Abstract
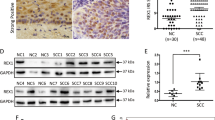
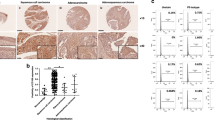
α-Actinin-4 (ACTN4) is frequently amplified and overexpressed in various cancers. Although ACTN4 functions in cancer cell migration and invasion, the roles of ACTN4 during the epithelial-to-mesenchymal transition (EMT) and cervical cancer tumorigenesis are unknown. In this study, we investigated the function of ACTN4 in the progression of cervical cancer and the mechanisms of EMT and tumorigenesis induced by ACTN4. We found that ACTN4 induced EMT by upregulating Snail, which was dependent on the Akt signaling pathway in cervical cancer. ACTN4 induced cell migration and invasion through Snail-mediated matrix metalloproteinase-9 expression. ACTN4 expression level was correlated with stabilization of β-catenin. Accumulatioin of β-catenin owing to ACTN4 induced tumorigenesis via upregulation of genes involved in cell proliferation, including cyclin D1 and c-myc. ACTN4 knockdown reduced cervical cancer cell proliferation and tumor formation in vivo. The expression level of ACTN4 is highly elevated in human cervical tumors, compared with that in normal cervical tissues. ACTN4-overexpressing MDCK cells induced tumor formation and metastatic nodules in nude mice. Our findings indicate that ACTN4 promotes EMT and tumorigenesis by regulating Snail expression and the Akt pathway in cervical cancer. We propose a novel mechanism for EMT and tumorigenesis in cervical cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Di Croce L, Pelicci PG . Tumour-associated hypermethylation: silencing E-cadherin expression enhances invasion and metastasis. Eur J Cancer 2003; 39: 413–414.
Eastham AM, Spencer H, Soncin F, Ritson S, Merry CL, Stern PL et al. Epithelial-mesenchymal transition events during human embryonic stem cell differentiation. Cancer Res 2007; 67: 11254–11262.
Chausovsky A, Bershadsky AD, Borisy GG . Cadherin-mediated regulation of microtubule dynamics. Nat Cell Biol 2000; 2: 797–804.
Gumbiner BM . Regulation of cadherin-mediated adhesion in morphogenesis. Nat Rev Mol Cell Biol 2005; 6: 622–634.
Jawhari A, Jordan S, Poole S, Browne P, Pignatelli M, Farthing MJ . Abnormal immunoreactivity of the E-cadherin-catenin complex in gastric carcinoma: relationship with patient survival. Gastroenterology 1997; 112: 46–54.
Nieman MT, Prudoff RS, Johnson KR, Wheelock MJ . N-cadherin promotes motility in human breast cancer cells regardless of their E-cadherin expression. J Cell Biol 1999; 147: 631–644.
Hazan RB, Phillips GR, Qiao RF, Norton L, Aaronson SA . Exogenous expression of N-cadherin in breast cancer cells induces cell migration, invasion, and metastasis. J Cell Biol 2000; 148: 779–790.
Seidel B, Braeg S, Adler G, Wedlich D, Menke A . E- and N-cadherin differ with respect to their associated p120ctn isoforms and their ability to suppress invasive growth in pancreatic cancer cells. Oncogene 2004; 23: 5532–5542.
Bolos V, Peinado H, Perez-Moreno MA, Fraga MF, Esteller M, Cano A . The transcription factor Slug represses E-cadherin expression and induces epithelial to mesenchymal transitions: a comparison with Snail and E47 repressors. J Cell Sci 2003; 116: 499–511.
Medici D, Hay ED, Olsen BR . Snail and Slug promote epithelial-mesenchymal transition through beta-catenin-T-cell factor-4-dependent expression of transforming growth factor-beta3. Mol Biol Cell 2008; 19: 4875–4887.
Ohkubo T, Ozawa M . The transcription factor Snail downregulates the tight junction components independently of E-cadherin downregulation. J Cell Sci 2004; 117: 1675–1685.
Jorda M, Olmeda D, Vinyals A, Valero E, Cubillo E, Llorens A et al. Upregulation of MMP-9 in MDCK epithelial cell line in response to expression of the Snail transcription factor. J Cell Sci 2005; 118: 3371–3385.
Cadigan KM, Nusse R . Wnt signaling: a common theme in animal development. Genes Dev 1997; 11: 3286–3305.
Logan CY, Nusse R . The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol 2004; 20: 781–810.
Ilyas M, Tomlinson IP . The interactions of APC, E-cadherin and beta-catenin in tumour development and progression. J Pathol 1997; 182: 128–137.
Polakis P . Wnt signaling and cancer. Genes Dev 2000; 14: 1837–1851.
Filali M, Cheng N, Abbott D, Leontiev V, Engelhardt JF . Wnt-3 A/beta-catenin signaling induces transcription from the LEF-1 promoter. J Biol Chem 2002; 277: 33398–33410.
Shimizu H, Julius MA, Giarre M, Zheng Z, Brown AM, Kitajewski J . Transformation by Wnt family proteins correlates with regulation of beta-catenin. Cell Growth Differ 1997; 8: 1349–1358.
Zeng L, Fagotto F, Zhang T, Hsu W, Vasicek TJ, Perry WL 3rd et al. The mouse Fused locus encodes Axin, an inhibitor of the Wnt signaling pathway that regulates embryonic axis formation. Cell 1997; 90: 181–192.
Altomare DA, Wang HQ, Skele KL, De Rienzo A, Klein-Szanto AJ, Godwin AK et al. AKT and mTOR phosphorylation is frequently detected in ovarian cancer and can be targeted to disrupt ovarian tumor cell growth. Oncogene 2004; 23: 5853–5857.
Hamada F, Tomoyasu Y, Takatsu Y, Nakamura M, Nagai S, Suzuki A et al. Negative regulation of Wingless signaling by D-axin, a Drosophila homolog of axin. Science 1999; 283: 1739–1742.
Honda K, Yamada T, Endo R, Ino Y, Gotoh M, Tsuda H et al. Actinin-4, a novel actin-bundling protein associated with cell motility and cancer invasion. J Cell Biol 1998; 140: 1383–1393.
Babakov VN, Petukhova OA, Turoverova LV, Kropacheva IV, Tentler DG, Bolshakova AV et al. RelA/NF-kappaB transcription factor associates with alpha-actinin-4. Exp Cell Res 2008; 314: 1030–1038.
Bolshakova A, Petukhova O, Turoverova L, Tentler D, Babakov V, Magnusson KE et al. Extra-cellular matrix proteins induce re-distribution of alpha-actinin-1 and alpha-actinin-4 in A431 cells. Cell Biol Int 2007; 31: 360–365.
Kumeta M, Yoshimura SH, Harata M, Takeyasu K . Molecular mechanisms underlying nucleocytoplasmic shuttling of actinin-4. J Cell Sci 2010; 123: 1020–1030.
Shao H, Wang JH, Pollak MR, Wells A . alpha-actinin-4 is essential for maintaining the spreading, motility and contractility of fibroblasts. PLoS One 2010; 5: e13921.
Honda K, Yamada T, Hayashida Y, Idogawa M, Sato S, Hasegawa F et al. Actinin-4 increases cell motility and promotes lymph node metastasis of colorectal cancer. Gastroenterology 2005; 128: 51–62.
Aksenova V, Turoverova L, Khotin M, Magnusson KE, Tulchinsky E, Melino G et al. Actin-binding protein alpha-actinin 4 (ACTN4) is a transcriptional co-activator of RelA/p65 sub-unit of NF-kB. Oncotarget 2013; 4: 362–372.
Chakraborty S, Reineke EL, Lam M, Li X, Liu Y, Gao C et al. Alpha-actinin 4 potentiates myocyte enhancer factor-2 transcription activity by antagonizing histone deacetylase 7. J Biol Chem 2006; 281: 35070–35080.
An HT, Kim J, Yoo S, Ko J . sLZIP negatively regulates skeletal muscle differentiation via interaction with alpha-actinin-4. J Biol Chem 2013; 289: 4969–4979.
Ding Z, Liang J, Lu Y, Yu Q, Songyang Z, Lin SY et al. A retrovirus-based protein complementation assay screen reveals functional AKT1-binding partners. Proc Natl Acad Sci USA 2006; 103: 15014–15019.
Hayashida Y, Honda K, Idogawa M, Ino Y, Ono M, Tsuchida A et al. E-cadherin regulates the association between beta-catenin and actinin-4. Cancer Res 2005; 65: 8836–8845.
Yamamoto S, Tsuda H, Honda K, Onozato K, Takano M, Tamai S et al. Actinin-4 gene amplification in ovarian cancer: a candidate oncogene associated with poor patient prognosis and tumor chemoresistance. Mod Pathol 2009; 22: 499–507.
Zhou BP, Deng J, Xia W, Xu J, Li YM, Gunduz M et al. Dual regulation of Snail by GSK-3beta-mediated phosphorylation in control of epithelial-mesenchymal transition. Nat Cell Biol 2004; 6: 931–940.
Nabeshima K, Inoue T, Shimao Y, Sameshima T . Matrix metalloproteinases in tumor invasion: role for cell migration. Pathol Int 2002; 52: 255–264.
Bjorklund M, Koivunen E . Gelatinase-mediated migration and invasion of cancer cells. Biochim Biophys Acta 2005; 1755: 37–69.
Xu D, McKee CM, Cao Y, Ding Y, Kessler BM, Muschel RJ . Matrix metalloproteinase-9 regulates tumor cell invasion through cleavage of protease nexin-1. Cancer Res 2010; 70: 6988–6998.
Testa JR, Bellacosa A . AKT plays a central role in tumorigenesis. Proc Natl Acad Sci USA 2001; 98: 10983–10985.
Bertelsen BI, Steine SJ, Sandvei R, Molven A, Laerum OD . Molecular analysis of the PI3K-AKT pathway in uterine cervical neoplasia: frequent PIK3CA amplification and AKT phosphorylation. Int J Cancer 2006; 118: 1877–1883.
Brognard J, Clark AS, Ni Y, Dennis PA . Akt/protein kinase B is constitutively active in non-small cell lung cancer cells and promotes cellular survival and resistance to chemotherapy and radiation. Cancer Res 2001; 61: 3986–3997.
Nakayama H, Ikebe T, Beppu M, Shirasuna K . High expression levels of nuclear factor kappaB, IkappaB kinase alpha and Akt kinase in squamous cell carcinoma of the oral cavity. Cancer 2001; 92: 3037–3044.
Prasad SB, Yadav SS, Das M, Govardhan HB, Pandey LK, Singh S et al. Down regulation of FOXO1 promotes cell proliferation in cervical cancer. J Cancer 2014; 5: 655–662.
Schmidt-Ott KM, Masckauchan TN, Chen X, Hirsh BJ, Sarkar A, Yang J et al. Beta-catenin/TCF/Lef controls a differentiation-associated transcriptional program in renal epithelial progenitors. Development 2007; 134: 3177–3190.
Rocheleau CE, Downs WD, Lin R, Wittmann C, Bei Y, Cha YH et al. Wnt signaling and an APC-related gene specify endoderm in early C. elegans embryos. Cell 1997; 90: 707–716.
Eastman Q, Grosschedl R . Regulation of LEF-1/TCF transcription factors by Wnt and other signals. Curr Opin Cell Biol 1999; 11: 233–240.
Barker N, Clevers H . Catenins, Wnt signaling and cancer. Bioessays 2000; 22: 961–965.
Kang H, Jang SW, Ko J . Human leucine zipper protein sLZIP induces migration and invasion of cervical cancer cells via expression of matrix metalloproteinase-9. J Biol Chem 2011; 286: 42072–42081.
Acknowledgements
This work was supported by Basic Science Research Program (NRF-2014R1A2A2A01002826) and Tunneling Nanotube Research Center (NRF-2015R1A5A1009024) through the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) and the Korea University Grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
An, HT., Yoo, S. & Ko, J. α-Actinin-4 induces the epithelial-to-mesenchymal transition and tumorigenesis via regulation of Snail expression and β-catenin stabilization in cervical cancer. Oncogene 35, 5893–5904 (2016). https://doi.org/10.1038/onc.2016.117
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.117
This article is cited by
-
H3K27ac-activated EGFR-AS1 promotes cell growth in cervical cancer through ACTN4-mediated WNT pathway
Biology Direct (2022)
-
CDX2 inhibits epithelial–mesenchymal transition in colorectal cancer by modulation of Snail expression and β-catenin stabilisation via transactivation of PTEN expression
British Journal of Cancer (2021)
-
Stopping transformed cancer cell growth by rigidity sensing
Nature Materials (2020)
-
Prognostic impact of ACTN4 gene copy number alteration in hormone receptor-positive, HER2-negative, node-negative invasive breast carcinoma
British Journal of Cancer (2020)
-
Replicative senescence in MSCWJ-1 human umbilical cord mesenchymal stem cells is marked by characteristic changes in motility, cytoskeletal organization, and RhoA localization
Molecular Biology Reports (2020)