Abstract
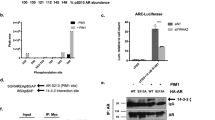
The androgen receptor (AR) has a vital role in the onset and progression of prostate cancer by promoting G1-S progression, possibly by functioning as a licensing factor for DNA replication. We here report that low dose 2-methoxyestradiol (2-ME), an endogenous estrogen metabolite, induces mitotic arrest in prostate cancer cells involving activation of the E3 ligase CHIP (C-terminus of Hsp70-interacting protein) and degradation of the AR. Depletion of the AR by small interfering RNA (siRNA) eliminates 2-ME-induced arrest and introducing AR into PC3-M cells confers 2-ME-induced mitotic arrest. Knockdown of CHIP or MDM2 (mouse homolog of double minute 2 protein) individually or in combination reduced AR degradation and abrogated M phase arrest induced by 2-ME. Our data link AR degradation via ubiquitination to mitotic arrest. Targeting the AR by activating E3 ligases such as CHIP represents a novel strategy for the treatment of prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Jemal A, Murray T, Ward E, Samuels A, Tiwari RC, Ghafoor A et al. Cancer statistics, 2005. CA Cancer J Clin 2005; 55: 10–30.
Schiewer MJ, Augello MA, Knudsen KE . The AR dependent cell cycle: mechanisms and cancer relevance. Mol Cell Endocrinol 2012; 352: 34–45.
Szostak MJ, Kyprianou N . Radiation-induced apoptosis: predictive and therapeutic significance in radiotherapy of prostate cancer (review). Oncol Rep 2000; 7: 699–706.
Lara PN, Meyers FJ . Treatment options in androgen-independent prostate cancer. Cancer Invest 1999; 17: 137–144.
Chen CD, Welsbie DS, Tran C, Baek SH, Chen R, Vessella R et al. Molecular determinants of resistance to antiandrogen therapy. Nat Med 2004; 10: 33–39.
Grossmann ME, Huang H, Tindall DJ . Androgen receptor signaling in androgen-refractory prostate cancer. J Natl Cancer Inst 2001; 93: 1687–1697.
Sharifi N, Farrar WL . Androgen receptor as a therapeutic target for androgen independent prostate cancer. Am J Ther 2006; 13: 166–170.
Pertschuk LP, Macchia RJ, Feldman JG, Brady KA, Levine M, Kim DS et al. Immunocytochemical assay for androgen receptors in prostate cancer: a prospective study of 63 cases with long-term follow-up. Ann Surg Oncol 1994; 1: 495–503.
Prins GS, Sklarew RJ, Pertschuk LP . Image analysis of androgen receptor immunostaining in prostate cancer accurately predicts response to hormonal therapy. J Urol 1998; 159: 641–649.
Fotsis T, Zhang Y, Pepper MS, Adlercreutz H, Montesano R, Nawroth PP et al. The endogenous oestrogen metabolite 2-methoxyoestradiol inhibits angiogenesis and suppresses tumour growth. Nature 1994; 368: 237–239.
Tsukamoto A, Kaneko Y, Yoshida T, Han K, Ichinose M, Kimura S . 2-Methoxyestradiol, an endogenous metabolite of estrogen, enhances apoptosis and beta-galactosidase expression in vascular endothelial cells. Biochem Biophys Res Commun 1998; 248: 9–12.
Yue TL, Wang X, Louden CS, Gupta S, Pillarisetti K, Gu JL et al. 2-Methoxyestradiol, an endogenous estrogen metabolite, induces apoptosis in endothelial cells and inhibits angiogenesis: possible role for stress-activated protein kinase signaling pathway and Fas expression. Mol Pharmacol 1997; 51: 951–962.
Day JM, Newman SP, Comninos A, Solomon C, Purohit A, Leese MP et al. The effects of 2-substituted oestrogen sulphamates on the growth of prostate and ovarian cancer cells. J Steroid Biochem Mol Biol 2003; 84: 317–325.
Kumar AP, Garcia GE, Slaga TJ . 2-methoxyestradiol blocks cell-cycle progression at G(2)/M phase and inhibits growth of human prostate cancer cells. Mol Carcinog 2001; 31: 111–124.
Qadan LR, Perez-Stable CM, Anderson C, D'Ippolito G, Herron A, Howard GA et al. 2-Methoxyestradiol induces G2/M arrest and apoptosis in prostate cancer. Biochem Biophys Res Commun 2001; 285: 1259–1266.
Casarez EV, Dunlap-Brown ME, Conaway MR, Amorino GP . Radiosensitization and modulation of p44/42 mitogen-activated protein kinase by 2-Methoxyestradiol in prostate cancer models. Cancer Res 2007; 67: 8316–8324.
Agarwal ML, Agarwal A, Taylor WR, Stark GR . p53 controls both the G2/M and the G1 cell cycle checkpoints and mediates reversible growth arrest in human fibroblasts. Proc Natl Acad Sci USA 1995; 92: 8493–8497.
Litvinov IV, Vander GDJ, Antony L, Dalrymple S, De Marzo AM, Drake CG et al. Androgen receptor as a licensing factor for DNA replication in androgen-sensitive prostate cancer cells. Proc Natl Acad Sci USA 2006; 103: 15085–15090.
Lee HJ, Chang C . Recent advances in androgen receptor action. Cell Mol Life Sci 2003; 60: 1613–1622.
Lee DK, Chang C . Endocrine mechanisms of disease: expression and degradation of androgen receptor: mechanism and clinical implication. J Clin Endocrinol Metab 2003; 88: 4043–4054.
Huang CY, Beliakoff J, Li X, Lee J, Sharma M, Lim B et al. hZimp7, a novel PIAS-like protein, enhances androgen receptor-mediated transcription and interacts with SWI/SNF-like BAF complexes. Mol Endocrinol 2005; 19: 2915–2929.
Sharma M, Li X, Wang Y, Zarnegar M, Huang CY, Palvimo JJ et al. hZimp10 is an androgen receptor co-activator and forms a complex with SUMO-1 at replication foci. EMBO J 2003; 22: 6101–6114.
D’Antonio JM, Vander GDJ, Isaacs JT . DNA licensing as a novel androgen receptor mediated therapeutic target for prostate cancer. Endocr Relat Cancer 2009; 16: 325–332.
Jin F, Fondell JD . A novel androgen receptor-binding element modulates Cdc6 transcription in prostate cancer cells during cell-cycle progression. Nucleic Acids Res 2009; 37: 4826–4838.
Mallik I, Davila M, Tapia T, Schanen B, Chakrabarti R . Androgen regulates Cdc6 transcription through interactions between androgen receptor and E2F transcription factor in prostate cancer cells. Biochim Biophys Acta 2008; 1783: 1737–1744.
Cushman M, He HM, Katzenellenbogen JA, Lin CM, Hamel E . Synthesis, antitubulin and antimitotic activity, and cytotoxicity of analogs of 2-methoxyestradiol, an endogenous mammalian metabolite of estradiol that inhibits tubulin polymerization by binding to the colchicine binding site. J Med Chem 1995; 38: 2041–2049.
Hamel E, Lin CM, Flynn E, D’Amato RJ . Interactions of 2-methoxyestradiol, an endogenous mammalian metabolite, with unpolymerized tubulin and with tubulin polymers. Biochemistry 1996; 35: 1304–1310.
Wang Q, Li W, Zhang Y, Yuan X, Xu K, Yu J et al. Androgen receptor regulates a distinct transcription program in androgen-independent prostate cancer. Cell 2009; 138: 245–256.
Gaughan L, Logan IR, Neal DE, Robson CN . Regulation of androgen receptor and histone deacetylase 1 by Mdm2-mediated ubiquitylation. Nucleic Acids Res 2005; 33: 13–26.
Lin HK, Wang L, Hu YC, Altuwaijri S, Chang C . Phosphorylation-dependent ubiquitylation and degradation of androgen receptor by Akt require Mdm2 E3 ligase. EMBO J 2002; 21: 4037–4048.
Gao X, Mohsin SK, Gatalica Z, Fu G, Sharma P, Nawaz Z . Decreased expression of e6-associated protein in breast and prostate carcinomas. Endocrinology 2005; 146: 1707–1712.
Cardozo CP, Michaud C, Ost MC, Fliss AE, Yang E, Patterson C et al. C-terminal Hsp-interacting protein slows androgen receptor synthesis and reduces its rate of degradation. Arch Biochem Biophys 2003; 410: 134–140.
He B, Bai S, Hnat AT, Kalman RI, Minges JT, Patterson C et al. An androgen receptor NH2-terminal conserved motif interacts with the COOH terminus of the Hsp70-interacting protein (CHIP). J Biol Chem 2004; 279: 30643–30653.
Wang AM, Morishima Y, Clapp KM, Peng HM, Pratt WB, Gestwicki JE et al. Inhibition of hsp70 by methylene blue affects signaling protein function and ubiquitination and modulates polyglutamine protein degradation. J Biol Chem 2010; 285: 15714–15723.
Morishima Y, Wang AM, Yu Z, Pratt WB, Osawa Y, Lieberman AP . CHIP deletion reveals functional redundancy of E3 ligases in promoting degradation of both signaling proteins and expanded glutamine proteins. Hum Mol Genet 2008; 17: 3942–3952.
Chymkowitch P, Le May N, Charneau P, Compe E, Egly JM . The phosphorylation of the androgen receptor by TFIIH directs the ubiquitin/proteasome process. EMBO J 2011; 30: 468–479.
Acknowledgements
This work was supported, in whole or in part, by the National Institutes of Health grant 5R01CA142823-02 (to J. M. L.) as well as the Charles R. Burnett Jr and W. Griffin Burnett Fund. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Sarkar, S., Brautigan, D., Parsons, S. et al. Androgen receptor degradation by the E3 ligase CHIP modulates mitotic arrest in prostate cancer cells. Oncogene 33, 26–33 (2014). https://doi.org/10.1038/onc.2012.561
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.561
Keywords
This article is cited by
-
A carboxy-terminal ubiquitylation site regulates androgen receptor activity
Communications Biology (2024)
-
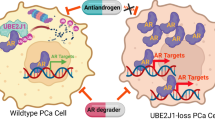
UBE2J1 is the E2 ubiquitin-conjugating enzyme regulating androgen receptor degradation and antiandrogen resistance
Oncogene (2024)
-
RETRACTED ARTICLE: Targeting KDM4A-AS1 represses AR/AR-Vs deubiquitination and enhances enzalutamide response in CRPC
Oncogene (2022)
-
Functional roles of E3 ubiquitin ligases in prostate cancer
Journal of Molecular Medicine (2022)
-
Multifaceted C-terminus of HSP70-interacting protein regulates tumorigenesis via protein quality control
Archives of Pharmacal Research (2019)