Abstract
The Ras superfamily of GTPases is involved in the modification of many cellular processes including cellular motility, proliferation and differentiation. Our laboratory has previously identified the RalGDS-related (Rgr) oncogene in a DMBA (7,12-dimethylbenz[α]anthracene)-induced rabbit squamous cell carcinoma and its human orthologue, hRgr. In this study, we analyzed the expression levels of the human hRgr transcript in a panel of human hematopoietic malignancies and found that a truncated form (diseased-truncated (Dtr-hrgr)) was significantly overexpressed in many T-cell-derived neoplasms. Although the Rgr proto-oncogene belongs to the RalGDS family of guanine nucleotide exchange factors (GEFs), we show that upon the introduction of hRgr into fibroblast cell lines, it is able to elicit the activation of both Ral and Ras GTPases. Moreover, in vitro guanine nucleotide exchange assays confirm that hRgr promotes Ral and Ras activation through GDP dissociation, which is a critical characteristic of GEF proteins. hRgr has guanine nucleotide exchange activity for both small GTPases and this activity was reduced when a point mutation within the catalytic domain (CDC25) of the protein, (cd) Dtr-hRgr, was utilized. These observations prompted the analysis of the biological effects of hRgr and (cd) hRgr expression in cultured cells. Here, we show that hRgr increases proliferation in low serum, increases invasion, reduces anchorage dependence and promotes the progression into the S phase of the cell cycle; properties that are abolished or severely reduced in the presence of the catalytic dead mutant. We conclude that the ability of hRgr to activate both Ral and Ras is responsible for its transformation-inducing phenotype and it could be an important contributor in the development of some T-cell malignancies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Albright CF, Giddings BW, Liu J, Vito M, Weinberg RA . (1993). Characterization of a guanine nucleotide dissociation stimulator for a ras-related GTPase. EMBO J 12: 339–347.
Bhattacharya M, Anborgh PH, Babwah AV, Dale LB, Dobransky T, Benovic JL et al. (2002). Beta-arrestins regulate a Ral-GDS Ral effector pathway that mediates cytoskeletal reorganization. Nat Cell Biol 4: 547–555.
Bos JL . (1989). ras oncogenes in human cancer: a review. Cancer Res 49: 4682–4689.
Boykevisch S, Zhao C, Sondermann H, Philippidou P, Halegoua S, Kuriyan J et al. (2006). Regulation of ras signaling dynamics by Sos-mediated positive feedback. Curr Biol 16: 2173–2179.
Camonis JH, White MA . (2005). Ral GTPases: corrupting the exocyst in cancer cells. Trends Cell Biol 15: 327–332.
Cascone I, Selimoglu R, Ozdemir C, Del Nery E, Yeaman C, White M et al. (2008). Distinct roles of RalA and RalB in the progression of cytokinesis are supported by distinct RalGEFs. EMBO J 27: 2375–2387.
Ceriani M, Scandiuzzi C, Amigoni L, Tisi R, Berruti G, Martegani E . (2007). Functional analysis of RalGPS2, a murine guanine nucleotide exchange factor for RalA GTPase. Exp Cell Res 313: 2293–2307.
Chardin P, Tavitian A . (1986). The ral gene: a new ras related gene isolated by the use of a synthetic probe. EMBO J 5: 2203–2208.
Chien Y, Kim S, Bumeister R, Loo YM, Kwon SW, Johnson CL et al. (2006). RalB GTPase-mediated activation of the IkappaB family kinase TBK1 couples innate immune signaling to tumor cell survival. Cell 127: 157–170.
Chissoe SL, Bodenteich A, Wang YF, Wang YP, Burian D, Clifton SW et al. (1995). Sequence and analysis of the human ABL gene, the BCR gene, and regions involved in the Philadelphia chromosomal translocation. Genomics 27: 67–82.
Corominas M, Kamino H, Leon J, Pellicer A . (1989). Oncogene activation in human benign tumors of the skin (keratoacanthomas): is HRAS involved in differentiation as well as proliferation? Proc Natl Acad Sci USA 86: 6372–6376.
D'Adamo DR, Novick S, Kahn JM, Leonardi P, Pellicer A . (1997). Rsc: a novel oncogene with structural and functional homology with the gene family of exchange factors for Ral. Oncogene 14: 1295–1305.
Das J, Ho M, Zikherman J, Govern C, Yang M, Weiss A et al. (2009). Digital signaling and hysteresis characterize ras activation in lymphoid cells. Cell 136: 337–351.
Frankel P, Aronheim A, Kavanagh E, Balda MS, Matter K, Bunney TD et al. (2005). RalA interacts with ZONAB in a cell density-dependent manner and regulates its transcriptional activity. EMBO J 24: 54–62.
Garcia-Espana A, Salazar E, Sun TT, Wu XR, Pellicer A . (2005). Differential expression of cell cycle regulators in phenotypic variants of transgenically induced bladder tumors: implications for tumor behavior. Cancer Res 65: 1150–1157.
Gotoh T, Cai D, Tian X, Feig LA, Lerner A . (2000). p130Cas regulates the activity of AND-34, a novel Ral, Rap1, and R-Ras guanine nucleotide exchange factor. J Biol Chem 275: 30118–30123.
Hall BE, Yang SS, Boriack-Sjodin PA, Kuriyan J, Bar-Sagi D . (2001). Structure-based mutagenesis reveals distinct functions for Ras switch 1 and switch 2 in Sos-catalyzed guanine nucleotide exchange. J Biol Chem 276: 27629–27637.
Hernandez-Munoz I, Benet M, Calero M, Jimenez M, Diaz R, Pellicer A . (2003). Rgr oncogene: activation by elimination of translational controls and mislocalization. Cancer Res 63: 4188–4195.
Hernandez-Munoz I, Malumbres M, Leonardi P, Pellicer A . (2000). The Rgr oncogene (homologous to RalGDS) induces transformation and gene expression by activating Ras, Ral and Rho mediated pathways. Oncogene 19: 2745–2757.
Hofer F, Berdeaux R, Martin GS . (1998). Ras-independent activation of Ral by a Ca(2+)-dependent pathway. Curr Biol 8: 839–842.
Hofer F, Fields S, Schneider C, Martin GS . (1994). Activated Ras interacts with the Ral guanine nucleotide dissociation stimulator. Proc Natl Acad Sci USA 91: 11089–11093.
Jullien-Flores V, Mahe Y, Mirey G, Leprince C, Meunier-Bisceuil B, Sorkin A et al. (2000). RLIP76, an effector of the GTPase Ral, interacts with the AP2 complex: involvement of the Ral pathway in receptor endocytosis. J Cell Sci 113 (Part 16): 2837–2844.
Kurzrock R, Kantarjian HM, Druker BJ, Talpaz M . (2003). Philadelphia chromosome-positive leukemias: from basic mechanisms to molecular therapeutics. Ann Intern Med 138: 819–830.
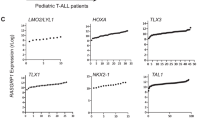
Leonardi P, Kassin E, Hernandez-Munoz I, Diaz R, Inghirami G, Pellicer A . (2002). Human rgr: transforming activity and alteration in T-cell malignancies. Oncogene 21: 5108–5116.
Lim KH, Baines AT, Fiordalisi JJ, Shipitsin M, Feig LA, Cox AD et al. (2005). Activation of RalA is critical for Ras-induced tumorigenesis of human cells. Cancer Cell 7: 533–545.
Lim KH, O'Hayer K, Adam SJ, Kendall SD, Campbell PM, Der CJ et al. (2006). Divergent roles for RalA and RalB in malignant growth of human pancreatic carcinoma cells. Curr Biol 16: 2385–2394.
Linnemann T, Kiel C, Herter P, Herrmann C . (2002). The activation of RalGDS can be achieved independently of its Ras binding domain. Implications of an activation mechanism in Ras effector specificity and signal distribution. J Biol Chem 277: 7831–7837.
Margarit SM, Sondermann H, Hall BE, Nagar B, Hoelz A, Pirruccello M et al. (2003). Structural evidence for feedback activation by Ras.GTP of the Ras-specific nucleotide exchange factor SOS. Cell 112: 685–695.
Mittar D, Sehajpal PK, Lander HM . (2004). Nitric oxide activates Rap1 and Ral in a Ras-independent manner. Biochem Biophys Res Commun 322: 203–209.
Muller AJ, Young JC, Pendergast AM, Pondel M, Landau NR, Littman DR et al. (1991). BCR first exon sequences specifically activate the BCR/ABL tyrosine kinase oncogene of Philadelphia chromosome-positive human leukemias. Mol Cell Biol 11: 1785–1792.
Ohta Y, Suzuki N, Nakamura S, Hartwig JH, Stossel TP . (1999). The small GTPase RalA targets filamin to induce filopodia. Proc Natl Acad Sci USA 96: 2122–2128.
Oxford G, Owens CR, Titus BJ, Foreman TL, Herlevsen MC, Smith SC et al. (2005). RalA and RalB: antagonistic relatives in cancer cell migration. Cancer Res 65: 7111–7120.
Oxford G, Theodorescu D . (2003). The role of Ras superfamily proteins in bladder cancer progression. J Urol 170: 1987–1993.
Paulsson K, Cazier JB, Macdougall F, Stevens J, Stasevich I, Vrcelj N et al. (2008). Microdeletions are a general feature of adult and adolescent acute lymphoblastic leukemia: unexpected similarities with pediatric disease. Proc Natl Acad Sci USA 105: 6708–6713.
Rodenhuis S . (1992). Ras and human tumors. Semin Cancer Biol 3: 241–247.
Roose JP, Mollenauer M, Ho M, Kurosaki T, Weiss A . (2007). Unusual interplay of two types of Ras activators, RasGRP and SOS, establishes sensitive and robust Ras activation in lymphocytes. Mol Cell Biol 27: 2732–2745.
Rosse C, Hatzoglou A, Parrini MC, White MA, Chavrier P, Camonis J . (2006). RalB mobilizes the exocyst to drive cell migration. Mol Cell Biol 26: 727–734.
Rowley JD . (1973). Letter: a new consistent chromosomal abnormality in chronic myelogenous leukaemia identified by quinacrine fluorescence and Giemsa staining. Nature 243: 290–293.
Sekido Y, Pass HI, Bader S, Mew DJ, Christman MF, Gazdar AF et al. (1995). Neurofibromatosis type 2 (NF2) gene is somatically mutated in mesothelioma but not in lung cancer. Cancer Res 55: 1227–1231.
Senga T, Iwamoto T, Kitamura T, Miyake Y, Hamaguchi M . (2001). JAK/STAT3-dependent activation of the RalGDS/Ral pathway in M1 mouse myeloid leukemia cells. J Biol Chem 276: 32678–32681.
Shao H, Andres DA . (2000). A novel RalGEF-like protein, RGL3, as a candidate effector for rit and Ras. J Biol Chem 275: 26914–26924.
Smith SC, Oxford G, Wu Z, Nitz MD, Conaway M, Frierson HF et al. (2006). The metastasis-associated gene CD24 is regulated by Ral GTPase and is a mediator of cell proliferation and survival in human cancer. Cancer Res 66: 1917–1922.
Sondermann H, Soisson SM, Boykevisch S, Yang SS, Bar-Sagi D, Kuriyan J . (2004). Structural analysis of autoinhibition in the Ras activator Son of sevenless. Cell 119: 393–405.
Sood R, Makalowska I, Carpten JD, Robbins CM, Stephan DA, Connors TD et al. (2000). The human RGL (RalGDS-like) gene: cloning, expression analysis and genomic organization. Biochim Biophys Acta 1491: 285–288.
Takai Y, Sasaki T, Matozaki T . (2001). Small GTP-binding proteins. Physiol Rev 81: 153–208.
Takaya A, Kamio T, Masuda M, Mochizuki N, Sawa H, Sato M et al. (2007). R-Ras regulates exocytosis by Rgl2/Rlf-mediated activation of RalA on endosomes. Mol Biol Cell 18: 1850–1860.
Tazmini G, Beaulieu N, Woo A, Zahedi B, Goulding RE, Kay RJ . (2009). Membrane localization of RasGRP1 is controlled by an EF-hand, and by the GEF domain. Biochim Biophys Acta 1793: 447–461.
Tchevkina E, Agapova L, Dyakova N, Martinjuk A, Komelkov A, Tatosyan A . (2005). The small G-protein RalA stimulates metastasis of transformed cells. Oncogene 24: 329–335.
Urano T, Emkey R, Feig LA . (1996). Ral-GTPases mediate a distinct downstream signaling pathway from Ras that facilitates cellular transformation. EMBO J 15: 810–816.
Wigler M, Pellicer A, Silverstein S, Axel R . (1978). Biochemical transfer of single-copy eucaryotic genes using total cellular DNA as donor. Cell 14: 725–731.
Wolthuis RM, Bauer B, vant't Veer LJ, de Vries-Smits AM, Cool RH, Spaargarer M et al. (1996). RalGDS-like factor (Rlf) is a novel Ras and Rap1A-associating protein. Oncogene 13: 353–362.
Zahir N, Lakins JN, Russell A, Ming W, Chatterjee C, Rozenberg GI et al. (2003). Autocrine laminin-5 ligates alpha6beta4 integrin and activates RAC and NFkappaB to mediate anchorage-independent survival of mammary tumors. J Cell Biol 163: 1397–1407.
Acknowledgements
We are grateful to Drs M Philips, D Bar-Sagi and J Bos for the gift of expression plasmids. We thank Kamlesh Yadav and Pamela Sung for their assistance with the mant-guanine nucleotide exchange and cellular proliferation assays, respectively, and Dr M Liu for her assistance with the statistical analysis for the cellular proliferation assay. These studies were supported by NIH Grants CA50434 and CA36327 to AP.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Osei-Sarfo, K., Martello, L., Ibrahim, S. et al. The human Rgr oncogene is overexpressed in T-cell malignancies and induces transformation by acting as a GEF for Ras and Ral. Oncogene 30, 3661–3671 (2011). https://doi.org/10.1038/onc.2011.93
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.93
Keywords
This article is cited by
-
Sindbis viral vectors target hematopoietic malignant cells
Cancer Gene Therapy (2012)