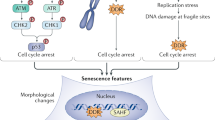
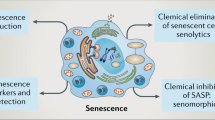
Abstract
p53 functions as a transcription factor involved in cell-cycle control, DNA repair, apoptosis and cellular stress responses. However, besides inducing cell growth arrest and apoptosis, p53 activation also modulates cellular senescence and organismal aging. Senescence is an irreversible cell-cycle arrest that has a crucial role both in aging and as a robust physiological antitumor response, which counteracts oncogenic insults. Therefore, via the regulation of senescence, p53 contributes to tumor growth suppression, in a manner strictly dependent by its expression and cellular context. In this review, we focus on the recent advances on the contribution of p53 to cellular senescence and its implication for cancer therapy, and we will discuss p53’s impact on animal lifespan. Moreover, we describe p53-mediated regulation of several physiological pathways that could mediate its role in both senescence and aging.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- DDR:
-
DNA damage response
- ROS:
-
reactive oxygen species.
References
Sherwood SW, Rush D, Ellsworth JL, Schimke RT . Defining cellular senescence in IMR-90 cells: a flow cytometric analysis. Proc Natl Acad Sci USA 1988; 85: 9086–9090.
Kuilman T, Michaloglou C, Mooi WJ, Peeper DS . The essence of senescence. Genes Dev 2010; 24: 2463–2479.
Romagosa C, Simonetti S, Lopez-Vicente L, Mazo A, Lleonart ME, Castellvi J et al. p16(Ink4a) overexpression in cancer: a tumor suppressor gene associated with senescence and high-grade tumors. Oncogene 2011; 30: 2087–2097.
Alessio N, Squillaro T, Cipollaro M, Bagella L, Giordano A, Galderisi U . The BRG1 ATPase of chromatin remodeling complexes is involved in modulation of mesenchymal stem cell senescence through RB-P53 pathways. Oncogene 2010; 29: 5452–5463.
Collado M, Gil J, Efeyan A, Guerra C, Schuhmacher AJ, Barradas M et al. Tumour biology: senescence in premalignant tumours. Nature 2005; 436: 642.
Krishnamurthy J, Torrice C, Ramsey MR, Kovalev GI, Al-Regaiey K, Su L et al. Ink4a/Arf expression is a biomarker of aging. J Clin Invest 2004; 114: 1299–1307.
Liu Y, Sanoff HK, Cho H, Burd CE, Torrice C, Ibrahim JG et al. Expression of p16(INK4a) in peripheral blood T-cells is a biomarker of human aging. Aging cell 2009; 8: 439–448.
Sharpless NE . Ink4a/Arf links senescence and aging. Exp Gerontol 2004; 39: 1751–1759.
Nogueira L, Ruiz-Ontanon P, Vazquez-Barquero A, Lafarga M, Berciano MT, Aldaz B et al. Blockade of the NFkappaB pathway drives differentiating glioblastoma-initiating cells into senescence both in vitro and in vivo. Oncogene 2011; 30: 3537–3548.
Caldwell ME, DeNicola GM, Martins CP, Jacobetz MA, Maitra A, Hruban RH et al. Cellular features of senescence during the evolution of human and murine ductal pancreatic cancer. Oncogene 2012; 31: 1599–1608.
Baker DJ, Wijshake T, Tchkonia T, LeBrasseur NK, Childs BG, van de Sluis B et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 2011; 479: 232–236.
Assmus B, Urbich C, Aicher A, Hofmann WK, Haendeler J, Rossig L et al. HMG-CoA reductase inhibitors reduce senescence and increase proliferation of endothelial progenitor cells via regulation of cell cycle regulatory genes. Circ Res 2003; 92: 1049–1055.
Beausejour CM, Krtolica A, Galimi F, Narita M, Lowe SW, Yaswen P et al. Reversal of human cellular senescence: roles of the p53 and p16 pathways. Embo J 2003; 22: 4212–4222.
Demidenko ZN, Korotchkina LG, Gudkov AV, Blagosklonny MV . Paradoxical suppression of cellular senescence by p53. Proc Natl Acad Sci USA 2010; 107: 9660–9664.
Kumar M, Lu Z, Takwi AA, Chen W, Callander NS, Ramos KS et al. Negative regulation of the tumor suppressor p53 gene by microRNAs. Oncogene 2011; 30: 843–853.
Campisi J . Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell 2005; 120: 513–522.
Campisi J, d'Adda di Fagagna F . Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol 2007; 8: 729–740.
Hayflick L . The limited in vitro lifetime of human diploid cell strains. Exp Cell Res 1965; 37: 614–636.
Wang X, Zeng L, Wang J, Chau JF, Lai KP, Jia D et al. A positive role for c-Abl in Atm and Atr activation in DNA damage response. Cell Death Differ 2011; 18: 5–15.
Lossaint G, Besnard E, Fisher D, Piette J, Dulic V . Chk1 is dispensable for G2 arrest in response to sustained DNA damage when the ATM/p53/p21 pathway is functional. Oncogene 2011; 30: 4261–4274.
Bodnar AG, Ouellette M, Frolkis M, Holt SE, Chiu CP, Morin GB et al. Extension of life-span by introduction of telomerase into normal human cells. Science 1998; 279: 349–352.
Magenta A, Cencioni C, Fasanaro P, Zaccagnini G, Greco S, Sarra-Ferraris G et al. miR-200c is upregulated by oxidative stress and induces endothelial cell apoptosis and senescence via ZEB1 inhibition. Cell Death Differ 2011; 18: 1628–1639.
Atzmon G, Cho M, Cawthon RM, Budagov T, Katz M, Yang X et al. Evolution in health and medicine Sackler colloquium: genetic variation in human telomerase is associated with telomere length in Ashkenazi centenarians. Proc Natl Acad Sci USA 2010; 107 (Suppl 1): 1710–1717.
Rudolph KL, Chang S, Lee HW, Blasco M, Gottlieb GJ, Greider C et al. Longevity, stress response, and cancer in aging telomerase-deficient mice. Cell 1999; 96: 701–712.
Lee HW, Blasco MA, Gottlieb GJ, Horner JW, Greider CW, DePinho RA . Essential role of mouse telomerase in highly proliferative organs. Nature 1998; 392: 569–574.
Flores I, Cayuela ML, Blasco MA . Effects of telomerase and telomere length on epidermal stem cell behavior. Science 2005; 309: 1253–1256.
Hande MP, Samper E, Lansdorp P, Blasco MA . Telomere length dynamics and chromosomal instability in cells derived from telomerase null mice. J Cell Biol 1999; 144: 589–601.
Chin L, Artandi SE, Shen Q, Tam A, Lee SL, Gottlieb GJ et al. p53 deficiency rescues the adverse effects of telomere loss and cooperates with telomere dysfunction to accelerate carcinogenesis. Cell 1999; 97: 527–538.
Di Micco R, Fumagalli M, Cicalese A, Piccinin S, Gasparini P, Luise C et al. Oncogene-induced senescence is a DNA damage response triggered by DNA hyper-replication. Nature 2006; 444: 638–642.
Rai P, Young JJ, Burton DG, Giribaldi MG, Onder TT, Weinberg RA . Enhanced elimination of oxidized guanine nucleotides inhibits oncogenic RAS-induced DNA damage and premature senescence. Oncogene 2011; 30: 1489–1496.
Weyemi U, Lagente-Chevallier O, Boufraqech M, Prenois F, Courtin F, Caillou B et al. ROS-generating NADPH oxidase NOX4 is a critical mediator in oncogenic H-Ras-induced DNA damage and subsequent senescence. Oncogene 2012; 31: 1117–1129.
Jenkins NC, Liu T, Cassidy P, Leachman SA, Boucher KM, Goodson AG et al. The p16(INK4A) tumor suppressor regulates cellular oxidative stress. Oncogene 2011; 30: 265–274.
Parrinello S, Samper E, Krtolica A, Goldstein J, Melov S, Campisi J . Oxygen sensitivity severely limits the replicative lifespan of murine fibroblasts. Nat Cell Biol 2003; 5: 741–747.
Rufini A, Niklison-Chirou MV, Inoue S, Tomasini R, Harris IS, Marino A et al. TAp73 depletion accelerates aging through metabolic dysregulation. Genes Dev 2012; 26: 2009–2014.
Balaban RS, Nemoto S, Finkel T . Mitochondria, oxidants, and aging. Cell 2005; 120: 483–495.
Harman D . Aging: a theory based on free radical and radiation chemistry. J Gerontol 1956; 11: 298–300.
Harman D . The biologic clock: the mitochondria? J Am Geriatr Soc 1972; 20: 145–147.
Gough DR, Cotter TG . Hydrogen peroxide: a Jekyll and Hyde signalling molecule. Cell Death Dis 2011; 2: e213.
Thomas E, Zeps N, Rigby P, Hartmann P . Reactive oxygen species initiate luminal but not basal cell death in cultured human mammary alveolar structures: a potential regulator of involution. Cell Death Dis 2011; 2: e189.
Lemarie A, Huc L, Pazarentzos E, Mahul-Mellier AL, Grimm S . Specific disintegration of complex II succinate:ubiquinone oxidoreductase links pH changes to oxidative stress for apoptosis induction. Cell Death Differ 2011; 18: 338–349.
Lambert AJ, Boysen HM, Buckingham JA, Yang T, Podlutsky A, Austad SN et al. Low rates of hydrogen peroxide production by isolated heart mitochondria associate with long maximum lifespan in vertebrate homeotherms. Aging cell 2007; 6: 607–618.
Lu T, Finkel T . Free radicals and senescence. Exp Cell Res 2008; 314: 1918–1922.
Halliwell B . The wanderings of a free radical. Free Radic Biol Med 2009; 46: 531–542.
Tiede LM, Cook EA, Morsey B, Fox HS . Oxygen matters: tissue culture oxygen levels affect mitochondrial function and structure as well as responses to HIV viroproteins. Cell Death Dis 2011; 2: e246.
Shalini S, Dorstyn L, Wilson C, Puccini J, Ho L, Kumar S . Impaired antioxidant defence and accumulation of oxidative stress in caspase-2-deficient mice. Cell Death Differ 2012; 19: 1370–1380.
Moiseeva O, Bourdeau V, Roux A, Deschenes-Simard X, Ferbeyre G . Mitochondrial dysfunction contributes to oncogene-induced senescence. Mol Cell Biol 2009; 29: 4495–4507.
Aksoy O, Chicas A, Zeng T, Zhao Z, McCurrach M, Wang X et al. The atypical E2F family member E2F7 couples the p53 and RB pathways during cellular senescence. Genes Dev 2012; 26: 1546–1557.
Jung JH, Bae S, Lee JY, Woo SR, Cha HJ, Yoon Y et al. E3 ubiquitin ligase Hades negatively regulates the exonuclear function of p53. Cell Death Differ 2011; 18: 1865–1875.
Gao W, Shen Z, Shang L, Wang X . Upregulation of human autophagy-initiation kinase ULK1 by tumor suppressor p53 contributes to DNA-damage-induced cell death. Cell Death Differ 2011; 18: 1598–1607.
Toledo F, Wahl GM . Regulating the p53 pathway: in vitro hypotheses, in vivo veritas. Nat Rev Cancer 2006; 6: 909–923.
Sermeus A, Michiels C . Reciprocal influence of the p53 and the hypoxic pathways. Cell Death Dis 2011; 2: e164.
Gogna R, Madan E, Kuppusamy P, Pati U . Re-oxygenation causes hypoxic tumor regression through restoration of p53 wild-type conformation and post-translational modifications. Cell Death Dis 2012; 3: e286.
Osawa T, Davies D, Hartley JA . Mechanism of cell death resulting from DNA interstrand cross-linking in mammalian cells. Cell Death Dis 2011; 2: e187.
Gatta R, Dolfini D, Mantovani R . NF-Y joins E2Fs, p53 and other stress transcription factors at the apoptosis table. Cell Death Dis 2011; 2: e162.
Medema RH, Macurek L . Checkpoint control and cancer. Oncogene 2012; 31: 2601–2613.
Koster R, Timmer-Bosscha H, Bischoff R, Gietema JA, de Jong S . Disruption of the MDM2-p53 interaction strongly potentiates p53-dependent apoptosis in cisplatin-resistant human testicular carcinoma cells via the Fas/FasL pathway. Cell Death Dis 2011; 2: e148.
Ahmed A, Yang J, Maya-Mendoza A, Jackson DA, Ashcroft M . Pharmacological activation of a novel p53-dependent S-phase checkpoint involving CHK-1. Cell Death Dis 2011; 2: e160.
Dixit D, Sharma V, Ghosh S, Mehta VS, Sen E . Inhibition of casein kinase-2 induces p53-dependent cell cycle arrest and sensitizes glioblastoma cells to tumor necrosis factor (TNFalpha)-induced apoptosis through SIRT1 inhibition. Cell Death Dis 2012; 3: e271.
Brown CJ, Lain S, Verma CS, Fersht AR, Lane DP . Awakening guardian angels: drugging the p53 pathway. Nat Rev Cancer 2009; 9: 862–873.
Markert EK, Levine AJ, Vazquez A . Proliferation and tissue remodeling in cancer: the hallmarks revisited. Cell Death Dis 2012; 3: e397.
Wiman KG . Pharmacological reactivation of mutant p53: from protein structure to the cancer patient. Oncogene 2010; 29: 4245–4252.
Kon N, Zhong J, Kobayashi Y, Li M, Szabolcs M, Ludwig T et al. Roles of HAUSP-mediated p53 regulation in central nervous system development. Cell Death Differ 2011; 18: 1366–1375.
Sah VP, Attardi LD, Mulligan GJ, Williams BO, Bronson RT, Jacks T . A subset of p53-deficient embryos exhibit exencephaly. Nat Genet 1995; 10: 175–180.
Levine AJ, Tomasini R, McKeon FD, Mak TW, Melino G . The p53 family: guardians of maternal reproduction. Nat Rev Mol Cell Biol 2011; 12: 259–265.
Gottlieb E, Vousden KH . p53 regulation of metabolic pathways. Cold Spring Harb Perspect Biol 2010; 2: a001040.
Maier B, Gluba W, Bernier B, Turner T, Mohammad K, Guise T et al. Modulation of mammalian life span by the short isoform of p53. Genes Dev 2004; 18: 306–319.
Marcel V, Dichtel-Danjoy ML, Sagne C, Hafsi H, Ma D, Ortiz-Cuaran S et al. Biological functions of p53 isoforms through evolution: lessons from animal and cellular models. Cell Death Differ 2011; 18: 1815–1824.
Kenyon CJ . The genetics of ageing. Nature 2010; 464: 504–512.
Liu D, Ou L, Clemenson GD, Chao C, Lutske ME, Zambetti GP et al. Puma is required for p53-induced depletion of adult stem cells. Nat Cell Biol 2010; 12: 993–998.
Pollina EA, Brunet A . Epigenetic regulation of aging stem cells. Oncogene 2011; 30: 3105–3126.
Donehower LA, Harvey M, Slagle BL, McArthur MJ, Montgomery CA, Butel JS et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 1992; 356: 215–221.
Armata HL, Garlick DS, Sluss HK . The ataxia telangiectasia-mutated target site Ser18 is required for p53-mediated tumor suppression. Cancer Res 2007; 67: 11696–11703.
Spinnler C, Hedstrom E, Li H, de Lange J, Nikulenkov F, Teunisse AF et al. Abrogation of Wip1 expression by RITA-activated p53 potentiates apoptosis induction via activation of ATM and inhibition of HdmX. Cell Death Differ 2011; 18: 1736–1745.
Matheu A, Maraver A, Klatt P, Flores I, Garcia-Cao I, Borras C et al. Delayed ageing through damage protection by the Arf/p53 pathway. Nature 2007; 448: 375–379.
Brooks CL, Gu W . Dynamics in the p53-Mdm2 ubiquitination pathway. Cell Cycle 2004; 3: 895–899.
d'Adda di Fagagna F . Living on a break: cellular senescence as a DNA-damage response. Nat Rev Cancer 2008; 8: 512–522.
Yasaei H, Gilham E, Pickles JC, Roberts TP, O'Donovan M, Newbold RF . Carcinogen-specific mutational and epigenetic alterations in INK4A, INK4B and p53 tumour-suppressor genes drive induced senescence bypass in normal diploid mammalian cells. Oncogene 2012; 32: 171–179.
Afanasyeva EA, Mestdagh P, Kumps C, Vandesompele J, Ehemann V, Theissen J et al. MicroRNA miR-885-5p targets CDK2 and MCM5, activates p53 and inhibits proliferation and survival. Cell Death Differ 2011; 18: 974–984.
Faraonio R, Salerno P, Passaro F, Sedia C, Iaccio A, Bellelli R et al. A set of miRNAs participates in the cellular senescence program in human diploid fibroblasts. Cell Death Differ 2012; 19: 713–721.
Herbig U, Wei W, Dutriaux A, Jobling WA, Sedivy JM . Real-time imaging of transcriptional activation in live cells reveals rapid up-regulation of the cyclin-dependent kinase inhibitor gene CDKN1A in replicative cellular senescence. Aging Cell 2003; 2: 295–304.
Dulic V, Drullinger LF, Lees E, Reed SI, Stein GH . Altered regulation of G1 cyclins in senescent human diploid fibroblasts: accumulation of inactive cyclin E-Cdk2 and cyclin D1-Cdk2 complexes. Proc Natl Acad Sci USA 1993; 90: 11034–11038.
Wang XD, Lapi E, Sullivan A, Ratnayaka I, Goldin R, Hay R et al. SUMO-modified nuclear cyclin D1 bypasses Ras-induced senescence. Cell Death Differ 2011; 18: 304–314.
Lanigan F, Geraghty JG, Bracken AP . Transcriptional regulation of cellular senescence. Oncogene 2011; 30: 2901–2911.
Brugarolas J, Chandrasekaran C, Gordon JI, Beach D, Jacks T, Hannon GJ . Radiation-induced cell cycle arrest compromised by p21 deficiency. Nature 1995; 377: 552–557.
Brown JP, Wei W, Sedivy JM . Bypass of senescence after disruption of p21CIP1/WAF1 gene in normal diploid human fibroblasts. Science 1997; 277: 831–834.
McConnell BB, Starborg M, Brookes S, Peters G . Inhibitors of cyclin-dependent kinases induce features of replicative senescence in early passage human diploid fibroblasts. Curr Biol 1998; 8: 351–354.
Chang BD, Watanabe K, Broude EV, Fang J, Poole JC, Kalinichenko TV et al. Effects of p21Waf1/Cip1/Sdi1 on cellular gene expression: implications for carcinogenesis, senescence, and age-related diseases. Proc Natl Acad Sci USA 2000; 97: 4291–4296.
Vousden KH, Prives C . Blinded by the light: the growing complexity of p53. Cell 2009; 137: 413–431.
Carvajal LA, Hamard PJ, Tonnessen C, Manfredi JJ . E2F7, a novel target, is up-regulated by p53 and mediates DNA damage-dependent transcriptional repression. Genes Dev 2012; 26: 1533–1545.
Di Stefano L, Jensen MR, Helin K . E2F7, a novel E2F featuring DP-independent repression of a subset of E2F-regulated genes. Embo J 2003; 22: 6289–6298.
Logan N, Delavaine L, Graham A, Reilly C, Wilson J, Brummelkamp TR et al. E2F-7: a distinctive E2F family member with an unusual organization of DNA-binding domains. Oncogene 2004; 23: 5138–5150.
Sage J, Miller AL, Perez-Mancera PA, Wysocki JM, Jacks T . Acute mutation of retinoblastoma gene function is sufficient for cell cycle re-entry. Nature 2003; 424: 223–228.
Laplante M, Sabatini DM . mTOR Signaling. Cold Spring Harb Perspect Biol 2012; 4: a011593.
van Veelen W, Korsse SE, van de Laar L, Peppelenbosch MP . The long and winding road to rational treatment of cancer associated with LKB1/AMPK/TSC/mTORC1 signaling. Oncogene 2011; 30: 2289–2303.
Fabrizio P, Pozza F, Pletcher SD, Gendron CM, Longo VD . Regulation of longevity and stress resistance by Sch9 in yeast. Science 2001; 292: 288–290.
Kaeberlein M, Powers RW, Steffen KK, Westman EA, Hu D, Dang N et al. Regulation of yeast replicative life span by TOR and Sch9 in response to nutrients. Science 2005; 310: 1193–1196.
Sharp ZD, Bartke A . Evidence for down-regulation of phosphoinositide 3-kinase/Akt/mammalian target of rapamycin (PI3K/Akt/mTOR)-dependent translation regulatory signaling pathways in Ames dwarf mice. J Gerontol A Biol Sci Med Sci 2005; 60: 293–300.
Powers RW, Kaeberlein M, Caldwell SD, Kennedy BK, Fields S . Extension of chronological life span in yeast by decreased TOR pathway signaling. Genes Dev 2006; 20: 174–184.
Jia K, Chen D, Riddle DL . The TOR pathway interacts with the insulin signaling pathway to regulate C. elegans larval development, metabolism and life span. Development 2004; 131: 3897–3906.
Kapahi P, Zid BM, Harper T, Koslover D, Sapin V, Benzer S . Regulation of lifespan in Drosophila by modulation of genes in the TOR signaling pathway. Curr Biol 2004; 14: 885–890.
Harrison DE, Strong R, Sharp ZD, Nelson JF, Astle CM, Flurkey K et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 2009; 460: 392–395.
Demidenko ZN, Blagosklonny MV . Growth stimulation leads to cellular senescence when the cell cycle is blocked. Cell Cycle 2008; 7: 3355–3361.
Martin-Montalvo A, Villalba JM, Navas P, de Cabo R . NRF2, cancer and calorie restriction. Oncogene 2011; 30: 505–520.
Grandison RC, Piper MD, Partridge L . Amino-acid imbalance explains extension of lifespan by dietary restriction in Drosophila. Nature 2009; 462: 1061–1064.
Zid BM, Rogers AN, Katewa SD, Vargas MA, Kolipinski MC, Lu TA et al. 4E-BP extends lifespan upon dietary restriction by enhancing mitochondrial activity in Drosophila. Cell 2009; 139: 149–160.
Zoncu R, Efeyan A, Sabatini DM . mTOR: from growth signal integration to cancer, diabetes and ageing. Nat Rev Mol Cell Biol 2011; 12: 21–35.
Romeo Y, Moreau J, Zindy PJ, Saba-El-Leil M, Lavoie G, Dandachi F et al. RSK regulates activated BRAF signalling to mTORC1 and promotes melanoma growth. Oncogene (e-pub ahead of print 16 July 2012; doi:10.1038/onc.2012.312).
Sully K, Akinduro O, Philpott MP, Naeem AS, Harwood CA, Reeve VE et al. The mTOR inhibitor rapamycin opposes carcinogenic changes to epidermal Akt1/PKBalpha isoform signaling. Oncogene (e-pub ahead of print 13 August 2012; doi:10.1038/onc.2012.338).
Wen ZH, Su YC, Lai PL, Zhang Y, Xu YF, Zhao A et al. Critical role of arachidonic acid-activated mTOR signaling in breast carcinogenesis and angiogenesis. Oncogene 2012; 32: 160–170.
Xiang T, Jia Y, Sherris D, Li S, Wang H, Lu D et al. Targeting the Akt/mTOR pathway in Brca1-deficient cancers. Oncogene 2011; 30: 2443–2450.
Nardella C, Chen Z, Salmena L, Carracedo A, Alimonti A, Egia A et al. Aberrant Rheb-mediated mTORC1 activation and Pten haploinsufficiency are cooperative oncogenic events. Genes Dev 2008; 22: 2172–2177.
Alimonti A, Nardella C, Chen Z, Clohessy JG, Carracedo A, Trotman LC et al. A novel type of cellular senescence that can be enhanced in mouse models and human tumor xenografts to suppress prostate tumorigenesis. J Clin Invest 2010; 120: 681–693.
Astle MV, Hannan KM, Ng PY, Lee RS, George AJ, Hsu AK et al. AKT induces senescence in human cells via mTORC1 and p53 in the absence of DNA damage: implications for targeting mTOR during malignancy. Oncogene 2012; 31: 1949–1962.
Ruggero D, Montanaro L, Ma L, Xu W, Londei P, Cordon-Cardo C et al. The translation factor eIF-4E promotes tumor formation and cooperates with c-Myc in lymphomagenesis. Nat Med 2004; 10: 484–486.
Demidenko ZN, Zubova SG, Bukreeva EI, Pospelov VA, Pospelova TV, Blagosklonny MV . Rapamycin decelerates cellular senescence. Cell Cycle 2009; 8: 1888–1895.
Young AR, Narita M, Ferreira M, Kirschner K, Sadaie M, Darot JF et al. Autophagy mediates the mitotic senescence transition. Genes Dev 2009; 23: 798–803.
Eisenberg T, Knauer H, Schauer A, Buttner S, Ruckenstuhl C, Carmona-Gutierrez D et al. Induction of autophagy by spermidine promotes longevity. Nat Cell Biol 2009; 11: 1305–1314.
Lepine S, Allegood JC, Park M, Dent P, Milstien S, Spiegel S . Sphingosine-1-phosphate phosphohydrolase-1 regulates ER stress-induced autophagy. Cell Death Differ 2011; 18: 350–361.
Tucci P . Caloric restriction: is mammalian life extension linked to p53? Aging 2012; 4: 525–534.
Budanov AV, Karin M . p53 target genes sestrin1 and sestrin2 connect genotoxic stress and mTOR signaling. Cell 2008; 134: 451–460.
Feng Z, Hu W, de Stanchina E, Teresky AK, Jin S, Lowe S et al. The regulation of AMPK beta1, TSC2, and PTEN expression by p53: stress, cell and tissue specificity, and the role of these gene products in modulating the IGF-1-AKT-mTOR pathways. Cancer Res 2007; 67: 3043–3053.
Lee JJ, Kim BC, Park MJ, Lee YS, Kim YN, Lee BL et al. PTEN status switches cell fate between premature senescence and apoptosis in glioma exposed to ionizing radiation. Cell Death Differ 2011; 18: 666–677.
Leontieva OV, Gudkov AV, Blagosklonny MV . Weak p53 permits senescence during cell cycle arrest. Cell Cycle 2010; 9: 4323–4327.
Klionsky DJ . Autophagy: from phenomenology to molecular understanding in less than a decade. Nat Rev Mol Cell Biol 2007; 8: 931–937.
Rubinsztein DC, Marino G, Kroemer G . Autophagy and aging. Cell 2011; 146: 682–695.
Hofius D, Munch D, Bressendorff S, Mundy J, Petersen M . Role of autophagy in disease resistance and hypersensitive response-associated cell death. Cell Death Differ 2011; 18: 1257–1262.
Wu WK, Coffelt SB, Cho CH, Wang XJ, Lee CW, Chan FK et al. The autophagic paradox in cancer therapy. Oncogene 2012; 31: 939–953.
Lee JH, Budanov AV, Park EJ, Birse R, Kim TE, Perkins GA et al. Sestrin as a feedback inhibitor of TOR that prevents age-related pathologies. Science 2010; 327: 1223–1228.
Matecic M, Smith DL, Pan X, Maqani N, Bekiranov S, Boeke JD et al. A microarray-based genetic screen for yeast chronological aging factors. PLoS Genet 2010; 6: e1000921.
Hars ES, Qi H, Ryazanov AG, Jin S, Cai L, Hu C et al. Autophagy regulates ageing in C. elegans. Autophagy 2007; 3: 93–95.
Minois N, Carmona-Gutierrez D, Bauer MA, Rockenfeller P, Eisenberg T, Brandhorst S et al. Spermidine promotes stress resistance in Drosophila melanogaster through autophagy-dependent and -independent pathways. Cell Death Dis 2012; 3: e401.
Levine B, Kroemer G . Autophagy in aging, disease and death: the true identity of a cell death impostor. Cell Death Differ 2009; 16: 1–2.
Kapahi P, Chen D, Rogers AN, Katewa SD, Li PW, Thomas EL et al. With TOR, less is more: a key role for the conserved nutrient-sensing TOR pathway in aging. Cell Metab 2010; 11: 453–465.
Kimmelman AC . The dynamic nature of autophagy in cancer. Genes Dev 2011; 25: 1999–2010.
Eng CH, Abraham RT . The autophagy conundrum in cancer: influence of tumorigenic metabolic reprogramming. Oncogene 2011; 30: 4687–4696.
Drullion C, Tregoat C, Lagarde V, Tan S, Gioia R, Priault M et al. Apoptosis and autophagy have opposite roles on imatinib-induced K562 leukemia cell senescence. Cell Death Dis 2012; 3: e373.
Degenhardt K, Mathew R, Beaudoin B, Bray K, Anderson D, Chen G et al. Autophagy promotes tumor cell survival and restricts necrosis, inflammation, and tumorigenesis. Cancer Cell 2006; 10: 51–64.
Shi WY, Xiao D, Wang L, Dong LH, Yan ZX, Shen ZX et al. Therapeutic metformin/AMPK activation blocked lymphoma cell growth via inhibition of mTOR pathway and induction of autophagy. Cell Death Dis 2012; 3: e275.
Ajabnoor GM, Crook T, Coley HM . Paclitaxel resistance is associated with switch from apoptotic to autophagic cell death in MCF-7 breast cancer cells. Cell Death Dis 2012; 3: e260.
Kuwahara Y, Oikawa T, Ochiai Y, Roudkenar MH, Fukumoto M, Shimura T et al. Enhancement of autophagy is a potential modality for tumors refractory to radiotherapy. Cell Death Dis 2011; 2: e177.
Galavotti S, Bartesaghi S, Faccenda D, Shaked-Rabi M, Sanzone S, McEvoy A et al. The autophagy-associated factors DRAM1 and p62 regulate cell migration and invasion in glioblastoma stem cells. Oncogene 2012; 32: 699–712.
Marino ML, Fais S, Djavaheri-Mergny M, Villa A, Meschini S, Lozupone F et al. Proton pump inhibition induces autophagy as a survival mechanism following oxidative stress in human melanoma cells. Cell Death Dis 2010; 1: e87.
Bellodi C, Kindle K, Bernassola F, Cossarizza A, Dinsdale D, Melino G et al. A cytoplasmic PML mutant inhibits p53 function. Cell Cycle 2006; 5: 2688–2692.
Yang D, Li L, Liu H, Wu L, Luo Z, Li H et al. Induction of autophagy and senescence by knockdown of ROC1 E3 ubiquitin ligase to suppress the growth of liver cancer cells. Cell Death Differ (e-pub ahead of print 31 August 2012; doi:10.1038/cdd.2012.113).
Gong C, Bauvy C, Tonelli G, Yue W, Delomenie C, Nicolas V et al. Beclin 1 and autophagy are required for the tumorigenicity of breast cancer stem-like/progenitor cells. Oncogene (e-pub ahead of print 25 June 2012; doi:10.1038/onc.2012.252).
Marino G, Salvador-Montoliu N, Fueyo A, Knecht E, Mizushima N, Lopez-Otin C . Tissue-specific autophagy alterations and increased tumorigenesis in mice deficient in Atg4C/autophagin-3. J Biol Chem 2007; 282: 18573–18583.
Qu X, Yu J, Bhagat G, Furuya N, Hibshoosh H, Troxel A et al. Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene. J Clin Invest 2003; 112: 1809–1820.
Takahashi Y, Coppola D, Matsushita N, Cualing HD, Sun M, Sato Y et al. Bif-1 interacts with Beclin 1 through UVRAG and regulates autophagy and tumorigenesis. Nat Cell Biol 2007; 9: 1142–1151.
Tomic T, Botton T, Cerezo M, Robert G, Luciano F, Puissant A et al. Metformin inhibits melanoma development through autophagy and apoptosis mechanisms. Cell Death Dis 2011; 2: e199.
Kang R, Zeh HJ, Lotze MT, Tang D . The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ 2011; 18: 571–580.
Maiuri MC, Galluzzi L, Morselli E, Kepp O, Malik SA, Kroemer G . Autophagy regulation by p53. Curr Opin Cell Biol 2010; 22: 181–185.
Tasdemir E, Maiuri MC, Galluzzi L, Vitale I, Djavaheri-Mergny M, D'Amelio M et al. Regulation of autophagy by cytoplasmic p53. Nat Cell Biol 2008; 10: 676–687.
Liang C . Negative regulation of autophagy. Cell Death Differ 2010; 17: 1807–1815.
Tavernarakis N, Pasparaki A, Tasdemir E, Maiuri MC, Kroemer G . The effects of p53 on whole organism longevity are mediated by autophagy. Autophagy 2008; 4: 870–873.
Kang MY, Kim HB, Piao C, Lee KH, Hyun JW, Chang IY et al. The critical role of catalase in prooxidant and antioxidant function of p53. Cell Death Differ 2012; 20: 117–129.
Polyak K, Xia Y, Zweier JL, Kinzler KW, Vogelstein B . A model for p53-induced apoptosis. Nature 1997; 389: 300–305.
Budanov AV, Sablina AA, Feinstein E, Koonin EV, Chumakov PM . Regeneration of peroxiredoxins by p53-regulated sestrins, homologs of bacterial AhpD. Science 2004; 304: 596–600.
Hussain SP, Amstad P, He P, Robles A, Lupold S, Kaneko I et al. p53-induced up-regulation of MnSOD and GPx but not catalase increases oxidative stress and apoptosis. Cancer Res 2004; 64: 2350–2356.
Tan M, Li S, Swaroop M, Guan K, Oberley LW, Sun Y . Transcriptional activation of the human glutathione peroxidase promoter by p53. J Biol Chem 1999; 274: 12061–12066.
Olovnikov IA, Kravchenko JE, Chumakov PM . Homeostatic functions of the p53 tumor suppressor: regulation of energy metabolism and antioxidant defense. Semin Cancer Biol 2009; 19: 32–41.
Johnson TM, Yu ZX, Ferrans VJ, Lowenstein RA, Finkel T . Reactive oxygen species are downstream mediators of p53-dependent apoptosis. Proc Natl Acad Sci USA 1996; 93: 11848–11852.
Macip S, Igarashi M, Berggren P, Yu J, Lee SW, Aaronson SA . Influence of induced reactive oxygen species in p53-mediated cell fate decisions. Mol Cell Biol 2003; 23: 8576–8585.
Bano D, Zanetti F, Mende Y, Nicotera P . Neurodegenerative processes in Huntington's disease. Cell Death Dis 2011; 2: e228.
Nguyen D, Alavi MV, Kim KY, Kang T, Scott RT, Noh YH et al. A new vicious cycle involving glutamate excitotoxicity, oxidative stress and mitochondrial dynamics. Cell Death Dis 2011; 2: e240.
Nakamura T, Lipton SA . Redox modulation by S-nitrosylation contributes to protein misfolding, mitochondrial dynamics, and neuronal synaptic damage in neurodegenerative diseases. Cell Death Differ 2011; 18: 1478–1486.
Cook CC, Kim A, Terao S, Gotoh A, Higuchi M . Consumption of oxygen: a mitochondrial-generated progression signal of advanced cancer. Cell Death Dis 2012; 3: e258.
Trifunovic A, Larsson NG . Mitochondrial dysfunction as a cause of ageing. J Int Med 2008; 263: 167–178.
Sahin E, Colla S, Liesa M, Moslehi J, Muller FL, Guo M et al. Telomere dysfunction induces metabolic and mitochondrial compromise. Nature 2011; 470: 359–365.
Cunningham JT, Rodgers JT, Arlow DH, Vazquez F, Mootha VK, Puigserver P . mTOR controls mitochondrial oxidative function through a YY1-PGC-1alpha transcriptional complex. Nature 2007; 450: 736–740.
Green DR, Galluzzi L, Kroemer G . Mitochondria and the autophagy-inflammation-cell death axis in organismal aging. Science 2011; 333: 1109–1112.
Ashrafi G, Schwarz TL . The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death Differ 2012; 20: 31–42.
Deng CX . SIRT1, is it a tumor promoter or tumor suppressor? Int J Biol Sci 2009; 5: 147–152.
Luo J, Nikolaev AY, Imai S, Chen D, Su F, Shiloh A et al. Negative control of p53 by Sir2alpha promotes cell survival under stress. Cell 2001; 107: 137–148.
Vaziri H, Dessain SK, Ng Eaton E, Imai SI, Frye RA, Pandita TK et al. hSIR2(SIRT1) functions as an NAD-dependent p53 deacetylase. Cell 2001; 107: 149–159.
Collavin L, Lunardi A, Del Sal G . p53-family proteins and their regulators: hubs and spokes in tumor suppression. Cell Death Differ 2010; 17: 901–911.
Campagna M, Herranz D, Garcia MA, Marcos-Villar L, Gonzalez-Santamaria J, Gallego P et al. SIRT1 stabilizes PML promoting its sumoylation. Cell Death Differ 2011; 18: 72–79.
Pellegrini L, Pucci B, Villanova L, Marino ML, Marfe G, Sansone L et al. SIRT3 protects from hypoxia and staurosporine-mediated cell death by maintaining mitochondrial membrane potential and intracellular pH. Cell Death Differ 2012; 19: 1815–1825.
Feldman JL, Dittenhafer-Reed KE, Denu JM . Sirtuin catalysis and regulation. J Biol Chem 2012; 287: 42419–42427.
Houtkooper RH, Pirinen E, Auwerx J . Sirtuins as regulators of metabolism and healthspan. Nat Rev Mol Cell Biol 2012; 13: 225–238.
Ota H, Akishita M, Eto M, Iijima K, Kaneki M, Ouchi Y . Sirt1 modulates premature senescence-like phenotype in human endothelial cells. J Mol Cell Cardiol 2007; 43: 571–579.
Pearson M, Carbone R, Sebastiani C, Cioce M, Fagioli M, Saito S et al. PML regulates p53 acetylation and premature senescence induced by oncogenic Ras. Nature 2000; 406: 207–210.
Brooks CL, Gu W . How does SIRT1 affect metabolism, senescence and cancer? Nat Rev Cancer 2009; 9: 123–128.
Li L, Wang L, Wang Z, Ho Y, McDonald T, Holyoake TL et al. Activation of p53 by SIRT1 inhibition enhances elimination of CML leukemia stem cells in combination with imatinib. Cancer Cell 2012; 21: 266–281.
Pospelova TV, Demidenko ZN, Bukreeva EI, Pospelov VA, Gudkov AV, Blagosklonny MV . Pseudo-DNA damage response in senescent cells. Cell Cycle 2009; 8: 4112–4118.
Langley E, Pearson M, Faretta M, Bauer UM, Frye RA, Minucci S et al. Human SIR2 deacetylates p53 and antagonizes PML/p53-induced cellular senescence. Embo J 2002; 21: 2383–2396.
Li D, Marchenko ND, Moll UM . SAHA shows preferential cytotoxicity in mutant p53 cancer cells by destabilizing mutant p53 through inhibition of the HDAC6-Hsp90 chaperone axis. Cell Death Differ 2011; 18: 1904–1913.
Krummel KA, Lee CJ, Toledo F, Wahl GM . The C-terminal lysines fine-tune P53 stress responses in a mouse model but are not required for stability control or transactivation. Proc Natl Acad Sci USA 2005; 102: 10188–10193.
Li T, Kon N, Jiang L, Tan M, Ludwig T, Zhao Y et al. Tumor suppression in the absence of p53-mediated cell-cycle arrest, apoptosis, and senescence. Cell 2012; 149: 1269–1283.
Herranz D, Munoz-Martin M, Canamero M, Mulero F, Martinez-Pastor B, Fernandez-Capetillo O et al. Sirt1 improves healthy ageing and protects from metabolic syndrome-associated cancer. Nat Commun 2010; 1: 3.
Oberdoerffer P, Michan S, McVay M, Mostoslavsky R, Vann J, Park SK et al. SIRT1 redistribution on chromatin promotes genomic stability but alters gene expression during aging. Cell 2008; 135: 907–918.
Wang RH, Sengupta K, Li C, Kim HS, Cao L, Xiao C et al. Impaired DNA damage response, genome instability, and tumorigenesis in SIRT1 mutant mice. Cancer Cell 2008; 14: 312–323.
Tissenbaum HA, Guarente L . Increased dosage of a sir-2 gene extends lifespan in Caenorhabditis elegans. Nature 2001; 410: 227–230.
Kaeberlein M, McVey M, Guarente L . The SIR2/3/4 complex and SIR2 alone promote longevity in Saccharomyces cerevisiae by two different mechanisms. Genes Dev 1999; 13: 2570–2580.
Rogina B, Helfand SL . Sir2 mediates longevity in the fly through a pathway related to calorie restriction. Proc Natl Acad Sci USA. 2004; 101: 15998–16003.
Burnett C, Valentini S, Cabreiro F, Goss M, Somogyvari M, Piper MD et al. Absence of effects of Sir2 overexpression on lifespan in C. elegans and Drosophila. Nature 2011; 477: 482–485.
Banks AS, Kon N, Knight C, Matsumoto M, Gutierrez-Juarez R, Rossetti L et al. SirT1 gain of function increases energy efficiency and prevents diabetes in mice. Cell Metab 2008; 8: 333–341.
Iaccarino I, Martins LM . Therapeutic targets in cancer cell metabolism and death. Cell Death Differ 2011; 18: 565–570.
Hanahan D, Weinberg RA . Hallmarks of cancer: the next generation. Cell 2011; 144: 646–674.
Collado M, Serrano M . Senescence in tumours: evidence from mice and humans. Nat Rev Cancer 2010; 10: 51–57.
Demaria M, Misale S, Giorgi C, Miano V, Camporeale A, Campisi J et al. STAT3 can serve as a hit in the process of malignant transformation of primary cells. Cell Death Differ 2012; 19: 1390–1397.
Durinck S, Ho C, Wang NJ, Liao W, Jakkula LR, Collisson EA et al. Temporal dissection of tumorigenesis in primary cancers. Cancer Discov 2011; 1: 137–143.
Wiman KG . Strategies for therapeutic targeting of the p53 pathway in cancer. Cell Death Differ 2006; 13: 921–926.
Ventura A, Kirsch DG, McLaughlin ME, Tuveson DA, Grimm J, Lintault L et al. Restoration of p53 function leads to tumour regression in vivo. Nature 2007; 445: 661–665.
Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V et al. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 2007; 445: 656–660.
Lu WJ, Lee NP, Kaul SC, Lan F, Poon RT, Wadhwa R et al. Mortalin-p53 interaction in cancer cells is stress dependent and constitutes a selective target for cancer therapy. Cell Death Differ 2011; 18: 1046–1056.
Verhaegen M, Checinska A, Riblett MB, Wang S, Soengas MS . E2F1-dependent oncogenic addiction of melanoma cells to MDM2. Oncogene 2012; 31: 828–841.
Vaseva AV, Yallowitz AR, Marchenko ND, Xu S, Moll UM . Blockade of Hsp90 by 17AAG antagonizes MDMX and synergizes with nutlin to induce p53-mediated apoptosis in solid tumors. Cell Death Dis 2011; 2: e156.
Michaelis M, Rothweiler F, Agha B, Barth S, Voges Y, Loschmann N et al. Human neuroblastoma cells with acquired resistance to the p53 activator RITA retain functional p53 and sensitivity to other p53 activating agents. Cell Death Dis 2012; 3: e294.
de Lange J, Ly LV, Lodder K, Verlaan-de Vries M, Teunisse AF, Jager MJ et al. Synergistic growth inhibition based on small-molecule p53 activation as treatment for intraocular melanoma. Oncogene 2012; 31: 1105–1116.
Gamble LD, Kees UR, Tweddle DA, Lunec J . MYCN sensitizes neuroblastoma to the MDM2-p53 antagonists nutlin-3 and MI-63. Oncogene 2012; 31: 752–763.
de Lange J, Verlaan-de Vries M, Teunisse AF, Jochemsen AG . Chk2 mediates RITA-induced apoptosis. Cell Death Differ 2012; 19: 980–989.
Junttila MR, Karnezis AN, Garcia D, Madriles F, Kortlever RM, Rostker F et al. Selective activation of p53-mediated tumour suppression in high-grade tumours. Nature 2010; 468: 567–571.
Feldser DM, Kostova KK, Winslow MM, Taylor SE, Cashman C, Whittaker CA et al. Stage-specific sensitivity to p53 restoration during lung cancer progression. Nature 2010; 468: 572–575.
Heiss EH, Schilder YD, Dirsch VM . Chronic treatment with resveratrol induces redox stress- and ataxia telangiectasia-mutated (ATM)-dependent senescence in p53-positive cancer cells. J Biol Chem 2007; 282: 26759–26766.
Agarwal B, Baur JA . Resveratrol and life extension. Ann N Y Acad Sci 2011; 1215: 138–143.
Feldser DM, Greider CW . Short telomeres limit tumor progression in vivo by inducing senescence. Cancer Cell 2007; 11: 461–469.
Cosme-Blanco W, Shen MF, Lazar AJ, Pathak S, Lozano G, Multani AS et al. Telomere dysfunction suppresses spontaneous tumorigenesis in vivo by initiating p53-dependent cellular senescence. EMBO Rep 2007; 8: 497–503.
Blasco MA, Lee HW, Hande MP, Samper E, Lansdorp PM, DePinho RA et al. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 1997; 91: 25–34.
Morton JP, Timpson P, Karim SA, Ridgway RA, Athineos D, Doyle B et al. Mutant p53 drives metastasis and overcomes growth arrest/senescence in pancreatic cancer. Proc Natl Acad Sci USA 2010; 107: 246–251.
Majumder PK, Grisanzio C, O'Connell F, Barry M, Brito JM, Xu Q et al. A prostatic intraepithelial neoplasia-dependent p27 Kip1 checkpoint induces senescence and inhibits cell proliferation and cancer progression. Cancer Cell 2008; 14: 146–155.
Sun P, Yoshizuka N, New L, Moser BA, Li Y, Liao R et al. PRAK is essential for ras-induced senescence and tumor suppression. Cell 2007; 128: 295–308.
Ha L, Ichikawa T, Anver M, Dickins R, Lowe S, Sharpless NE et al. ARF functions as a melanoma tumor suppressor by inducing p53-independent senescence. Proc Natl Acad Sci USA 2007; 104: 10968–10973.
Kim NW, Piatyszek MA, Prowse KR, Harley CB, West MD, Ho PL et al. Specific association of human telomerase activity with immortal cells and cancer. Science 1994; 266: 2011–2015.
Shay JW, Bacchetti S . A survey of telomerase activity in human cancer. Eur J Cancer 1997; 33: 787–791.
Dotsch V, Bernassola F, Coutandin D, Candi E, Melino G . p63 and p73, the ancestors of p53. Cold Spring Harb Perspect Biol 2010; 2: a004887.
Melino G, De Laurenzi V, Vousden KH . p73: friend or foe in tumorigenesis. Nat Rev Cancer 2002; 2: 605–615.
Gonfloni S, Di Tella L, Caldarola S, Cannata SM, Klinger FG, Di Bartolomeo C et al. Inhibition of the c-Abl-TAp63 pathway protects mouse oocytes from chemotherapy-induced death. Nat Med 2009; 15: 1179–1185.
Agami R, Blandino G, Oren M, Shaul Y . Interaction of c-Abl and p73alpha and their collaboration to induce apoptosis. Nature 1999; 399: 809–813.
Gong JG, Costanzo A, Yang HQ, Melino G, Kaelin WG, Levrero M et al. The tyrosine kinase c-Abl regulates p73 in apoptotic response to cisplatin-induced DNA damage. Nature 1999; 399: 806–809.
Yuan ZM, Shioya H, Ishiko T, Sun X, Gu J, Huang YY et al. p73 is regulated by tyrosine kinase c-Abl in the apoptotic response to DNA damage. Nature 1999; 399: 814–817.
Meltser V, Ben-Yehoyada M, Shaul Y . c-Abl tyrosine kinase in the DNA damage response: cell death and more. Cell Death Differ 2011; 18: 2–4.
Conforti F, Sayan AE, Sreekumar R, Sayan BS . Regulation of p73 activity by post-translational modifications. Cell Death Dis 2012; 3: e285.
Gressner O, Schilling T, Lorenz K, Schulze Schleithoff E, Koch A, Schulze-Bergkamen H et al. TAp63alpha induces apoptosis by activating signaling via death receptors and mitochondria. Embo J 2005; 24: 2458–2471.
Ramadan S, Terrinoni A, Catani MV, Sayan AE, Knight RA, Mueller M et al. p73 induces apoptosis by different mechanisms. Biochem Biophys Res Commun 2005; 331: 713–717.
Rufini A, Agostini M, Grespi F, Tomasini R, Sayan BS, Niklison-Chirou MV et al. p73 in cancer. Genes Cancer 2011; 2: 491–502.
Tomasini R, Secq V, Pouyet L, Thakur AK, Wilhelm M, Nigri J et al. TAp73 is required for macrophage-mediated innate immunity and the resolution of inflammatory responses. Cell Death Differ (e-pub ahead of print 14 September 2012; doi:10.1038/cdd.2012.123).
Yang A, Walker N, Bronson R, Kaghad M, Oosterwegel M, Bonnin J et al. p73-deficient mice have neurological, pheromonal and inflammatory defects but lack spontaneous tumours. Nature 2000; 404: 99–103.
Flores ER . p73 is critical for the persistence of memory. Cell Death Differ 2011; 18: 381–382.
Killick R, Niklison-Chirou M, Tomasini R, Bano D, Rufini A, Grespi F et al. p73: a multifunctional protein in neurobiology. Mol Neurobiol 2011; 43: 139–146.
Talos F, Abraham A, Vaseva AV, Holembowski L, Tsirka SE, Scheel A et al. p73 is an essential regulator of neural stem cell maintenance in embryonal and adult CNS neurogenesis. Cell Death Differ 2010; 17: 1816–1829.
Fujitani M, Cancino GI, Dugani CB, Weaver IC, Gauthier-Fisher A, Paquin A et al. TAp73 acts via the bHLH Hey2 to promote long-term maintenance of neural precursors. Curr Biol 2010; 20: 2058–2065.
Agostini M, Tucci P, Steinert JR, Shalom-Feuerstein R, Rouleau M, Aberdam D et al. microRNA-34a regulates neurite outgrowth, spinal morphology, and function. Proc Natl Acad Sci USA 2011; 108: 21099–21104.
Agostini M, Tucci P, Killick R, Candi E, Sayan BS, Rivetti di Val Cervo P et al. Neuronal differentiation by TAp73 is mediated by microRNA-34a regulation of synaptic protein targets. Proc Natl Acad Sci USA 2011; 108: 21093–21098.
Wilhelm MT, Rufini A, Wetzel MK, Tsuchihara K, Inoue S, Tomasini R et al. Isoform-specific p73 knockout mice reveal a novel role for delta Np73 in the DNA damage response pathway. Genes Dev 2010; 24: 549–560.
Wetzel MK, Naska S, Laliberte CL, Rymar VV, Fujitani M, Biernaskie JA et al. p73 regulates neurodegeneration and phospho-tau accumulation during aging and Alzheimer's disease. Neuron 2008; 59: 708–721.
Tomasini R, Tsuchihara K, Tsuda C, Lau SK, Wilhelm M, Ruffini A et al. TAp73 regulates the spindle assembly checkpoint by modulating BubR1 activity. Proc Natl Acad Sci USA 2009; 106: 797–802.
Tomasini R, Tsuchihara K, Wilhelm M, Fujitani M, Rufini A, Cheung CC et al. TAp73 knockout shows genomic instability with infertility and tumor suppressor functions. Genes Dev 2008; 22: 2677–2691.
Talos F, Nemajerova A, Flores ER, Petrenko O, Moll UM . p73 suppresses polyploidy and aneuploidy in the absence of functional p53. Mol Cell 2007; 27: 647–659.
Candi E, Dinsdale D, Rufini A, Salomoni P, Knight RA, Mueller M et al. TAp63 and DeltaNp63 in cancer and epidermal development. Cell Cycle 2007; 6: 274–285.
Melino G . p63 is a suppressor of tumorigenesis and metastasis interacting with mutant p53. Cell Death Differ 2011; 18: 1487–1499.
Tucci P, Agostini M, Grespi F, Markert EK, Terrinoni A, Vousden KH et al. Loss of p63 and its microRNA-205 target results in enhanced cell migration and metastasis in prostate cancer. Proc Natl Acad Sci USA 2012; 109: 15312–15317.
Lena AM, Shalom-Feuerstein R, Rivetti di Val Cervo P, Aberdam D, Knight RA, Melino G et al. miR-203 represses 'stemness' by repressing DeltaNp63. Cell Death Differ 2008; 15: 1187–1195.
Senoo M, Pinto F, Crum CP, McKeon F . p63 Is essential for the proliferative potential of stem cells in stratified epithelia. Cell 2007; 129: 523–536.
Yi R, Poy MN, Stoffel M, Fuchs E . A skin microRNA promotes differentiation by repressing 'stemness'. Nature 2008; 452: 225–229.
Candi E, Cipollone R, Rivetti di Val Cervo P, Gonfloni S, Melino G, Knight R . p63 in epithelial development. Cell Mol Life Sci 2008; 65: 3126–3133.
Candi E, Rufini A, Terrinoni A, Dinsdale D, Ranalli M, Paradisi A et al. Differential roles of p63 isoforms in epidermal development: selective genetic complementation in p63 null mice. Cell Death Differ 2006; 13: 1037–1047.
Shalom-Feuerstein R, Lena AM, Zhou H, De La Forest Divonne S, Van Bokhoven H, Candi E et al. DeltaNp63 is an ectodermal gatekeeper of epidermal morphogenesis. Cell Death Differ 2011; 18: 887–896.
Leonard MK, Kommagani R, Payal V, Mayo LD, Shamma HN, Kadakia MP . DeltaNp63alpha regulates keratinocyte proliferation by controlling PTEN expression and localization. Cell Death Differ 2011; 18: 1924–1933.
Suh EK, Yang A, Kettenbach A, Bamberger C, Michaelis AH, Zhu Z et al. p63 protects the female germ line during meiotic arrest. Nature 2006; 444: 624–628.
Yang A, Schweitzer R, Sun D, Kaghad M, Walker N, Bronson RT et al. p63 is essential for regenerative proliferation in limb, craniofacial and epithelial development. Nature 1999; 398: 714–718.
Mills AA, Zheng B, Wang XJ, Vogel H, Roop DR, Bradley A . p63 is a p53 homologue required for limb and epidermal morphogenesis. Nature 1999; 398: 708–713.
Flores ER, Sengupta S, Miller JB, Newman JJ, Bronson R, Crowley D et al. Tumor predisposition in mice mutant for p63 and p73: evidence for broader tumor suppressor functions for the p53 family. Cancer Cell 2005; 7: 363–373.
Keyes WM, Wu Y, Vogel H, Guo X, Lowe SW, Mills AA . p63 deficiency activates a program of cellular senescence and leads to accelerated aging. Genes Dev 2005; 19: 1986–1999.
Salomoni P, Dvorkina M, Michod D . Role of the promyelocytic leukaemia protein in cell death regulation. Cell Death Dis 2012; 3: e247.
Peche LY, Scolz M, Ladelfa MF, Monte M, Schneider C . MageA2 restrains cellular senescence by targeting the function of PMLIV/p53 axis at the PML-NBs. Cell Death Differ 2012; 19: 926–936.
Su X, Paris M, Gi YJ, Tsai KY, Cho MS, Lin YL et al. TAp63 prevents premature aging by promoting adult stem cell maintenance. Cell Stem Cell 2009; 5: 64–75.
Paris M, Rouleau M, Puceat M, Aberdam D . Regulation of skin aging and heart development by TAp63. Cell Death Differ 2012; 19: 186–193.
Guo X, Keyes WM, Papazoglu C, Zuber J, Li W, Lowe SW et al. TAp63 induces senescence and suppresses tumorigenesis in vivo. Nat Cell Biol 2009; 11: 1451–1457.
Rivetti di Val Cervo P, Lena AM, Nicoloso M, Rossi S, Mancini M, Zhou H et al. p63-microRNA feedback in keratinocyte senescence. Proc Natl Acad Sci USA 2012; 109: 1133–1138.
Keyes WM, Pecoraro M, Aranda V, Vernersson-Lindahl E, Li W, Vogel H et al. DeltaNp63alpha is an oncogene that targets chromatin remodeler Lsh to drive skin stem cell proliferation and tumorigenesis. Cell stem cell 2011; 8: 164–176.
Zaika A, Irwin M, Sansome C, Moll UM . Oncogenes induce and activate endogenous p73 protein. J Biol Chem 2001; 276: 11310–11316.
Sahin E, Depinho RA . Axis of ageing: telomeres, p53 and mitochondria. Nat Rev Mol Cell Biol 2012; 13: 397–404.
Francis SP, Kramarenko II, Brandon CS, Lee FS, Baker TG, Cunningham LL . Celastrol inhibits aminoglycoside-induced ototoxicity via heat shock protein 32. Cell Death Dis 2011; 2: e195.
Colombi M, Molle KD, Benjamin D, Rattenbacher-Kiser K, Schaefer C, Betz C et al. Genome-wide shRNA screen reveals increased mitochondrial dependence upon mTORC2 addiction. Oncogene 2011; 30: 1551–1565.
Donadelli M, Dando I, Zaniboni T, Costanzo C, Dalla Pozza E, Scupoli MT et al. Gemcitabine/cannabinoid combination triggers autophagy in pancreatic cancer cells through a ROS-mediated mechanism. Cell Death Dis 2011; 2: e152.
Ling LU, Tan KB, Lin H, Chiu GN . The role of reactive oxygen species and autophagy in safingol-induced cell death. Cell Death Dis 2011; 2: e129.
Karna P, Zughaier S, Pannu V, Simmons R, Narayan S, Aneja R . Induction of reactive oxygen species-mediated autophagy by a novel microtubule-modulating agent. J Biol Chem 2010; 285: 18737–18748.
Rodgers JT, Lerin C, Haas W, Gygi SP, Spiegelman BM, Puigserver P . Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1. Nature 2005; 434: 113–118.
Chen YF, Kao CH, Chen YT, Wang CH, Wu CY, Tsai CY et al. Cisd2 deficiency drives premature aging and causes mitochondria-mediated defects in mice. Genes Dev 2009; 23: 1183–1194.
Harman D . The biologic clock: the mitochondria? J Am Geriatr Soc 1972; 20: 145–147.
Lee HY, Choi CS, Birkenfeld AL, Alves TC, Jornayvaz FR, Jurczak MJ et al. Targeted expression of catalase to mitochondria prevents age-associated reductions in mitochondrial function and insulin resistance. Cell Metab 2010; 12: 668–674.
Linnane AW, Marzuki S, Ozawa T, Tanaka M . Mitochondrial DNA mutations as an important contributor to ageing and degenerative diseases. Lancet 1989; 1: 642–645.
Liu J, Cao L, Chen J, Song S, Lee IH, Quijano C et al. Bmi1 regulates mitochondrial function and the DNA damage response pathway. Nature 2009; 459: 387–392.
Reilly SM, Bhargava P, Liu S, Gangl MR, Gorgun C, Nofsinger RR et al. Nuclear receptor corepressor SMRT regulates mitochondrial oxidative metabolism and mediates aging-related metabolic deterioration. Cell Metab 2010; 12: 643–653.
Kang S, Louboutin JP, Datta P, Landel CP, Martinez D, Zervos AS et al. Loss of HtrA2/Omi activity in non-neuronal tissues of adult mice causes premature aging. Cell Death Differ (e-pub ahead of print 14 September 2012; doi:10.1038/cdd.2012.117).
Schriner SE, Linford NJ, Martin GM, Treuting P, Ogburn CE, Emond M et al. Extension of murine life span by overexpression of catalase targeted to mitochondria. Science 2005; 308: 1909–1911.
Su X, Gi YJ, Chakravarti D, Chan LI, Zhang A, Xia X et al. TAp63 is a master transcriptional regulator of lipid and glucose metabolism. Cell Metab 2012; 16: 511–525.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Rufini, A., Tucci, P., Celardo, I. et al. Senescence and aging: the critical roles of p53. Oncogene 32, 5129–5143 (2013). https://doi.org/10.1038/onc.2012.640
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.640
Keywords
This article is cited by
-
Plasma membrane damage limits replicative lifespan in yeast and induces premature senescence in human fibroblasts
Nature Aging (2024)
-
The potential role of hydrogen sulfide in cancer cell apoptosis
Cell Death Discovery (2024)
-
Senolytic treatment fails to improve ovarian reserve or fertility in female mice
GeroScience (2024)
-
TRIM25 inhibition attenuates inflammation, senescence, and oxidative stress in microvascular endothelial cells induced by hyperglycemia
Graefe's Archive for Clinical and Experimental Ophthalmology (2024)
-
LncRNA DANCR counteracts premature ovarian insufficiency by regulating the senescence process of granulosa cells through stabilizing the interaction between p53 and hNRNPC
Journal of Ovarian Research (2023)