Abstract
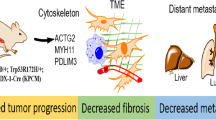
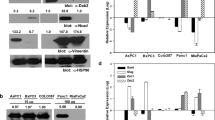
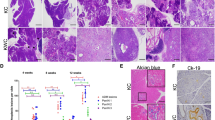
Pancreatic ductal adenocarcinoma (PDA) is often detected at a late stage, hence the identification of new therapies that have potential to block tumor progression is critical for this lethal disease. N-cadherin upregulation has been observed in many cancers including PDA, however, a causal role for this cell adhesion receptor in disease progression has yet to be defined. The concomitant expression of oncogenic KrasG12D and mutant p53 (Trp53R172H) in the murine pancreas results in metastatic PDA that recapitulates the cognate features of human pancreatic cancer providing an excellent animal model to identify genes required for tumor progression. Here we determine the consequences of genetically manipulating N-cadherin expression in a mouse model of PDA. Remarkably, mice with reduced N-cadherin expression (that is, Ncad−/+) survived 25% longer (177 vs 142 days, P<0.05) than animals expressing two wild-type N-cadherin (Cdh2) alleles. The survival benefit is likely due to a cumulative effect of N-cadherin's role in different aspects of tumorigenesis including tumor-cell survival, growth, migration and invasion. Interestingly, reduced hedgehog signaling may contribute to the better prognosis for the Ncad−/+ mice. Moreover, the matrix metalloproteinase MMP-7, associated with poor prognosis in PDA, was reduced in Ncad−/+ tumors. Finally, Ncad−/+ tumor cells exhibited decreased FGF-stimulated ERK1/2 activation consistent with N-cadherin's ability to promote FGFR signaling. These data support a critical role for N-cadherin in PDA and its potential prognostic value. Additionally, this study provides in vivo genetic evidence that the cell-surface protein N-cadherin represents a promising therapeutic target for the treatment of pancreatic cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hezel AF, Kimmelman AC, Stanger BZ, Bardeesy N, Depinho RA . Genetics and biology of pancreatic ductal adenocarcinoma. Genes Dev 2006; 20: 1218–1249.
Wheelock MJ, Shintani Y, Maeda M, Fukumoto Y, Johnson KR . Cadherin switching. J Cell Sci 2008; 121 (Part 6): 727–735.
Hazan RB, Phillips GR, Qiao RF, Norton L, Aaronson SA . Exogenous expression of N-cadherin in breast cancer cells induces cell migration, invasion, and metastasis. J Cell Biol 2000; 148: 779–790.
Nieman MT, Prudoff RS, Johnson KR, Wheelock MJ . N-cadherin promotes motility in human breast cancer cells regardless of their E-cadherin expression. J Cell Biol 1999; 147: 631–644.
Hingorani SR, Petricoin EF, Maitra A, Rajapakse V, King C, Jacobetz MA et al. Preinvasive and invasive ductal pancreatic cancer and its early detection in the mouse. Cancer Cell 2003; 4: 437–450.
Agbunag C, Bar-Sagi D . Oncogenic K-ras drives cell cycle progression and phenotypic conversion of primary pancreatic duct epithelial cells. Cancer Res 2004; 64: 5659–5663.
Brembeck FH, Schreiber FS, Deramaudt TB, Craig L, Rhoades B, Swain G et al. The mutant K-ras oncogene causes pancreatic periductal lymphocytic infiltration and gastric mucous neck cell hyperplasia in transgenic mice. Cancer Res 2003; 63: 2005–2009.
Deramaudt TB, Takaoka M, Upadhyay R, Bowser MJ, Porter J, Lee A et al. N-cadherin and keratinocyte growth factor receptor mediate the functional interplay between Ki-RASG12V and p53V143A in promoting pancreatic cell migration, invasion, and tissue architecture disruption. Mol Cell Biol 2006; 26: 4185–4200.
Shintani Y, Hollingsworth MA, Wheelock MJ, Johnson KR . Collagen I promotes metastasis in pancreatic cancer by activating c-Jun NH(2)-terminal kinase 1 and up-regulating N-cadherin expression. Cancer Res 2006; 66: 11745–11753.
Hingorani SR, Wang L, Multani AS, Combs C, Deramaudt TB, Hruban RH et al. Trp53R172H and KrasG12D cooperate to promote chromosomal instability and widely metastatic pancreatic ductal adenocarcinoma in mice. Cancer Cell 2005; 7: 469–483.
Kostetskii I, Li J, Xiong Y, Zhou R, Ferrari VA, Patel VV et al. Induced deletion of the N-cadherin gene in the heart leads to dissolution of the intercalated disc structure. Circ Res 2005; 96: 346–354.
Luo Y, Kostetskii I, Radice GL . N-cadherin is not essential for limb mesenchymal chondrogenesis. Dev Dyn 2005; 232: 336–344.
Johansson JK, Voss U, Kesavan G, Kostetskii I, Wierup N, Radice GL et al. N-cadherin is dispensable for pancreas development but required for beta-cell granule turnover. Genesis 2010; 48: 374–381.
Shintani Y, Fukumoto Y, Chaika N, Grandgenett PM, Hollingsworth MA, Wheelock MJ et al. ADH-1 suppresses N-cadherin-dependent pancreatic cancer progression. Int J Cancer 2008; 122: 71–77.
Suyama K, Shapiro I, Guttman M, Hazan RB . A signaling pathway leading to metastasis is controlled by N-cadherin and the FGF receptor. Cancer Cell 2002; 2: 301–314.
Schreiber FS, Deramaudt TB, Brunner TB, Boretti MI, Gooch KJ, Stoffers DA et al. Successful growth and characterization of mouse pancreatic ductal cells: functional properties of the Ki-RAS(G12V) oncogene. Gastroenterology 2004; 127: 250–260.
Berman DM, Karhadkar SS, Maitra A, Montes De Oca R, Gerstenblith MR, Briggs K et al. Widespread requirement for Hedgehog ligand stimulation in growth of digestive tract tumours. Nature 2003; 425: 846–851.
Morton JP, Mongeau ME, Klimstra DS, Morris JP, Lee YC, Kawaguchi Y et al. Sonic hedgehog acts at multiple stages during pancreatic tumorigenesis. Proc Natl Acad Sci USA 2007; 104: 5103–5108.
Thayer SP, di Magliano MP, Heiser PW, Nielsen CM, Roberts DJ, Lauwers GY et al. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 2003; 425: 851–856.
Karafin MS, Cummings CT, Fu B, Iacobuzio-Donahue CA . The developmental transcription factor Gata4 is overexpressed in pancreatic ductal adenocarcinoma. Int J Clin Exp Pathol 2009; 3: 47–55.
Crawford HC, Scoggins CR, Washington MK, Matrisian LM, Leach SD . Matrix metalloproteinase-7 is expressed by pancreatic cancer precursors and regulates acinar-to-ductal metaplasia in exocrine pancreas. J Clin Invest 2002; 109: 1437–1444.
Fukuda A, Wang SC, Morris IV JP, Folias AE, Liou A, Kim GE et al. Stat3 and MMP7 contribute to pancreatic ductal adenocarcinoma initiation and progression. Cancer Cell 2011; 19: 441–455.
Nomura S, Yoshitomi H, Takano S, Shida T, Kobayashi S, Ohtsuka M et al. FGF10/FGFR2 signal induces cell migration and invasion in pancreatic cancer. Br J Cancer 2008; 99: 305–313.
Cavallaro U, Niedermeyer J, Fuxa M, Christofori G . N-CAM modulates tumour-cell adhesion to matrix by inducing FGF-receptor signalling. Nat Cell Biol 2001; 3: 650–657.
Lehembre F, Yilmaz M, Wicki A, Schomber T, Strittmatter K, Ziegler D et al. NCAM-induced focal adhesion assembly: a functional switch upon loss of E-cadherin. EMBO J 2008; 27: 2603–2615.
Olive KP, Jacobetz MA, Davidson CJ, Gopinathan A, McIntyre D, Honess D et al. Inhibition of hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science 2009; 324: 1457–1461.
Hulit J, Suyama K, Chung S, Keren R, Agiostratidou G, Shan W et al. N-cadherin signaling potentiates mammary tumor metastasis via enhanced extracellular signal-regulated kinase activation. Cancer Res 2007; 67: 3106–3116.
Kang JS, Feinleib JL, Knox S, Ketteringham MA, Krauss RS . Promyogenic members of the Ig and cadherin families associate to positively regulate differentiation. Proc Natl Acad Sci USA 2003; 100: 3989–3994.
Tenzen T, Allen BL, Cole F, Kang JS, Krauss RS, McMahon AP . The cell surface membrane proteins Cdo and Boc are components and targets of the hedgehog signaling pathway and feedback network in mice. Dev Cell 2006; 10: 647–656.
Augustine CK, Yoshimoto Y, Gupta M, Zipfel PA, Selim MA, Febbo P et al. Targeting N-cadherin enhances antitumor activity of cytotoxic therapies in melanoma treatment. Cancer Res 2008; 68: 3777–3784.
Tanaka H, Kono E, Tran CP, Miyazaki H, Yamashiro J, Shimomura T et al. Monoclonal antibody targeting of N-cadherin inhibits prostate cancer growth, metastasis and castration resistance. Nat Med 2010; 16: 1414–1420.
Acknowledgements
We thank Dr D Tuveson (Cancer Research UK, Cambridge) for the LSL-K-rasG12D and LSL-Trp53R172H mice and Dr A Lowy (UCSD) for the Pdx1/Cre mice. We are grateful to Han Du, Craig Riley, David Kurz, Leeanne Griffith and Andrew Ho for technical assistance. This work was supported by the Center for Molecular Studies in Digestive and Liver Disease (NIH P30DK050306) and University Research Foundation—University of Pennsylvania, NIH R21 CA133609, Pilot Research Award—Thomas Jefferson University (to GLR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Su, Y., Li, J., Witkiewicz, A. et al. N-cadherin haploinsufficiency increases survival in a mouse model of pancreatic cancer. Oncogene 31, 4484–4489 (2012). https://doi.org/10.1038/onc.2011.574
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.574
Keywords
This article is cited by
-
N-cadherin in cancer metastasis, its emerging role in haematological malignancies and potential as a therapeutic target in cancer
BMC Cancer (2018)
-
N-cadherin functions as a growth suppressor in a model of K-ras-induced PanIN
Oncogene (2016)
-
Hypoxia promotes the invasion and metastasis of laryngeal cancer cells via EMT
Medical Oncology (2016)
-
Beyond E-cadherin: roles of other cadherin superfamily members in cancer
Nature Reviews Cancer (2014)
-
Bex2 is Critical for Migration and Invasion in Malignant Glioma Cells
Journal of Molecular Neuroscience (2013)