei P, Sharma R, Walter KM, Klein AP, Eshleman JR et al. Palladin is overexpressed in the non-neoplastic stroma of infiltrating ductal adenocarcinomas of the pancreas, but is only rarely overexpressed in neoplastic cells. Cancer Biol Ther 2007; 6: 324–328.
McLane JS, Ligon LA . Palladin mediates stiffness-induced fibroblast activation in the tumor microenvironment. Biophys J 2015; 109: 249–264.
Chin YR, Toker A . The actin-bundling protein palladin is an Akt1-specific substrate that regulates breast cancer cell migration. Mol Cell 2010; 38: 333–344.
Chin YR, Toker A . Akt2 regulates expression of the actin-bundling protein palladin. FEBS Lett 2010; 584: 4769–4774.
Cheng GZ, Zhang W, Wang LH . Regulation of cancer cell survival, migration, and invasion by Twist: AKT2 comes to interplay. Cancer Res 2008; 68: 957–960.
Cannon AR, Owen MK, Guerrero MS, Kerber ML, Goicoechea SM, Hemstreet KC et al. Palladin expression is a conserved characteristic of the desmoplastic tumor microenvironment and contributes to altered gene expression. Cytoskeleton (Hoboken) 2015; 72: 402–411.
Jin L, Gan Q, Zieba BJ, Goicoechea SM, Owens GK, Otey CA et al. The actin associated protein palladin is important for the early smooth muscle cell differentiation. PLoS One 2010; 5: e12823.
Somasundaram R, Schuppan D . Type I, II, III, IV, V, and VI collagens serve as extracellular ligands for the isoforms of platelet-derived growth factor (AA, BB, and AB). J Biol Chem 1996; 271: 26884–26891.
Groulx JF, Gagne D, Benoit YD, Martel D, Basora N, Beaulieu JF . Collagen VI is a basement membrane component that regulates epithelial cell-fibronectin interactions. Matrix Biol 2011; 30: 195–206.
Chen P, Cescon M, Bonaldo P . Collagen VI in cancer and its biological mechanisms. Trends Mol Med 2013; 19: 410–417.
Torres S, Garcia-Palmero I, Herrera M, Bartolome RA, Pena C, Fernandez-Acenero MJ et al. LOXL2 is highly expressed in cancer-associated fibroblasts and associates to poor colon cancer survival. Clin Cancer Res 2015; 21: 4892–4902.
Bodnar AG, Ouellette M, Frolkis M, Holt SE, Chiu CP, Morin GB et al. Extension of life-span by introduction of telomerase into normal human cells. Science 1998; 279: 349–352.
Mayne LV, Priestley A, James MR, Burke JF . Efficient immortalization and morphological transformation of human fibroblasts by transfection with SV40 DNA linked to a dominant marker. Exp Cell Res 1986; 162: 530–538.
Larriba MJ, Martin-Villar E, Garcia JM, Pereira F, Pena C, de Herreros AG et al. Snail2 cooperates with Snail1 in the repression of vitamin D receptor in colon cancer. Carcinogenesis 2009; 30: 1459–1468.
Bartolome RA, Garcia-Palmero I, Torres S, Lopez-Lucendo M, Balyasnikova IV, Casal JI . IL13 receptor alpha2 signaling requires a scaffold protein, FAM120A, to activate the FAK and PI3K pathways in colon cancer metastasis. Cancer Res 2015; 75: 2434–2444.
Bartolome RA, Pelaez-Garcia A, Gomez I, Torres S, Fernandez-Acenero MJ, Escudero-Paniagua B et al. An RGD motif present in cadherin 17 induces integrin activation and tumor growth. J Biol Chem 2014; 289: 34801–34814.
Bartolome RA, Barderas R, Torres S, Fernandez-Acenero MJ, Mendes M, Garcia-Foncillas J et al. Cadherin-17 interacts with alpha2beta1 integrin to regulate cell proliferation and adhesion in colorectal cancer cells causing liver metastasis. Oncogene 2014; 33: 1658–1669.
Acknowledgements
IG-P was supported by a contract S2010/BMD-2344/ Colomics2 from the Comunidad de Madrid. ST was a recipient of a Juan de la Cierva programme. RAB was supported by a grant to established research groups of the Asociación Española Contra el Cáncer (AECC). ML-L was a recipient of a ProteoRed contract. AP-G and BE-P were FPI fellows from the Ministry of Economy and Competitiveness (MINECO). This research was supported by grants to established research groups of the S2010/BMD-2344/Colomics2 from the Comunidad de Madrid, ‘Asociación Española Contra el Cancer (AECC)’, BIO2012-31023 from the MINECO, PRB2 (IPT13/0001-ISCIII-SGEFI/FEDER), RD12/0036/0041 and RD12/0036/0021 from the Instituto de Salud Carlos III-FEDER.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
García-Palmero, I., Torres, S., Bartolomé, R. et al. Twist1-induced activation of human fibroblasts promotes matrix stiffness by upregulating palladin and collagen α1(VI). Oncogene 35, 5224–5236 (2016). https://doi.org/10.1038/onc.2016.57
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.57
This article is cited by
-
Extracellular matrix remodeling in tumor progression and immune escape: from mechanisms to treatments
Molecular Cancer (2023)
-
What is new in cancer-associated fibroblast biomarkers?
Cell Communication and Signaling (2023)
-
CNS tumor stroma transcriptomics identify perivascular fibroblasts as predictors of immunotherapy resistance in glioblastoma patients
npj Genomic Medicine (2023)
-
Role of Interleukins in Inflammation-Mediated Tumor Immune Microenvironment Modulation in Colorectal Cancer Pathogenesis
Digestive Diseases and Sciences (2023)
-
Targeting extracellular matrix stiffness and mechanotransducers to improve cancer therapy
Journal of Hematology & Oncology (2022)