Abstract
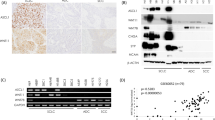
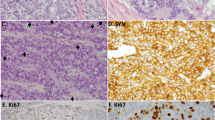
ASCL1 is an important regulatory transcription factor in pulmonary neuroendocrine (NE) cell development, but its value as a biomarker of NE differentiation in lung adenocarcinoma (AD) and as a potential prognostic biomarker remains unclear. We examined ASCL1 expression in lung cancer samples of varied histologic subtype, clinical outcome and smoking status and compared with expression of traditional NE markers. ASCL1 mRNA expression was found almost exclusively in smokers with AD, in contrast to non-smokers and other lung cancer subtypes. ASCL1 protein expression by immunohistochemical (IHC) analysis correlated best with synaptophysin compared with chromogranin and CD56/NCAM. Analysis of a compendium of 367 microarray-based gene expression profiles in stage I lung adenocarcinomas identified significantly higher expression levels of the RET oncogene in ASCL1-positive tumors (ASCL1+) compared with ASCL1− tumors (q-value <10−9). High levels of RET expression in ASCL1+ but not in ASCL1- tumors was associated with significantly shorter overall survival (OS) in stage 1 (P=0.007) and in all AD (P=0.037). RET protein expression by IHC had an association with OS in the context of ASCL1 expression. In silico gene set analysis and in vitro experiments by ASCL1 shRNA in AD cells with high endogenous expression of ASCL1 and RET implicated ASCL1 as a potential upstream regulator of the RET oncogene. Also, silencing ASCL1 in AD cells markedly reduced cell growth and motility. These results suggest that ASCL1 and RET expression defines a clinically relevant subgroup of ∼10% of AD characterized by NE differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kageyama R, Nakanishi S . Helix-loop-helix factors in growth and differentiation of the vertebrate nervous system. Curr Opin Genet Dev 1997; 7: 659–665.
Borges M, Linnoila RI, van de Velde HJ, Chen H, Nelkin BD, Mabry M et al. An achaete-scute homologue essential for neuroendocrine differentiation in the lung. Nature 1997; 386: 852–855.
Linnoila RI, Zhao B, DeMayo JL, Nelkin BD, Baylin SB, DeMayo FJ et al. Constitutive achaete-scute homologue-1 promotes airway dysplasia and lung neuroendocrine tumors in transgenic mice. Cancer Res 2000; 60: 4005–4009.
Bryant CM, Albertus DL, Kim S, Chen G, Brambilla C, Guedj M et al. Clinically relevant characterization of lung adenocarcinoma subtypes based on cellular pathways: an international validation study. PLoS ONE 2010; 5: e11712.
Storey JD, Tibshirani R . Statistical significance for genomewide studies. Proc Natl Acad Sci USA 2003; 100: 9440–9445.
Osada H, Tomida S, Yatabe Y, Tatematsu Y, Takeuchi T, Murakami H et al. Roles of achaete-scute homologue 1 in DKK1 and E-cadherin repression and neuroendocrine differentiation in lung cancer. Cancer Res 2008; 68: 1647–1655.
Kang HC, Kim IJ, Park JH, Shin Y, Ku JL, Jung MS et al. Identification of genes with differential expression in acquired drug-resistant gastric cancer cells using high-density oligonucleotide microarrays. Clin Cancer Res 2004; 10 (1 Pt 1): 272–284.
Arighi E, Borrello MG, Sariola H . RET tyrosine kinase signaling in development and cancer. Cytokine Growth Factor Rev 2005; 16: 441–467.
Ionescu DN, Treaba D, Gilks CB, Leung S, Renouf D, Laskin J et al. Nonsmall cell lung carcinoma with neuroendocrine differentiation-an entity of no clinical or prognostic significance. Am J Surg Pathol 2007; 31: 26–32.
Fujiwara T, Hiramatsu M, Isagawa T, Ninomiya H, Inamura K, Ishikawa S et al. ASCL1-coexpression profiling but not single gene expression profiling defines lung adenocarcinomas of neuroendocrine nature with poor prognosis. Lung Cancer 2012; 75: 119–125.
Dauger S, Renolleau S, Vardon G, Nepote V, Mas C, Simonneau M et al. Ventilatory responses to hypercapnia and hypoxia in Mash-1 heterozygous newborn and adult mice. Pediatr Res 1999; 46: 535–542.
Burton MD, Kawashima A, Brayer JA, Kazemi H, Shannon DC, Schuchardt A et al. RET proto-oncogene is important for the development of respiratory CO2 sensitivity. J Auton Nerv Syst 1997; 63: 137–143.
Dauger S, Guimiot F, Renolleau S, Levacher B, Boda B, Mas C et al. MASH-1/RET pathway involvement in development of brain stem control of respiratory frequency in newborn mice. Physiol Genomics 2001; 7: 149–157.
Huber K, Bruhl B, Guillemot F, Olson EN, Ernsberger U, Unsicker K . Development of chromaffin cells depends on MASH1 function. Development 2002; 129: 4729–4738.
Shoba T, Dheen ST, Tay SS . Retinoic acid influences the expression of the neuronal regulatory genes Mash-1 and c-ret in the developing rat heart. Neurosci Lett 2002; 318: 129–132.
Lodish MB, Stratakis CA . RET oncogene in MEN2, MEN2B, MTC and other forms of thyroid cancer. Expert Rev Anticancer Ther 2008; 8: 625–632.
Marsh DJ, Gimm O . Multiple endocrine neoplasia: types 1 and 2. Adv Otorhinolaryngol 2011; 70: 84–90.
Kouvaraki MA, Shapiro SE, Perrier ND, Cote GJ, Gagel RF, Hoff AO et al. RET proto-oncogene: a review and update of genotype-phenotype correlations in hereditary medullary thyroid cancer and associated endocrine tumors. Thyroid 2005; 15: 531–544.
Rheinbay E, Suva ML, Gillespie SM, Wakimoto H, Patel AP, Shahid M et al. An aberrant transcription factor network essential for wnt signaling and stem cell maintenance in glioblastoma. Cell Rep 2013; 3: 1567–1579.
Lo L, Tiveron MC, Anderson DJ . MASH1 activates expression of the paired homeodomain transcription factor Phox2a, and couples pan-neuronal and subtype-specific components of autonomic neuronal identity. Development 1998; 125: 609–620.
Chen H, Carson-Walter EB, Baylin SB, Nelkin BD, Ball DW . Differentiation of medullary thyroid cancer by C-Raf-1 silences expression of the neural transcription factor human achaete-scute homolog-1. Surgery 1996; 120: 168–172 discussion 73.
Huber K, Karch N, Ernsberger U, Goridis C, Unsicker K . The role of Phox2B in chromaffin cell development. Dev Biol 2005; 279: 501–508.
Kohno T, Ichikawa H, Totoki Y, Yasuda K, Hiramoto M, Nammo T et al. KIF5B-RET fusions in lung adenocarcinoma. Nat Med 2012; 18: 375–377.
Wang R, Hu H, Pan Y, Li Y, Ye T, Li C et al. RET fusions define a unique molecular and clinicopathologic subtype of non-small-cell lung cancer. J Clin Oncol 2012; 30: 4352–4359.
Freedman ND, Leitzmann MF, Hollenbeck AR, Schatzkin A, Abnet CC . Cigarette smoking and subsequent risk of lung cancer in men and women: analysis of a prospective cohort study. Lancet Oncol 2008; 9: 649–656.
Khuder SA . Effect of cigarette smoking on major histological types of lung cancer: a meta-analysis. Lung Cancer 2001; 31: 139–148.
Landi MT, Dracheva T, Rotunno M, Figueroa JD, Liu H, Dasgupta A et al. Gene expression signature of cigarette smoking and its role in lung adenocarcinoma development and survival. PLoS ONE 2008; 3: e1651.
Massion PP, Zou Y, Chen H, Jiang A, Coulson P, Amos CI et al. Smoking-related genomic signatures in non-small cell lung cancer. Am J Respir Crit Care Med 2008; 178: 1164–1172.
Powell CA, Spira A, Derti A, DeLisi C, Liu G, Borczuk A et al. Gene expression in lung adenocarcinomas of smokers and nonsmokers. Am J Respir Cell Mol Biol 2003; 29: 157–162.
Boldrini L, Ali G, Gisfredi S, Ursino S, Baldini E, Melfi F et al. Epidermal growth factor receptor and K-RAS mutations in 411 lung adenocarcinoma: a population-based prospective study. Oncol Rep 2009; 22: 683–691.
Shaw AT, Yeap BY, Mino-Kenudson M, Digumarthy SR, Costa DB, Heist RS et al. Clinical features and outcome of patients with non-small-cell lung cancer who harbor EML4-ALK. J Clin Oncol 2009; 27: 4247–4253.
Takahashi T, Sonobe M, Kobayashi M, Yoshizawa A, Menju T, Nakayama E et al. Clinicopathologic features of non-small-cell lung cancer with EML4-ALK fusion gene. Ann Surg Oncol 2010; 17: 889–897.
Osada H, Tatematsu Y, Yatabe Y, Horio Y, Takahashi T . ASH1 gene is a specific therapeutic target for lung cancers with neuroendocrine features. Cancer Res 2005; 65: 10680–10685.
Poulsen TT, Pedersen N, Juel H, Poulsen HS . A chimeric fusion of the hASH1 and EZH2 promoters mediates high and specific reporter and suicide gene expression and cytotoxicity in small cell lung cancer cells. Cancer Gene Ther 2008; 15: 563–575.
Travis WD, World Health OrganizationInternational Agency for Research on Cancer., International Association for the Study of Lung Cancer., International Academy of Pathology. Pathology and Genetics of Tumours of The Lung, Pleura, Thymus and Heart. Lyon Oxford: IARC Press Oxford University Press (distributor), 2004.
Acknowledgements
This work was supported by a Waterman Biomarker Discovery grant and by the Mayo Clinic Center for Individualized Medicine.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Kosari, F., Ida, C., Aubry, MC. et al. ASCL1 and RET expression defines a clinically relevant subgroup of lung adenocarcinoma characterized by neuroendocrine differentiation. Oncogene 33, 3776–3783 (2014). https://doi.org/10.1038/onc.2013.359
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.359
Keywords
This article is cited by
-
Aggressive variants of prostate cancer: underlying mechanisms of neuroendocrine transdifferentiation
Journal of Experimental & Clinical Cancer Research (2022)
-
Pulmonary large cell neuroendocrine carcinoma with adenocarcinoma-like features: napsin A expression and genomic alterations
Modern Pathology (2018)
-
EZH2 promotes progression of small cell lung cancer by suppressing the TGF-β-Smad-ASCL1 pathway
Cell Discovery (2015)