Abstract
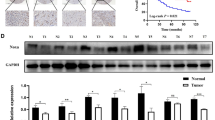
Zinc-finger protein 331 (ZNF331), a Kruppel-associated box zinc-finger protein gene, was identified as a putative tumor suppressor in our previous study. However, the role of ZNF331 in tumorigenesis remains elusive. We aimed to clarify its epigenetic regulation and biological functions in gastric cancer. ZNF331 was silenced or downregulated in 71% (12/17) gastric cancer cell lines. A significant downregulation was also detected in paired gastric tumors compared with adjacent non-cancer tissues. In contrast, ZNF331 was readily expressed in various normal adult tissues. The downregulation of ZNF331 was closely linked to the promoter hypermethylation as evidenced by methylation-specific PCR, bisulfite genomic sequencing and reexpression by demethylation agent treatment. DNA sequencing showed no genetic mutation/deletion of ZNF331 in gastric cancer cell lines. Ectopic expression of ZNF331 in the silenced cancer cell lines MKN28 and HCT116 significantly reduced colony formation and cell viability, induced cell cycle arrests and repressed cell migration and invasive ability. Concordantly, knockdown of ZNF331 increased cell viability and colony formation ability of gastric cancer cell line MKN45. Two-dimensional gel electrophoresis and mass spectrometry-based comparative proteomic approach were applied to analyze the molecular basis of the biological functions of ZNF331. In all, 10 downstream targets of ZNF331 were identified to be associated with regulation of cell growth and metastasis. The tumor-suppressive effect of ZNF331 is mediated at least by downregulation of genes involved in cell growth promotion (DSTN, EIF5A, GARS, DDX5, STAM, UQCRFS1 and SET) and migration/invasion (DSTN and ACTR3), and upregulation of genome-stability gene (SSBP1) and cellular senescence gene (PNPT1). A novel target of ZNF331 (DSTN) was functionally validated. Overexpression of DSTN in BGC-823 cells increased colony formation and migration ability. In conclusion, our results suggest that ZNF331 possesses important functions for the suppression of gastric carcinogenesis as a novel functional tumor-suppressor gene.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Abbreviations
- 2-DE:
-
two-dimensional gel electrophoresis
- 5-Aza:
-
5-Aza-2′-deoxycytidine
- ACTR3:
-
ARP3 actin-related protein 3 homolog
- DDX5:
-
DEAD (Asp-Glu-Ala-Asp) box proteins 5
- DSTN:
-
destrin
- EIF5A:
-
ukaryotic translation initiation factor 5A
- GARS:
-
glycyl-tRNA synthetase
- MSP:
-
methylation specific PCR
- PNPT:
-
the human PNPase gene
- qRT–PCR:
-
quantitative RT–PCR
- RT–PCR:
-
reverse transcript PCR
- SSBP:
-
single-stranded DNA binding protein
- STAM:
-
signal transducing adaptor molecule
- UQCRFS:
-
Ubiquinol-cytochrome-c reductase, Rieske iron-sulfur polypeptide
- ZNF:
-
zinc-finger protein
References
Kang GH, Lee S, Kim JS, Jung HY . Profile of aberrant CpG island methylation along multistep gastric carcinogenesis. Lab Invest 2003; 83: 519–526.
Lee JH, Park SJ, Abraham SC, Seo JS, Nam JH, Choi C et al. Frequent CpG island methylation in precursor lesions and early gastric adenocarcinomas. Oncogene 2004; 23: 4646–4654.
Li L, Tao Q, Jin H, van Hasselt A, Poon FF, Wang X et al. The tumor suppressor UCHL1 forms a complex with p53/MDM2/ARF to promote p53 signaling and is frequently silenced in nasopharyngeal carcinoma. Clin Cancer Res 2010; 16: 2949–2958.
Rippe V, Belge G, Meiboom M, Kazmierczak B, Fusco A, Bullerdiek J . A KRAB zinc finger protein gene is the potential target of 19q13 translocation in benign thyroid tumors. Genes Chromosomes Cancer 1999; 26: 229–236.
Wu H, Zhang S, Qiu W, Zhang G, Xia Q, Xiao C et al. Isolation, characterization, and mapping of a novel human KRAB zinc finger protein encoding gene ZNF463. Biochim Biophys Acta 2001; 1518: 190–193.
Laity JH, Lee BM, Wright PE . Zinc finger proteins: new insights into structural and functional diversity. Curr Opin Struct Biol 2001; 11: 39–46.
Margolin JF, Friedman JR, Meyer WK, Vissing H, Thiesen HJ, Rauscher 3rd FJ . Kruppel-associated boxes are potent transcriptional repression domains. Proc Natl Acad Sci USA 1994; 91: 4509–4513.
Vissing H, Meyer WK, Aagaard L, Tommerup N, Thiesen HJ . Repression of transcriptional activity by heterologous KRAB domains present in zinc finger proteins. FEBS Lett 1995; 369: 153–157.
Witzgall R, O’Leary E, Leaf A, Onaldi D, Bonventre JV . The Kruppel-associated box-A (KRAB-A) domain of zinc finger proteins mediates transcriptional repression. Proc Natl Acad Sci USA 1994; 91: 4514–4518.
Meiboom M, Murua Escobar H, Pentimalli F, Fusco A, Belge G, Bullerdiek J . A 3.4-kbp transcript of ZNF331 is solely expressed in follicular thyroid adenomas. Cytogenet Genome Res 2003; 101: 113–117.
Fredericks WJ, Ayyanathan K, Herlyn M, Friedman JR, Rauscher 3rd FJ . An engineered PAX3-KRAB transcriptional repressor inhibits the malignant phenotype of alveolar rhabdomyosarcoma cells harboring the endogenous PAX3-FKHR oncogene. Mol Cell Biol 2000; 20: 5019–5031.
Sohn JH, Yeh BI, Choi JW, Yoon J, Namkung J, Park KK et al. Repression of human telomerase reverse transcriptase using artificial zinc finger transcription factors. Mol Cancer Res 2010; 8: 246–253.
Estornes Y, Gay F, Gevrey JC, Navoizat S, Nejjari M, Scoazec JY et al. Differential involvement of destrin and cofilin-1 in the control of invasive properties of Isreco1 human colon cancer cells. Int J Cancer 2007; 121: 2162–2171.
Janknecht R . Multi-talented DEAD-box proteins and potential tumor promoters: p68 RNA helicase (DDX5) and its paralog, p72 RNA helicase (DDX17). Am J Transl Res 2010; 2: 223–234.
Jalal C, Uhlmann-Schiffler H, Stahl H . Redundant role of DEAD box proteins p68 (Ddx5) and p72/p82 (Ddx17) in ribosome biogenesis and cell proliferation. Nucleic Acids Res 2007; 35: 3590–3601.
Jasiulionis MG, Luchessi AD, Moreira AG, Souza PP, Suenaga AP, Correa M et al. Inhibition of eukaryotic translation initiation factor 5A (eIF5A) hypusination impairs melanoma growth. Cell Biochem Funct 2007; 25: 109–114.
Freist W, Logan DT, Gauss DH . Glycyl-tRNA synthetase. Biol Chem Hoppe Seyler 1996; 377: 343–356.
Takeshita T, Arita T, Higuchi M, Asao H, Endo K, Kuroda H et al. STAM, signal transducing adaptor molecule, is associated with Janus kinases and involved in signaling for cell growth and c-myc induction. Immunity 1997; 6: 449–457.
Pennacchio LA, Bergmann A, Fukushima A, Okubo K, Salemi A, Lennon GG . Structure, sequence and location of the UQCRFS1 gene for the human Rieske Fe-S protein. Gene 1995; 155: 207–211.
Ohashi Y, Kaneko SJ, Cupples TE, Young SR . Ubiquinol cytochrome c reductase (UQCRFS1) gene amplification in primary breast cancer core biopsy samples. Gynecol Oncol 2004; 93: 54–58.
Kaneko SJ, Gerasimova T, Smith ST, Lloyd KO, Suzumori K, Young SR . CA125 and UQCRFS1 FISH studies of ovarian carcinoma. Gynecol Oncol 2003; 90: 29–36.
Leung SY, Ho C, Tu IP, Li R, So S, Chu KM et al. Comprehensive analysis of 19q12 amplicon in human gastric cancers. Mod Pathol 2006; 19: 854–863.
Anazawa Y, Nakagawa H, Furihara M, Ashida S, Tamura K, Yoshioka H et al. PCOTH, a novel gene overexpressed in prostate cancers, promotes prostate cancer cell growth through phosphorylation of oncoprotein TAF-Ibeta/SET. Cancer Res 2005; 65: 4578–4586.
Fan Z, Beresford PJ, Oh DY, Zhang D, Lieberman J . Tumor suppressor NM23-H1 is a granzyme A-activated DNase during CTL-mediated apoptosis, and the nucleosome assembly protein SET is its inhibitor. Cell 2003; 112: 659–672.
Wassarman DA, Steitz JA . RNA splicing. Alive with DEAD proteins. Nature 1991; 349: 463–464.
Choi YJ, Lee SG . The DEAD-box RNA helicase DDX3 interacts with DDX5, co-localizes with it in the cytoplasm during the G2/M phase of the cycle, and affects its shuttling during mRNP export. J Cell Biochem 2011; 113: 985–996; e-pub ahead of print 27 October 2011; doi: 10.1002/jcb.23428.
Otsubo T, Iwaya K, Mukai Y, Mizokami Y, Serizawa H, Matsuoka T et al. Involvement of Arp2/3 complex in the process of colorectal carcinogenesis. Mod Pathol 2004; 17: 461–467.
Leszczyniecka M, Kang DC, Sarkar D, Su ZZ, Holmes M, Valerie K et al. Identification and cloning of human polynucleotide phosphorylase, hPNPase old-35, in the context of terminal differentiation and cellular senescence. Proc Natl Acad Sci USA 2002; 99: 16636–16641.
Sarkar D, Park ES, Emdad L, Randolph A, Valerie K, Fisher PB . Defining the domains of human polynucleotide phosphorylase (hPNPaseOLD-35) mediating cellular senescence. Mol Cell Biol 2005; 25: 7333–7343.
Sarkar D, Leszczyniecka M, Kang DC, Lebedeva IV, Valerie K, Dhar S et al. Down-regulation of Myc as a potential target for growth arrest induced by human polynucleotide phosphorylase (hPNPaseold-35) in human melanoma cells. J Biol Chem 2003; 278: 24542–24551.
Tiranti V, Rossi E, Ruiz-Carrillo A, Rossi G, Rocchi M, DiDonato S et al. Chromosomal localization of mitochondrial transcription factor A (TCF6), single-stranded DNA-binding protein (SSBP), and endonuclease G (ENDOG), three human housekeeping genes involved in mitochondrial biogenesis. Genomics 1995; 25: 559–564.
Huang J, Gong Z, Ghosal G, Chen J . SOSS complexes participate in the maintenance of genomic stability. Mol Cell 2009; 35: 384–393.
Pollard KS, Serre D, Wang X, Tao H, Grundberg E, Hudson TJ et al. A genome-wide approach to identifying novel-imprinted genes. Hum Genet 2008; 122: 625–634.
Yu J, Leung WK, Ebert MP, Ng EK, Go MY, Wang HB et al. Increased expression of survivin in gastric cancer patients and in first degree relatives. Br J Cancer 2002; 87: 91–97.
Li G, Zhang XA, Wang H, Wang X, Meng CL, Chan CY et al. Comparative proteomic analysis of mesenchymal stem cells derived from human bone marrow, umbilical cord, and placenta: implication in the migration. Proteomics 2009; 9: 20–30.
Acknowledgements
This project was supported by the National Basic Research Program of China (973 Program, 2010CB529305), Research Grants Council RGC CERG CUHK (473008), CUHK Focused Investment Grant (1903026) and RFCID (10090942, 11100022).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Yu, J., Liang, Q., Wang, J. et al. Zinc-finger protein 331, a novel putative tumor suppressor, suppresses growth and invasiveness of gastric cancer. Oncogene 32, 307–317 (2013). https://doi.org/10.1038/onc.2012.54
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.54
Keywords
This article is cited by
-
Molecular cloning, characterization, and functional analysis of the uncharacterized C11orf96 gene
BMC Veterinary Research (2022)
-
Gamma-glutamyltransferase 7 suppresses gastric cancer by cooperating with RAB7 to induce mitophagy
Oncogene (2022)
-
MiR-183-5p-PNPT1 Axis Enhances Cisplatin-induced Apoptosis in Bladder Cancer Cells
Current Medical Science (2022)
-
Association of ZNF331 and WIF1 methylation in peripheral blood leukocytes with the risk and prognosis of gastric cancer
BMC Cancer (2021)
-
ZNF471 modulates EMT and functions as methylation regulated tumor suppressor with diagnostic and prognostic significance in cervical cancer
Cell Biology and Toxicology (2021)