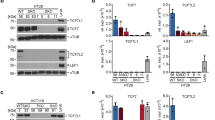
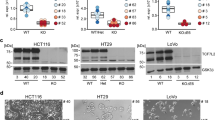
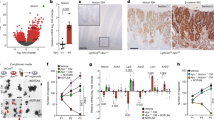
Abstract
Human colorectal cancer is often initiated by the aberrant activation of Wnt signaling, notably following adenomatous polyposis coli (Apc) inactivation. Recent studies identified adult intestinal stem cells (ISCs) and demonstrated their role as the cells of origin for intestinal tumors. However, the early consequences of aberrant Wnt signaling activation remain to be fully elucidated. Here, using organoid cultures established from conditional knockout mice and in vitro gene ablation, we show that Apc inactivation led to aberrant ISC proliferation and the expansion of the crypt domain. This system was used to evaluate the potential of a cancer-related spindle protein, Tacc3, as a target of cancer therapy, as its disruption led to the suppression of tumor formation in an animal model of intestinal tumors. We found that Tacc3 is required for the proper mitosis of Apc-deficient ISCs, and its disruption significantly attenuated the expansion of the crypt domain. In vivo analysis of corresponding mutant mice demonstrated that Tacc3 disruption led to a significant decrease in tumor number and prolonged survival. These observations demonstrated that Tacc3 is a potential chemotherapeutic target for intestinal tumors by perturbing the aberrant cell proliferation of Apc-deficient ISCs and provides an opportunity for the development of novel cancer prevention and treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Sato T, van Es JH, Snippert HJ, Stange DE, Vries RG, van den Born M et al. Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature 2011; 469: 415–418.
Farin HF, Van Es JH, Clevers H . Redundant sources of Wnt regulate intestinal stem cells and promote formation of Paneth cells. Gastroenterology 2012; 143: 1518–1529 e1517.
Kabiri Z, Greicius G, Madan B, Biechele S, Zhong Z, Zaribafzadeh H et al. Stroma provides an intestinal stem cell niche in the absence of epithelial Wnts. Development 2014; 141: 2206–2215.
San Roman AK, Jayewickreme CD, Murtaugh LC, Shivdasani RA . Wnt secretion from epithelial cells and subepithelial myofibroblasts is not required in the mouse intestinal stem cell niche in vivo. Stem Cell Rep 2014; 2: 127–134.
Najdi R, Proffitt K, Sprowl S, Kaur S, Yu J, Covey TM et al. A uniform human Wnt expression library reveals a shared secretory pathway and unique signaling activities. Differentiation 2012; 84: 203–213.
Clevers H, Loh KM, Nusse R . Stem cell signaling. An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control. Science 2014; 346: 1248012.
Cancer Genome Atlas N.. Comprehensive molecular characterization of human colon and rectal cancer. Nature 2012; 487: 330–337.
Barker N, Ridgway RA, van Es JH, van de Wetering M, Begthel H, van den Born M et al. Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 2009; 457: 608–611.
Powell AE, Wang Y, Li Y, Poulin EJ, Means AL, Washington MK et al. The pan-ErbB negative regulator Lrig1 is an intestinal stem cell marker that functions as a tumor suppressor. Cell 2012; 149: 146–158.
Sangiorgi E, Capecchi MR . Bmi1 is expressed in vivo in intestinal stem cells. Nat Genet 2008; 40: 915–920.
Harrison MR, Holen KD, Liu G . Beyond taxanes: a review of novel agents that target mitotic tubulin and microtubules, kinases, and kinesins. Clin Adv Hematol Oncol 2009; 7: 54–64.
Jordan MA, Wilson L . Microtubules as a target for anticancer drugs. Nat Rev Cancer 2004; 4: 253–265.
Sudakin V, Yen TJ . Targeting mitosis for anti-cancer therapy. BioDrugs 2007; 21: 225–233.
Lauffart B, Vaughan MM, Eddy R, Chervinsky D, DiCioccio RA, Black JD et al. Aberrations of TACC1 and TACC3 are associated with ovarian cancer. BMC Womens Health 2005; 5: 8.
Still IH, Vince P, Cowell JK . The third member of the transforming acidic coiled coil-containing gene family, TACC3, maps in 4p16, close to translocation breakpoints in multiple myeloma, and is upregulated in various cancer cell lines. Genomics 1999; 58: 165–170.
Brouhard GJ, Stear JH, Noetzel TL, Al-Bassam J, Kinoshita K, Harrison SC et al. XMAP215 is a processive microtubule polymerase. Cell 2008; 132: 79–88.
Lee MJ, Gergely F, Jeffers K, Peak-Chew SY, Raff JW . Msps/XMAP215 interacts with the centrosomal protein D-TACC to regulate microtubule behaviour. Nat Cell Biol 2001; 3: 643–649.
Charrasse S, Mazel M, Taviaux S, Berta P, Chow T, Larroque C . Characterization of the cDNA and pattern of expression of a new gene over-expressed in human hepatomas and colonic tumors. Eur J Biochem 1995; 234: 406–413.
Sato T, Vries RG, Snippert HJ, van de Wetering M, Barker N, Stange DE et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 2009; 459: 262–265.
Yao R, Natsume Y, Noda T . TACC3 is required for the proper mitosis of sclerotome mesenchymal cells during formation of the axial skeleton. Cancer Sci 2007; 98: 555–562.
Yao R, Natsume Y, Saiki Y, Shioya H, Takeuchi K, Yamori T et al. Disruption of Tacc3 function leads to in vivo tumor regression. Oncogene 2012; 31: 135–148.
Sansom OJ, Meniel VS, Muncan V, Phesse TJ, Wilkins JA, Reed KR et al. Myc deletion rescues Apc deficiency in the small intestine. Nature 2007; 446: 676–679.
Munoz J, Stange DE, Schepers AG, van de Wetering M, Koo BK, Itzkovitz S et al. The Lgr5 intestinal stem cell signature: robust expression of proposed quiescent '+4' cell markers. EMBO J 2012; 31: 3079–3091.
Snippert HJ, van Es JH, van den Born M, Begthel H, Stange DE, Barker N et al. Prominin-1/CD133 marks stem cells and early progenitors in mouse small intestine. Gastroenterology 2009; 136: 2187–2194 e2181.
Dalerba P, Dylla SJ, Park IK, Liu R, Wang X, Cho RW et al. Phenotypic characterization of human colorectal cancer stem cells. Proc Natl Acad Sci USA 2007; 104: 10158–10163.
O'Brien CA, Pollett A, Gallinger S, Dick JE . A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature 2007; 445: 106–110.
Ricci-Vitiani L, Lombardi DG, Pilozzi E, Biffoni M, Todaro M, Peschle C et al. Identification and expansion of human colon-cancer-initiating cells. Nature 2007; 445: 111–115.
Todaro M, Francipane MG, Medema JP, Stassi G . Colon cancer stem cells: promise of targeted therapy. Gastroenterology 2010; 138: 2151–2162.
Batlle E, Henderson JT, Beghtel H, van den Born MM, Sancho E, Huls G et al. Beta-catenin and TCF mediate cell positioning in the intestinal epithelium by controlling the expression of EphB/ephrinB. Cell 2002; 111: 251–263.
Jensen KB, Collins CA, Nascimento E, Tan DW, Frye M, Itami S et al. Lrig1 expression defines a distinct multipotent stem cell population in mammalian epidermis. Cell Stem Cell 2009; 4: 427–439.
Fatehullah A, Appleton PL, Nathke IS . Cell and tissue polarity in the intestinal tract during tumourigenesis: cells still know the right way up, but tissue organization is lost. Philos Trans R Soc Lond B Biol Sci 2013; 368: 20130014.
Sansom OJ, Reed KR, Hayes AJ, Ireland H, Brinkmann H, Newton IP et al. Loss of Apc in vivo immediately perturbs Wnt signaling, differentiation, and migration. Genes Dev 2004; 18: 1385–1390.
van de Wetering M, Sancho E, Verweij C, de Lau W, Oving I, Hurlstone A et al. The beta-catenin/TCF-4 complex imposes a crypt progenitor phenotype on colorectal cancer cells. Cell 2002; 111: 241–250.
Gergely F, Draviam VM, Raff JW . The ch-TOG/XMAP215 protein is essential for spindle pole organization in human somatic cells. Genes Dev 2003; 17: 336–341.
Conte N, Delaval B, Ginestier C, Ferrand A, Isnardon D, Larroque C et al. TACC1-chTOG-Aurora A protein complex in breast cancer. Oncogene 2003; 22: 8102–8116.
Merlos-Suarez A, Barriga FM, Jung P, Iglesias M, Cespedes MV, Rossell D et al. The intestinal stem cell signature identifies colorectal cancer stem cells and predicts disease relapse. Cell Stem Cell 2011; 8: 511–524.
Wong VW, Stange DE, Page ME, Buczacki S, Wabik A, Itami S et al. Lrig1 controls intestinal stem-cell homeostasis by negative regulation of ErbB signalling. Nat Cell Biol 2012; 14: 401–408.
Drost J, van Jaarsveld RH, Ponsioen B, Zimberlin C, van Boxtel R, Buijs A et al. Sequential cancer mutations in cultured human intestinal stem cells. Nature 2015; 521: 43–47.
Matano M, Date S, Shimokawa M, Takano A, Fujii M, Ohta Y et al. Modeling colorectal cancer using CRISPR-Cas9-mediated engineering of human intestinal organoids. Nat Med 2015; 21: 256–262.
Holik AZ, Young M, Krzystyniak J, Williams GT, Metzger D, Shorning BY et al. Brg1 loss attenuates aberrant wnt-signalling and prevents wnt-dependent tumourigenesis in the murine small intestine. PLoS Genet 2014; 10: e1004453.
Phesse TJ, Parry L, Reed KR, Ewan KB, Dale TC, Sansom OJ et al. Deficiency of Mbd2 attenuates Wnt signaling. Mol Cell Biol 2008; 28: 6094–6103.
Shibata H, Toyama K, Shioya H, Ito M, Hirota M, Hasegawa S et al. Rapid colorectal adenoma formation initiated by conditional targeting of the Apc gene. Science 1997; 278: 120–123.
Madison BB, Dunbar L, Qiao XT, Braunstein K, Braunstein E, Gumucio DL . Cis elements of the villin gene control expression in restricted domains of the vertical (crypt) and horizontal (duodenum, cecum) axes of the intestine. J Biol Chem 2002; 277: 33275–33283.
Barker N, van Es JH, Kuipers J, Kujala P, van den Born M, Cozijnsen M et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 2007; 449: 1003–1007.
Ventura A, Kirsch DG, McLaughlin ME, Tuveson DA, Grimm J, Lintault L et al. Restoration of p53 function leads to tumour regression in vivo. Nature 2007; 445: 661–665.
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 2005; 102: 15545–15550.
Acknowledgements
We thank Dr Tomoyuki Kitagawa for continuous support and Ms Yuki Ota for technical assistance with the microarray analysis. We also thank Dr Toshiro Sato for valuable advice on the mouse intestine organoid culture. This study is partially supported by the research program for the Project for Development of Innovative Research on Cancer Therapeutics, P-Direct from the Japan Agency for Medical Research and Development, AMED (RY), Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (RY), and The Vehicle Racing Commemorative Foundation (RY).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Yao, R., Oyanagi, J., Natsume, Y. et al. Suppression of intestinal tumors by targeting the mitotic spindle of intestinal stem cells. Oncogene 35, 6109–6119 (2016). https://doi.org/10.1038/onc.2016.148
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.148
This article is cited by
-
E2F1 promotes cell cycle progression by stabilizing spindle fiber in colorectal cancer cells
Cellular & Molecular Biology Letters (2022)