Abstract
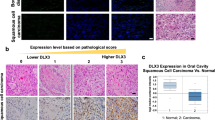
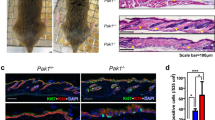
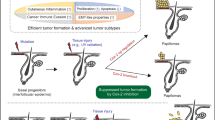
The small GTPase Rac1 is crucial for maintaining stem cells (SCs) in mammalian epidermis, and Rac1 activation leads to SC expansion. Loss or inhibition of Rac1 correlates with decreased frequency of skin cancer formation in a chemical carcinogenesis model. Here, we have addressed whether Rac1 activation would enhance carcinogenesis and result in tumor progression. We used K14ΔNLef1 mice, a model for differentiated sebaceous adenomas (SAs), and activated Rac1 in an epidermis-specific manner (K14L61Rac1). Surprisingly, Rac1 activation did not change the incidence and frequency of sebaceous tumors. However, tumors, which occurred exclusively in K14ΔNLef1/K14L61Rac1 double-transgenic mice, were poorly differentiated resembling malignant sebaceous tumors and were termed sebaceous carcinoma-like tumors (SCLTs). Compared with SAs, SCLTs showed an aberrant pattern of cell proliferation, invasive growth and less abundant expression of sebocyte differentiation markers, including stearoyl-CoA desaturase-1 and adipophilin. Interestingly, the adnexal SC marker Lrig1 was upregulated in SCLTs, showing that active Rac1 leads to the accumulation of sebocyte precursors in the context of K14ΔNLef1-induced skin tumors. In a search for targets of Rac1, we found cancer progression-related proteins, Dhcr24/Seladin1 and Nuclear protein 1/P8, to be strongly regulated in SCLTs. At last, Rac1 and Dhcr24/Seladin1 were detected in human sebaceous tumors demonstrating a potential high impact of our findings for human skin disease. This is the first study showing that Rac1 activity can lead to malignant progression of skin tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- DMBA:
-
7,12-Dimethylbenz[a]anthracen
- GEF:
-
guanine nucleotide exchange factor
- HF:
-
hair follicle
- IFE:
-
interfollicular epidermis
- K10:
-
keratin 10
- K14:
-
keratin 14
- SC:
-
stem cell
- SCD-1:
-
stearoyl-CoA desaturase-1
- SA:
-
sebaceous adenoma
- SCLT:
-
sebaceous carcinoma-like tumor
- SG:
-
sebaceous gland.
References
Malliri A, van der Kammen R A, Clark K, van der Valk M, Michiels F, Collard J G . Mice deficient in the Rac activator Tiam1 are resistant to Ras-induced skin tumours. Nature 2002; 417: 867–871.
Karlsson R, Pedersen E D, Wang Z, Brakebusch C . Rho GTPase function in tumorigenesis. Biochim Biophys Acta 2009; 1796: 91–98.
Wang Z, Pedersen E, Basse A, Lefever T, Peyrollier K, Kapoor S et al. Rac1 is crucial for Ras-dependent skin tumor formation by controlling Pak1-Mek-Erk hyperactivation and hyperproliferation in vivo. Oncogene 2010; 29: 3362–3373.
Chrostek A, Wu X, Quondamatteo F, Hu R, Sanecka A, Niemann C et al. Rac1 is crucial for hair follicle integrity but is not essential for maintenance of the epidermis. Mol Cell Biol 2006; 26: 6957–6970.
Castilho R M, Squarize C H, Patel V, Millar S E, Zheng Y, Molinolo A et al. Requirement of Rac1 distinguishes follicular from interfollicular epithelial stem cells. Oncogene 2007; 26: 5078–5085.
Benitah S A, Frye M, Glogauer M, Watt F M . Stem cell depletion through epidermal deletion of Rac1. Science 2005; 309: 933–935.
Stachelscheid H, Ibrahim H, Koch L, Schmitz A, Tscharntke M, Wunderlich F T et al. Epidermal insulin/IGF-1 signalling control interfollicular morphogenesis and proliferative potential through Rac activation. EMBO J 2008; 27: 2091–2101.
Niemann C, Owens D M, Hülsken J, Birchmeier W, Watt F. M . Expression of DeltaNLef1 in mouse epidermis results in differentiation of hair follicles into squamous epidermal cysts and formation of skin tumours. Development 2002; 129: 95–109.
Takeda H, Lyle S, Lazar A J F, Zouboulis C C, Smyth I, Watt F. M . Human sebaceous tumors harbor inactivating mutations in LEF1. Nat Med 2006; 12: 395–397.
Niemann C, Owens D M, Schettina P, Watt F M . Dual role of inactivating Lef1 mutations in epidermis: tumor promotion and specification of tumor type. Cancer Res 2007; 67: 2916–2921.
Behrendt K, Klatte J, Pofahl R, Bloch W, Smyth N, Tscharntke M et al. A function for Rac1 in the terminal differentiation and pigmentation of hair. J Cell Sci 2012; 125: 896–905.
Fehrenschild D, Galli U, Breiden B, Bloch W, Schettina P, Brodesser S et al. TCF/Lef1-mediated control of lipid metabolism regulates skin barrier function. J Investig Dermatol 2012; 132: 337–345.
Kakanj P, Reuter K, Séquaris G, Wodtke C, Schettina P, Frances D et al. Indian hedgehog controls proliferation and differentiation in skin tumorigenesis and protects against malignant progression. Cell Rep 2013; 4: 340–351.
Calonje J E, Lazar AJ, McKee PH, B T . McKees Pathology of the Skin, 4th edn. Saunders Elsevier (USA, Philadelphia): China, 2011.
Gu L-H, Coulombe P A . Hedgehog signaling, keratin 6 induction, and sebaceous gland morphogenesis: implications for pachyonychia congenita and related conditions. Am J Pathol 2008; 173: 752–761.
Olson M F, Ashworth A, Hall A . An essential role for Rho, Rac, and Cdc42 GTPases in cell cycle progression through G1. Science 1995; 269: 1270–1272.
Kamai T, Shirataki H, Nakanishi K, Furuya N, Kambara T, Abe H et al. Increased Rac1 activity and Pak1 overexpression are associated with lymphovascular invasion and lymph node metastasis of upper urinary tract cancer. BMC Cancer 2010; 10: 164.
Jaks V, Kasper M, Toftgård R . The hair follicle-a stem cell zoo. Exp Cell Res 2010; 316: 1422–1428.
Battista M-C, Guimond M-O, Roberge C, Doueik A A, Fazli L, Gleave M et al. Inhibition of DHCR24/seladin-1 impairs cellular homeostasis in prostate cancer. Prostate 2010; 70: 921–933.
Goruppi S, Iovanna J L . Stress-inducible protein p8 is involved in several physiological and pathological processes. J Biol Chem 2010; 285: 1577–1581.
Ree A H, Tvermyr M, Engebraaten O, Rooman M, Røsok O, Hovig E et al. Expression of a novel factor in human breast cancer cells with metastatic potential. Cancer Res 1999; 59: 4675–4680.
Jiang W G, Davies G, Martin T A, Kynaston H, Mason M D, Fodstad O . Com-1/p8 acts as a putative tumour suppressor in prostate cancer. Int J Mol Med 2006; 18: 981–986.
Hartmann C H, Klein C A . Gene expression profiling of single cells on large-scale oligonucleotide arrays. Nucleic Acids Res 2006; 34: e143.
Smyth G K, Speed T . Normalization of cDNA microarray data. Methods 2003; 31: 265–273.
Scheid S, Spang R . twilight; a Bioconductor package for estimating the local false discovery rate. Bioinformatics 2005; 21: 2921–2922.
Storey J D, Tibshirani R . Statistical significance for genomewide studies. Proc Natl Acad Sci USA 2003; 100: 9440–9445.
Acknowledgements
We thank Vera Czichowski, Dagmar Fehrenschild, Peter Schettina, Margot Junker and Michael Tscharntke for expert technical support and are grateful for technical support by Wilhelm Bloch and Cornelia Mauch (SFB 829, Z2). This work was funded by the Deutsche Krebshilfe (to CN and CK) and the SFB 829 (A3 to CN, B10 to IH).
Author Contributions
CN and IH designed the experiments. DF, NS, RP, MM, KB, KR and CN performed the experiments and data analysis. CK established and supervised rare cell microarray analysis. TK discussed the data, and IH and CN wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Frances, D., Sharma, N., Pofahl, R. et al. A role for Rac1 activity in malignant progression of sebaceous skin tumors. Oncogene 34, 5505–5512 (2015). https://doi.org/10.1038/onc.2014.471
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.471
This article is cited by
-
The E3 ubiquitin ligase MG53 inhibits hepatocellular carcinoma by targeting RAC1 signaling
Oncogenesis (2022)
-
Rho GTPases: Anti- or pro-neoplastic targets?
Oncogene (2017)