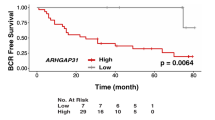
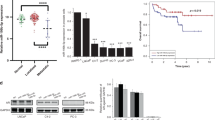
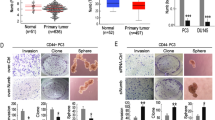
Abstract
MicroRNA-375 (miR-375) is frequently elevated in prostate tumors and cell-free fractions of patient blood, but its role in genesis and progression of prostate cancer is poorly understood. In this study, we demonstrated that miR-375 is inversely correlated with epithelial–mesenchymal transition signatures (EMT) in clinical samples and can drive mesenchymal–epithelial transition (MET) in model systems. Indeed, miR-375 potently inhibited invasion and migration of multiple prostate cancer lines. The transcription factor YAP1 was found to be a direct target of miR-375 in prostate cancer. Knockdown of YAP1 phenocopied miR-375 overexpression, and overexpression of YAP1 rescued anti-invasive effects mediated by miR-375. Furthermore, transcription of the miR-375 gene was shown to be directly repressed by the EMT transcription factor, ZEB1. Analysis of multiple patient cohorts provided evidence for this ZEB1-miR-375-YAP1 regulatory circuit in clinical samples. Despite its anti-invasive and anti-EMT capacities, plasma miR-375 was found to be correlated with circulating tumor cells in men with metastatic disease. Collectively, this study provides new insight into the function of miR-375 in prostate cancer, and more broadly identifies a novel pathway controlling epithelial plasticity and tumor cell invasion in this disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Bray F, Ren JS, Masuyer E, Ferlay J . Global estimates of cancer prevalence for 27 sites in the adult population in 2008. Int J Cancer 2013; 132: 1133–1145.
Wu JN, Fish KM, Evans CP, Devere White RW, Dall'Era MA . No improvement noted in overall or cause-specific survival for men presenting with metastatic prostate cancer over a 20-year period. Cancer 2014; 120: 818–823.
Das R, Gregory PA, Hollier BG, Tilley WD, Selth LA . Epithelial plasticity in prostate cancer: principles and clinical perspectives. Trends Mol Med 2014; 20: 643–651.
De Craene B, Berx G . Regulatory networks defining EMT during cancer initiation and progression. Nat Rev Cancer 2013; 13: 97–110.
Thiery JP . Epithelial-mesenchymal transitions in tumour progression. Nat Rev Cancer 2002; 2: 442–454.
Banyard J, Chung I, Wilson AM, Vetter G, Le Bechec A, Bielenberg DR et al. Regulation of epithelial plasticity by miR-424 and miR-200 in a new prostate cancer metastasis model. Sci Rep 2013; 3: 3151.
Celia-Terrassa T, Meca-Cortes O, Mateo F, de Paz AM, Rubio N, Arnal-Estape A et al. Epithelial-mesenchymal transition can suppress major attributes of human epithelial tumor-initiating cells. J Clin Invest 2012; 122: 1849–1868.
Korpal M, Ell BJ, Buffa FM, Ibrahim T, Blanco MA, Celia-Terrassa T et al. Direct targeting of Sec23a by miR-200 s influences cancer cell secretome and promotes metastatic colonization. Nature medicine 2011; 17: 1101–1108.
Ocana OH, Corcoles R, Fabra A, Moreno-Bueno G, Acloque H, Vega S et al. Metastatic colonization requires the repression of the epithelial-mesenchymal transition inducer Prrx1. Cancer Cell 2012; 22: 709–724.
Tsai JH, Donaher JL, Murphy DA, Chau S, Yang J . Spatiotemporal regulation of epithelial-mesenchymal transition is essential for squamous cell carcinoma metastasis. Cancer Cell 2012; 22: 725–736.
Gregory PA, Bracken CP, Bert AG, Goodall GJ . MicroRNAs as regulators of epithelial-mesenchymal transition. Cell cycle 2008; 7: 3112–3118.
Zhang J, Ma L . MicroRNA control of epithelial-mesenchymal transition and metastasis. Cancer Metast Rev 2012; 31: 653–662.
Selth LA, Tilley WD, Butler LM . Circulating microRNAs: macro-utility as markers of prostate cancer? Endocr Relat Cancer 2012; 19: R99–R113.
Ambs S, Prueitt RL, Yi M, Hudson RS, Howe TM, Petrocca F et al. Genomic profiling of microRNA and messenger RNA reveals deregulated microRNA expression in prostate cancer. Cancer Res 2008; 68: 6162–6170.
Martens-Uzunova ES, Jalava SE, Dits NF, van Leenders GJ, Moller S, Trapman J et al. Diagnostic and prognostic signatures from the small non-coding RNA transcriptome in prostate cancer. Oncogene 2011; 31: 978–991.
Schaefer A, Stephan C, Busch J, Yousef GM, Jung K . Diagnostic, prognostic and therapeutic implications of microRNAs in urologic tumors. Nat Rev Urol 2010; 7: 286–297.
Taylor BS, Schultz N, Hieronymus H, Gopalan A, Xiao Y, Carver BS et al. Integrative genomic profiling of human prostate cancer. Cancer Cell 2010; 18: 11–22.
Wach S, Nolte E, Szczyrba J, Stohr R, Hartmann A, Orntoft T et al. MicroRNA profiles of prostate carcinoma detected by multiplatform microRNA screening. Int J Cancer 2011; 130: 611–621.
Lin PC, Chiu YL, Banerjee S, Park K, Mosquera JM, Giannopoulou E et al. Epigenetic repression of miR-31 disrupts androgen receptor homeostasis and contributes to prostate cancer progression. Cancer Res 2013; 73: 1232–1244.
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 2005; 102: 15545–15550.
Kohn KW, Zeeberg BR, Reinhold WC, Sunshine M, Luna A, Pommier Y . Gene expression profiles of the NCI-60 human tumor cell lines define molecular interaction networks governing cell migration processes. PLoS One 2012; 7: e35716.
Yuan TC, Veeramani S, Lin FF, Kondrikou D, Zelivianski S, Igawa T et al. Androgen deprivation induces human prostate epithelial neuroendocrine differentiation of androgen-sensitive LNCaP cells. Endocr Relat Cancer 2006; 13: 151–167.
Ward A, Balwierz A, Zhang JD, Kublbeck M, Pawitan Y, Hielscher T et al. Re-expression of microRNA-375 reverses both tamoxifen resistance and accompanying EMT-like properties in breast cancer. Oncogene 2012; 32: 1173–1182.
Lokman NA, Elder AS, Ricciardelli C, Oehler MK . Chick chorioallantoic membrane (CAM) assay as an in vivo model to study the effect of newly identified molecules on ovarian cancer invasion and metastasis. Int J Mol Sci 2012; 13: 9959–9970.
Nishikawa E, Osada H, Okazaki Y, Arima C, Tomida S, Tatematsu Y et al. miR-375 is activated by ASH1 and inhibits YAP1 in a lineage-dependent manner in lung cancer. Cancer Res 2011; 71: 6165–6173.
Christensen LL, Holm A, Rantala J, Kallioniemi O, Rasmussen MH, Ostenfeld MS et al. Functional screening identifies miRNAs influencing apoptosis and proliferation in colorectal cancer. PLoS One 2014; 9: e96767.
Pan D . The hippo signaling pathway in development and cancer. Dev Cell 2010; 19: 491–505.
Ota M, Sasaki H . Mammalian Tead proteins regulate cell proliferation and contact inhibition as transcriptional mediators of Hippo signaling. Development 2008; 135: 4059–4069.
Shao DD, Xue W, Krall EB, Bhutkar A, Piccioni F, Wang X et al. KRAS and YAP1 converge to regulate EMT and tumor survival. Cell 2014; 158: 171–184.
Bracken CP, Gregory PA, Kolesnikoff N, Bert AG, Wang J, Shannon MF et al. A double-negative feedback loop between ZEB1-SIP1 and the microRNA-200 family regulates epithelial-mesenchymal transition. Cancer Res 2008; 68: 7846–7854.
Burk U, Schubert J, Wellner U, Schmalhofer O, Vincan E, Spaderna S et al. A reciprocal repression between ZEB1 and members of the miR-200 family promotes EMT and invasion in cancer cells. EMBO Rep 2008; 9: 582–589.
de Souza Rocha Simonini P, Breiling A, Gupta N, Malekpour M, Youns M, Omranipour R et al. Epigenetically deregulated microRNA-375 is involved in a positive feedback loop with estrogen receptor alpha in breast cancer cells. Cancer Res 2010; 70: 9175–9184.
Kolesnikoff N, Attema JL, Roslan S, Bert AG, Schwarz QP, Gregory PA et al. Specificity protein 1 (Sp1) maintains basal epithelial expression of the miR-200 family: implications for epithelial-mesenchymal transition. J Biol Chem 2014; 289: 11194–11205.
Consortium EP, Bernstein BE, Birney E, Dunham I, Green ED, Gunter C et al. An integrated encyclopedia of DNA elements in the human genome. Nature 2012; 489: 57–74.
Brase JC, Johannes M, Schlomm T, Falth M, Haese A, Steuber T et al. Circulating miRNAs are correlated with tumor progression in prostate cancer. Int J Cancer 2011; 128: 608–616.
Bryant RJ, Pawlowski T, Catto JW, Marsden G, Vessella RL, Rhees B et al. Changes in circulating microRNA levels associated with prostate cancer. Br J Cancer 2012; 106: 768–774.
Selth LA, Townley S, Gillis JL, Ochnik AM, Murti K, Macfarlane RJ et al. Discovery of circulating microRNAs associated with human prostate cancer using a mouse model of disease. Int J Cancer 2011; 131: 652–661.
Nauseef JT, Henry MD . Epithelial-to-mesenchymal transition in prostate cancer: paradigm or puzzle? Nat Rev Urol 2011; 8: 428–439.
Selth LA, Townley SL, Bert AG, Stricker PD, Sutherland PD, Horvath LG et al. Circulating microRNAs predict biochemical recurrence in prostate cancer patients. Br J Cancer 2013; 109: 641–650.
Madhavan D, Zucknick M, Wallwiener M, Cuk K, Modugno C, Scharpff M et al. Circulating miRNAs as surrogate markers for circulating tumor cells and prognostic markers in metastatic breast cancer. Clin Cancer Res 2012; 18: 5972–5982.
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008; 133: 704–715.
Chu M, Chang Y, Li P, Guo Y, Zhang K, Gao W . Androgen receptor is negatively correlated with the methylation-mediated transcriptional repression of miR-375 in human prostate cancer cells. Oncol Rep 2014; 31: 34–40.
Costa-Pinheiro P, Ramalho-Carvalho J, Vieira FQ, Torres-Ferreira J, Oliveira J, Goncalves CS et al. MicroRNA-375 plays a dual role in prostate carcinogenesis. Clin Epigenet 2015; 7: 42.
Bitting RL, Schaeffer D, Somarelli JA, Garcia-Blanco MA, Armstrong AJ . The role of epithelial plasticity in prostate cancer dissemination and treatment resistance. Cancer Metast Rev 2014; 33: 441–468.
Blower PE, Verducci JS, Lin S, Zhou J, Chung JH, Dai Z et al. MicroRNA expression profiles for the NCI-60 cancer cell panel. Mol Cancer Ther 2007; 6: 1483–1491.
Liu H, D'Andrade P, Fulmer-Smentek S, Lorenzi P, Kohn KW, Weinstein JN et al. mRNA and microRNA expression profiles of the NCI-60 integrated with drug activities. Mol Cancer Ther 2010; 9: 1080–1091.
Sokilde R, Kaczkowski B, Podolska A, Cirera S, Gorodkin J, Moller S et al. Global microRNA analysis of the NCI-60 cancer cell panel. Mol Cancer Ther 2011; 10: 375–384.
Moore NL, Buchanan G, Harris JM, Selth LA, Bianco-Miotto T, Hanson AR et al. An androgen receptor mutation in the MDA-MB-453 cell line model of molecular apocrine breast cancer compromises receptor activity. Endocr Relat Cancer 2012; 19: 599–613.
Yu X, Machesky LM . Cells assemble invadopodia-like structures and invade into matrigel in a matrix metalloprotease dependent manner in the circular invasion assay. PLoS One 2012; 7: e30605.
Oka T, Mazack V, Sudol M . Mst2 and Lats kinases regulate apoptotic function of Yes kinase-associated protein (YAP). J Biol Chem 2008; 283: 27534–27546.
Neilsen PM, Noll JE, Mattiske S, Bracken CP, Gregory PA, Schulz RB et al. Mutant p53 drives invasion in breast tumors through up-regulation of miR-155. Oncogene 2013; 32: 2992–3000.
Marrocco DL, Tilley WD, Bianco-Miotto T, Evdokiou A, Scher HI, Rifkind RA et al. Suberoylanilide hydroxamic acid (vorinostat) represses androgen receptor expression and acts synergistically with an androgen receptor antagonist to inhibit prostate cancer cell proliferation. Mol Cancer Ther 2007; 6: 51–60.
Dupont S, Morsut L, Aragona M, Enzo E, Giulitti S, Cordenonsi M et al. Role of YAP/TAZ in mechanotransduction. Nature 2011; 474: 179–183.
Schmidt D, Wilson MD, Spyrou C, Brown GD, Hadfield J, Odom DT . ChIP-seq: using high-throughput sequencing to discover protein-DNA interactions. Methods 2009; 48: 240–248.
Gasch C, Plummer PN, Jovanovic L, McInnes LM, Wescott D, Saunders CM et al. Heterogeneity of miR-10b expression in circulating tumor cells. Sci Rep 2015; 5: 15980.
Acknowledgements
The YAP1 3′-UTR plasmids were a kind gift from Prof Takashi Takahashi, Nagoya University. The results published here are in part based on the data generated by The Cancer Genome Atlas, established by the National Cancer Institute and the National Human Genome Research Institute, and we are grateful to the specimen donors and relevant research groups associated with this project. We also acknowledge the ENCODE Consortium and the Myers laboratory at the HudsonAlpha Institute for Biotechnology for generating the ZEB1 ChIP-seq data set used in this study. This work was supported by: Young Investigator Awards from the Prostate Cancer Foundation (the Foundation 14 award; LAS) and the Prostate Cancer Foundation of Australia (PCFA)/Movember (YI 0810; LAS, YI 0412; MMC) and grants from PCFA/Movember/Cancer Australia (Grant IDs 1012337 and 1043482; WDT, LAS and LMB) and the National Health and Medical Research Council (Grant ID 1083961). PAG is supported by a Beat Cancer Project fellowship from the Cancer Council of South Australia. BGH is supported by a Queensland government Smart Futures Fund Fellowship. The research programs of LAS, MMC, LMB, CCN, BGH and WDT are supported by the Movember Foundation and the Prostate Cancer Foundation of Australia through Movember Revolutionary Team Awards.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Selth, L., Das, R., Townley, S. et al. A ZEB1-miR-375-YAP1 pathway regulates epithelial plasticity in prostate cancer. Oncogene 36, 24–34 (2017). https://doi.org/10.1038/onc.2016.185
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.185
This article is cited by
-
An integrated ceRNA network identifies miR-375 as an upregulated miRNA playing a tumor suppressive role in aggressive prostate cancer
Oncogene (2024)
-
The interplay between noncoding RNA and YAP/TAZ signaling in cancers: molecular functions and mechanisms
Journal of Experimental & Clinical Cancer Research (2022)
-
Cancer-derived exosomal miR-375 targets DIP2C and promotes osteoblastic metastasis and prostate cancer progression by regulating the Wnt signaling pathway
Cancer Gene Therapy (2022)
-
MicroRNA-375 is a therapeutic target for castration-resistant prostate cancer through the PTPN4/STAT3 axis
Experimental & Molecular Medicine (2022)
-
The deadly cross-talk between Hippo pathway and epithelial–mesenchymal transition (EMT) in cancer
Molecular Biology Reports (2022)