Abstract
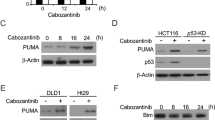
Aberrant Ras/Raf/MEK/ERK signaling is one of the most prevalent oncogenic alterations and confers survival advantage to tumor cells. Inhibition of this pathway can effectively suppress tumor cell growth. For example, sorafenib, a multi-kinase inhibitor targeting c-Raf and other oncogenic kinases, has been used clinically for treating advanced liver and kidney tumors, and also has shown efficacy against other malignancies. However, how inhibition of oncogenic signaling by sorafenib and other drugs suppresses tumor cell growth remains unclear. In this study, we found that sorafenib kills cancer cells by activating PUMA (p53-upregulated modulator of apoptosis), a p53 target and a BH3-only Bcl-2 family protein. Sorafenib treatment induces PUMA in a variety of cancer cells irrespective of their p53 status. Surprisingly, the induction of PUMA by sorafenib is mediated by IκB-independent activation of nuclear factor (NF)-κB, which directly binds to the PUMA promoter to activate its transcription. NF-κB activation by sorafenib requires glycogen synthase kinase 3β activation, subsequent to ERK inhibition. Deficiency in PUMA abrogates sorafenib-induced apoptosis and caspase activation, and renders sorafenib resistance in colony formation and xenograft tumor assays. Furthermore, the chemosensitization effect of sorafenib is dependent on PUMA, and involves concurrent PUMA induction through different pathways. BH3 mimetics potentiate the anti-cancer effects of sorafenib, and restore sorafenib sensitivity in resistant cells. Together, these results demonstrate a key role of PUMA-dependent apoptosis in therapeutic inhibition of Ras/Raf/MEK/ERK signaling. They provide a rationale for manipulating the apoptotic machinery to improve sensitivity and overcome resistance to the therapies that target oncogenic kinase signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Abbreviations
- eIF4E:
-
eukaryotic translation initiation factor 4E
- HCC:
-
hepatocellular carcinoma
- FoxO3a:
-
forkhead box O3a
- KO:
-
knockout
- GSK3β:
-
glycogen synthase kinase 3β
- Mcl-1, myeloid cell leukemia 1; NF-κB:
-
nuclear factor-κB
- PUMA:
-
p53-upregulated modulator of apoptosis
- siRNA:
-
small interfering RNA
- TNF-α:
-
tumor necrosis factor-α
- WT:
-
wild type
References
Vogelstein B, Kinzler KW . Cancer genes and the pathways they control. Nat Med 2004; 10: 789–799.
Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med 2007; 356: 125–134.
Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 2008; 359: 378–390.
Green DR, Kroemer G . Pharmacological manipulation of cell death: clinical applications in sight? J Clin Invest 2005; 115: 2610–2617.
Sordella R, Bell DW, Haber DA, Settleman J . Gefitinib-sensitizing EGFR mutations in lung cancer activate anti-apoptotic pathways. Science 2004; 305: 1163–1167.
Wilhelm S, Carter C, Lynch M, Lowinger T, Dumas J, Smith RA et al. Discovery and development of sorafenib: a multikinase inhibitor for treating cancer. Nat Rev Drug Discov 2006; 5: 835–844.
Wilhelm SM, Adnane L, Newell P, Villanueva A, Llovet JM, Lynch M . Preclinical overview of sorafenib, a multikinase inhibitor that targets both Raf and VEGF and PDGF receptor tyrosine kinase signaling. Mol Cancer Ther 2008; 7: 3129–3140.
Yu C, Bruzek LM, Meng XW, Gores GJ, Carter CA, Kaufmann SH et al. The role of Mcl-1 downregulation in the proapoptotic activity of the multikinase inhibitor BAY 43-9006. Oncogene 2005; 24: 6861–6869.
Rahmani M, Davis EM, Bauer C, Dent P, Grant S . Apoptosis induced by the kinase inhibitor BAY 43-9006 in human leukemia cells involves down-regulation of Mcl-1 through inhibition of translation. J Biol Chem 2005; 280: 35217–35227.
Liu L, Cao Y, Chen C, Zhang X, McNabola A, Wilkie D et al. Sorafenib blocks the RAF/MEK/ERK pathway, inhibits tumor angiogenesis, and induces tumor cell apoptosis in hepatocellular carcinoma model PLC/PRF/5. Cancer Res 2006; 66: 11851–11858.
Yu J, Zhang L . PUMA, a potent killer with or without p53. Oncogene 2008; 27 (Suppl 1): S71–S83.
Yu J, Yue W, Wu B, Zhang L . PUMA sensitizes lung cancer cells to chemotherapeutic agents and irradiation. Clin Cancer Res 2006; 12: 2928–2936.
Dudgeon C, Wang P, Sun X, Peng R, Sun Q, Yu J et al. PUMA induction by FoxO3a mediates the anticancer activities of the broad-range kinase inhibitor UCN-01. Mol Cancer Ther 2010; 9: 2893–2902.
Sun Q, Ming L, Thomas SM, Wang Y, Chen ZG, Ferris RL et al. PUMA mediates EGFR tyrosine kinase inhibitor-induced apoptosis in head and neck cancer cells. Oncogene 2009; 18: 2348–2357.
Wang P, Qiu W, Dudgeon C, Liu H, Huang C, Zambetti GP et al. PUMA is directly activated by NF-kappaB and contributes to TNF-alpha-induced apoptosis. Cell Death Differ 2009; 16: 1192–1202.
Ming L, Sakaida T, Yue W, Jha A, Zhang L, Yu J . Sp1 and p73 Activate PUMA Following Serum Starvation. Carcinogenesis 2008; 29: 1878–1884.
Ming L, Wang P, Bank A, Yu J, Zhang L . PUMA dissociates Bax and BCL-XL to induce apoptosis in colon cancer cells. J Biol Chem 2006; 281: 16034–16042.
Yu J, Wang P, Ming L, Wood MA, Zhang L . SMAC/Diablo mediates the proapoptotic function of PUMA by regulating PUMA-induced mitochondrial events. Oncogene 2007; 26: 4189–4198.
Yu J, Wang Z, Kinzler KW, Vogelstein B, Zhang L . PUMA mediates the apoptotic response to p53 in colorectal cancer cells. Proc Natl Acad Sci USA 2003; 100: 1931–1936.
Wilhelm SM, Carter C, Tang L, Wilkie D, McNabola A, Rong H et al. BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res 2004; 64: 7099–7109.
Plastaras JP, Kim SH, Liu YY, Dicker DT, Dorsey JF, McDonough J et al. Cell cycle dependent and schedule-dependent antitumor effects of sorafenib combined with radiation. Cancer Res 2007; 67: 9443–9454.
You H, Pellegrini M, Tsuchihara K, Yamamoto K, Hacker G, Erlacher M et al. FOXO3a-dependent regulation of Puma in response to cytokine/growth factor withdrawal. J Exp Med 2006; 203: 1657–1663.
Huang S, Sinicrope FA . Sorafenib inhibits STAT3 activation to enhance TRAIL-mediated apoptosis in human pancreatic cancer cells. Mol Cancer Ther 2010; 9: 742–750.
Baud V, Karin M . Is NF-kappaB a good target for cancer therapy? Hopes and pitfalls. Nat Rev Drug Discov 2009; 8: 33–40.
Cross DA, Alessi DR, Cohen P, Andjelkovich M, Hemmings BA . Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 1995; 378: 785–789.
Ding Q, Xia W, Liu JC, Yang JY, Lee DF, Xia J et al. Erk associates with and primes GSK-3beta for its inactivation resulting in upregulation of beta-catenin. Mol Cell 2005; 19: 159–170.
Zhang L, Ming L, Yu J . BH3 mimetics to improve cancer therapy; mechanisms and examples. Drug Resist Updat 2007; 10: 207–217.
Wilhelm S, Chien DS . BAY 43-9006: preclinical data. Curr Pharm Des 2002; 8: 2255–2257.
Clark JW, Eder JP, Ryan D, Lathia C, Lenz HJ . Safety and pharmacokinetics of the dual action Raf kinase and vascular endothelial growth factor receptor inhibitor, BAY 43-9006, in patients with advanced, refractory solid tumors. Clin Cancer Res 2005; 11: 5472–5480.
Opferman JT, Iwasaki H, Ong CC, Suh H, Mizuno S, Akashi K et al. Obligate role of anti-apoptotic MCL-1 in the survival of hematopoietic stem cells. Science 2005; 307: 1101–1104.
Zhang W, Konopleva M, Ruvolo VR, McQueen T, Evans RL, Bornmann WG et al. Sorafenib induces apoptosis of AML cells via Bim-mediated activation of the intrinsic apoptotic pathway. Leukemia 2008; 22: 808–818.
Dutta J, Fan Y, Gupta N, Fan G, Gelinas C . Current insights into the regulation of programmed cell death by NF-kappaB. Oncogene 2006; 25: 6800–6816.
Biton S, Ashkenazi A . NEMO and RIP1 control cell fate in response to extensive DNA damage via TNF-alpha feedforward signaling. Cell 2011; 145: 92–103.
Augustine CK, Toshimitsu H, Jung SH, Zipfel PA, Yoo JS, Yoshimoto Y et al. Sorafenib, a multikinase inhibitor, enhances the response of melanoma to regional chemotherapy. Mol Cancer Ther 2010; 9: 2090–2101.
Sun W, Powell M, O’Dwyer PJ, Catalano P, Ansari RH, Benson III AB . Phase II study of sorafenib in combination with docetaxel and cisplatin in the treatment of metastatic or advanced gastric and gastroesophageal junction adenocarcinoma: ECOG 5203. J Clin Oncol 2010; 28: 2947–2951.
Kim C, Lee JL, Choi YH, Kang BW, Ryu MH, Chang HM et al. Phase I dose-finding study of sorafenib in combination with capecitabine and cisplatin as a first-line treatment in patients with advanced gastric cancer. Invest New Drugs 2010 (e-pub ahead of print 14 September 2010).
Molhoek KR, Brautigan DL, Slingluff Jr CL . Synergistic inhibition of human melanoma proliferation by combination treatment with B-Raf inhibitor BAY43-9006 and mTOR inhibitor rapamycin. J Transl Med 2005; 3: 39.
Martinelli E, Troiani T, Morgillo F, Rodolico G, Vitagliano D, Morelli MP et al. Synergistic antitumor activity of sorafenib in combination with epidermal growth factor receptor inhibitors in colorectal and lung cancer cells. Clin Cancer Res 2010; 16: 4990–5001.
Lind JS, Dingemans AM, Groen HJ, Thunnissen FB, Bekers O, Heideman DA et al. A multicenter phase II study of erlotinib and sorafenib in chemotherapy-naive patients with advanced non-small cell lung cancer. Clin Cancer Res 2010; 16: 3078–3087.
Hong DS, Sebti SM, Newman RA, Blaskovich MA, Ye L, Gagel RF et al. Phase I trial of a combination of the multikinase inhibitor sorafenib and the farnesyltransferase inhibitor tipifarnib in advanced malignancies. Clin Cancer Res 2009; 15: 7061–7068.
Gomez-Benito M, Marzo I, Anel A, Naval J . Farnesyltransferase inhibitor BMS-214662 induces apoptosis in myeloma cells through PUMA up-regulation, Bax and Bak activation, and Mcl-1 elimination. Mol Pharmacol 2005; 67: 1991–1998.
Zhang G, Park MA, Mitchell C, Hamed H, Rahmani M, Martin AP et al. Vorinostat and sorafenib synergistically kill tumor cells via FLIP suppression and CD95 activation. Clin Cancer Res 2008; 14: 5385–5399.
Martin AP, Park MA, Mitchell C, Walker T, Rahmani M, Thorburn A et al. BCL-2 family inhibitors enhance histone deacetylase inhibitor and sorafenib lethality via autophagy and overcome blockade of the extrinsic pathway to facilitate killing. Mol Pharmacol 2009; 76: 327–341.
Hikita H, Takehara T, Shimizu S, Kodama T, Shigekawa M, Iwase K et al. The Bcl-xL inhibitor, ABT-737, efficiently induces apoptosis and suppresses growth of hepatoma cells in combination with sorafenib. Hepatology 2010; 52: 1310–1321.
Chen KF, Chen HL, Tai WT, Feng WC, Hsu CH, Chen PJ et al. Activation of PI3K/Akt signaling pathway mediates acquired resistance to sorafenib in hepatocellular carcinoma cells. J Pharmacol Exp Ther 2011; 337: 155–161.
Gedaly R, Angulo P, Hundley J, Daily MF, Chen C, Koch A et al. PI-103 and sorafenib inhibit hepatocellular carcinoma cell proliferation by blocking Ras/Raf/MAPK and PI3K/AKT/mTOR pathways. Anticancer Res 2010; 30: 4951–4958.
Wang P, Yu J, Zhang L . The nuclear function of p53 is required for PUMA-mediated apoptosis induced by DNA damage. Proc Natl Acad Sci USA 2007; 104: 4054–4059.
Yue W, Sun Q, Dacic S, Landreneau RJ, Siegfried JM, Yu J et al. Downregulation of Dkk3 activates beta-catenin/TCF-4 signaling in lung cancer. Carcinogenesis 2008; 29: 84–92.
Kohli M, Yu J, Seaman C, Bardelli A, Kinzler KW, Vogelstein B et al. SMAC/Diablo-dependent apoptosis induced by nonsteroidal antiinflammatory drugs (NSAIDs) in colon cancer cells. Proc Natl Acad Sci USA 2004; 101: 16897–16902.
Li H, Wang P, Sun Q, Ding WX, Yin XM, Sobol RW et al. Following cytochrome c release, autophagy is inhibited during chemotherapy-induced apoptosis by caspase 8-mediated cleavage of Beclin 1. Cancer Res 2011; 71: 3625–3634.
Qiu W, Wang X, Leibowitz B, Liu H, Barker N, Okada H et al. Chemoprevention by nonsteroidal anti-inflammatory drugs eliminates oncogenic intestinal stem cells via SMAC-dependent apoptosis. Proc Natl Acad Sci USA 2010; 107: 20027–20032.
Acknowledgements
We thank Monica E Buchanan, Brian Leibowitz, Mathew F Brown and other lab members for critical reading and helpful discussion, and Drs Jennifer R Grandis and Jing Hu for help with analyses of STAT1, STAT3, eIF4E and GSK3β. This work is supported by NIH grants CA106348 and CA121105 and American Cancer Society grant RSG-07-156-01-CNE (LZ); Flight Attendant Medical Research Institute, NIH grant CA129829 and American Cancer Society grant RGS-10-124-01-CCE (JY); NIH National Research Service Award postdoctoral fellowship grant F32CA139882 (CD); and China Scholarship Council (RP). LZ is a scholar of the V Foundation for Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Dudgeon, C., Peng, R., Wang, P. et al. Inhibiting oncogenic signaling by sorafenib activates PUMA via GSK3β and NF-κB to suppress tumor cell growth. Oncogene 31, 4848–4858 (2012). https://doi.org/10.1038/onc.2011.644
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.644
Keywords
This article is cited by
-
The GSK3β/Mcl-1 axis is regulated by both FLT3-ITD and Axl and determines the apoptosis induction abilities of FLT3-ITD inhibitors
Cell Death Discovery (2023)
-
Cytochrome P450 1A2 overcomes nuclear factor kappa B-mediated sorafenib resistance in hepatocellular carcinoma
Oncogene (2021)
-
Anti-miR-518d-5p overcomes liver tumor cell death resistance through mitochondrial activity
Cell Death & Disease (2021)
-
Targeting mTOR suppressed colon cancer growth through 4EBP1/eIF4E/PUMA pathway
Cancer Gene Therapy (2020)
-
Chemogenomic screening identifies the Hsp70 co-chaperone DNAJA1 as a hub for anticancer drug resistance
Scientific Reports (2020)