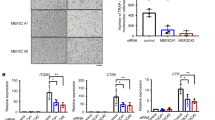
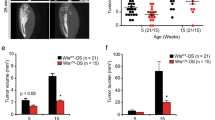
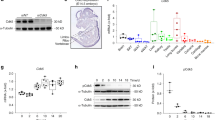
Abstract
Mice lacking c-fos develop osteopetrosis due to a block in osteoclast differentiation. Carboxy-terminal phosphorylation of Fos on serine 374 by ERK1/2 and serine 362 by RSK1/2 regulates Fos stability and transactivation potential in vitro. To assess the physiological relevance of Fos phosphorylation in vivo, serine 362 and/or serine 374 was replaced by alanine (Fos362A, Fos374A and FosAA) or by phospho-mimetic aspartic acid (FosDD). Homozygous mutants were healthy and skeletogenesis was largely unaffected. Fos C-terminal phosphorylation, predominantly on serine 374, was found important for osteoclast differentiation in vitro and affected lipopolysaccharide (LPS)-induced cytokine response in vitro and in vivo. Importantly, skin papilloma development was delayed in FosAA, Fos362A and Rsk2-deficient mice, accelerated in FosDD mice and unaffected in Fos374A mutants. Furthermore, the related Fos protein and putative RSK2 target Fra1 failed to substitute for Fos in papilloma development. This indicates that phosphorylation of serines 362 and 374 exerts context-dependent roles in modulating Fos activity in vivo. Inhibition of Fos C-terminal phosphorylation on serine 362 by targeting RSK2 might be of therapeutic relevance for skin tumours.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bakiri L, Takada Y, Radolf M, Eferl R, Yaniv M, Wagner EF et al. (2007). Role of heterodimerization of c-Fos and Fra1 proteins in osteoclast differentiation. Bone 40: 867–875.
Barber JR, Verma IM . (1987). Modification of fos proteins: phosphorylation of c-fos, but not v-fos, is stimulated by 12-tetradecanoyl-phorbol-13-acetate and serum. Mol Cell Biol 7: 2201–2211.
Basbous J, Chalbos D, Hipskind R, Jariel-Encontre I, Piechaczyk M . (2007). Ubiquitin-independent proteasomal degradation of Fra-1 is antagonized by Erk1/2 pathway-mediated phosphorylation of a unique C-terminal destabilizer. Mol Cell Biol 27: 3936–3950.
Casalino L, Bakiri L, Talotta F, Weitzman JB, Fusco A, Yaniv M et al. (2007). Fra-1 promotes growth and survival in RAS-transformed thyroid cells by controlling cyclin A transcription. EMBO J 26: 1878–1890.
Chen RH, Abate C, Blenis J . (1993). Phosphorylation of the c-Fos transrepression domain by mitogen-activated protein kinase and 90-kDa ribosomal S6 kinase. Proc Natl Acad Sci USA 90: 10952–10956.
Chen RH, Juo PC, Curran T, Blenis J . (1996). Phosphorylation of c-Fos at the C-terminus enhances its transforming activity. Oncogene 12: 1493–1502.
Clausen BE, Burkhardt C, Reith W, Renkawitz R, Forster I . (1999). Conditional gene targeting in macrophages and granulocytes using LysMcre mice. Transgenic Res 8: 265–277.
Curran T, Miller AD, Zokas L, Verma IM . (1984). Viral and cellular Fos proteins: a comparative analysis. Cell 36: 259–268.
David JP, Mehic D, Bakiri L, Schilling AF, Mandic V, Priemel M et al. (2005). Essential role of RSK2 in c-Fos-dependent osteosarcoma development. J Clin Invest 115: 664–672.
Deng T, Karin M . (1994). c-Fos transcriptional activity stimulated by H-ras-activated protein kinase distinct from JNK and ERK. Nature 371: 171–175.
Doehn U, Hauge C, Frank SR, Jensen CJ, Duda K, Nielsen JV et al. (2009). RSK is a principal effector of the RAS-ERK pathway for eliciting a coordinate promotile/invasive gene program and phenotype in epithelial cells. Mol Cell 35: 511–522.
Durchdewald M, Guinea-Viniegra J, Haag D, Riehl A, Lichter P, Hahn M et al. (2008). Podoplanin is a novel Fos target gene in skin carcinogenesis. Cancer Res 68: 6877–6883.
Dymecki SM . (1996). Flp recombinase promotes site-specific DNA recombination in embryonic stem cells and transgenic mice. Proc Natl Acad Sci USA 93: 6191–6196.
Eferl R, Wagner EF . (2003). AP-1: a double-edged sword in tumorigenesis. Nat Rev Cancer 3: 859–868.
Ferrara P, Andermarcher E, Bossis G, Acquaviva C, Brockly F, Jariel-Encontre I et al. (2003). The structural determinants responsible for c-Fos protein proteasomal degradation differ according to the conditions of expression. Oncogene 22: 1461–1474.
Fleischmann A, Hafezi F, Elliott C, Reme CE, Ruther U, Wagner EF . (2000). Fra-1 replaces c-Fos-dependent functions in mice. Genes Dev 14: 2695–2700.
Fleischmann A, Hvalby O, Jensen V, Strekalova T, Zacher C, Layer LE et al. (2003a). Impaired long-term memory and NR2A-type NMDA receptor-dependent synaptic plasticity in mice lacking c-Fos in the CNS. J Neurosci 23: 9116–9122.
Fleischmann A, Jochum W, Eferl R, Witowsky J, Wagner EF . (2003b). Rhabdomyosarcoma development in mice lacking Trp53 and Fos: tumor suppression by the Fos protooncogene. Cancer Cell 4: 477–482.
Fu L, Patel MS, Bradley A, Wagner EF, Karsenty G . (2005). The molecular clock mediates leptin-regulated bone formation. Cell 122: 803–815.
Gius D, Cao XM, Rauscher III FJ, Cohen DR, Curran T, Sukhatme VP . (1990). Transcriptional activation and repression by Fos are independent functions: the C terminus represses immediate-early gene expression via CArG elements. Mol Cell Biol 10: 4243–4255.
Grigoriadis AE, Wang ZQ, Cecchini MG, Hofstetter W, Felix R, Fleisch HA et al. (1994). c-Fos: a key regulator of osteoclast-macrophage lineage determination and bone remodeling. Science 266: 443–448.
Hess J, Angel P, Schorpp-Kistner M . (2004). AP-1 subunits: quarrel and harmony among siblings. J Cell Sci 117: 5965–5973.
Koga K, Takaesu G, Yoshida R, Nakaya M, Kobayashi T, Kinjyo I et al. (2009). Cyclic adenosine monophosphate suppresses the transcription of proinflammatory cytokines via the phosphorylated c-Fos protein. Immunity 30: 372–383.
Monje P, Marinissen MJ, Gutkind JS . (2003). Phosphorylation of the carboxyl-terminal transactivation domain of c-Fos by extracellular signal-regulated kinase mediates the transcriptional activation of AP-1 and cellular transformation induced by platelet-derived growth factor. Mol Cell Biol 23: 7030–7043.
Murphy LO, Blenis J . (2006). MAPK signal specificity: the right place at the right time. Trends Biochem Sci 31: 268–275.
Murphy LO, Smith S, Chen RH, Fingar DC, Blenis J . (2002). Molecular interpretation of ERK signal duration by immediate early gene products. Nat Cell Biol 4: 556–564.
Nakakuki T, Birtwistle MR, Saeki Y, Yumoto N, Ide K, Nagashima T et al. (2010). Ligand-specific c-Fos expression emerges from the spatiotemporal control of ErbB network dynamics. Cell 141: 884–896.
Ofir R, Dwarki VJ, Rashid D, Verma IM . (1990). Phosphorylation of the C terminus of Fos protein is required for transcriptional transrepression of the c-fos promoter. Nature 348: 80–82.
Okazaki K, Sagata N . (1995). The Mos/MAP kinase pathway stabilizes c-Fos by phosphorylation and augments its transforming activity in NIH 3T3 cells. EMBO J 14: 5048–5059.
Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ et al. (1987). Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res 2: 595–610.
Pellegrino MJ, Stork PJ . (2006). Sustained activation of extracellular signal-regulated kinase by nerve growth factor regulates c-fos protein stabilization and transactivation in PC12 cells. J Neurochem 99: 1480–1493.
Ray N, Kuwahara M, Takada Y, Maruyama K, Kawaguchi T, Tsubone H et al. (2006). c-Fos suppresses systemic inflammatory response to endotoxin. Int Immunol 18: 671–677.
Saez E, Rutberg SE, Mueller E, Oppenheim H, Smoluk J, Yuspa SH et al. (1995). c-fos is required for malignant progression of skin tumors. Cell 82: 721–732.
Sasaki T, Kojima H, Kishimoto R, Ikeda A, Kunimoto H, Nakajima K . (2006). Spatiotemporal regulation of c-Fos by ERK5 and the E3 ubiquitin ligase UBR1, and its biological role. Mol Cell 24: 63–75.
Sassone-Corsi P, Sisson JC, Verma IM . (1988). Transcriptional autoregulation of the proto-oncogene fos. Nature 334: 314–319.
Shin S, Dimitri CA, Yoon SO, Dowdle W, Blenis J . (2010). ERK2 but not ERK1 induces epithelial-to-mesenchymal transformation via DEF motif-dependent signaling events. Mol Cell 38: 114–127.
Sibilia M, Fleischmann A, Behrens A, Stingl L, Carroll J, Watt FM et al. (2000). The EGF receptor provides an essential survival signal for SOS-dependent skin tumor development. Cell 102: 211–220.
Takada Y, Ray N, Ikeda E, Kawaguchi T, Kuwahara M, Wagner EF et al. (2010). Fos proteins suppress dextran sulfate sodium-induced colitis through inhibition of NF-kappaB. J Immunol 184: 1014–1021.
Tratner I, Ofir R, Verma IM . (1992). Alteration of a cyclic AMP-dependent protein kinase phosphorylation site in the c-Fos protein augments its transforming potential. Mol Cell Biol 12: 998–1006.
Wang ZQ, Liang J, Schellander K, Wagner EF, Grigoriadis AE . (1995). c-fos-induced osteosarcoma formation in transgenic mice: cooperativity with c-jun and the role of endogenous c-fos. Cancer Res 55: 6244–6251.
Wang ZQ, Ovitt C, Grigoriadis AE, Mohle-Steinlein U, Ruther U, Wagner EF . (1992). Bone and haematopoietic defects in mice lacking c-fos. Nature 360: 741–745.
Yang X, Matsuda K, Bialek P, Jacquot S, Masuoka HC, Schinke T et al. (2004). ATF4 is a substrate of RSK2 and an essential regulator of osteoblast biology; implication for Coffin-Lowry Syndrome. Cell 117: 387–398.
Zenz R, Scheuch H, Martin P, Frank C, Eferl R, Kenner L et al. (2003). c-Jun regulates eyelid closure and skin tumor development through EGFR signaling. Dev Cell 4: 879–889.
Acknowledgements
We are very grateful to Drs K Matsuo, Y Takada and R Khokha for critical comments and helpful suggestions, Drs F Mulero and RP Marshall for help with micro-computed tomography, U Moehle-Steinlein, E Andres, HC Theussl and V Komnenovic for technical assistance. The project was initiated at the IMP, which is funded by Boehringer Ingelheim (BI). EFW is funded by the BBVA-Foundation and by an ERC advanced Grant. Part of this work was funded by an EMBO postdoctoral fellowship to LB and the NoE Cells into Organs (LSHM-CT-2003-504468) program of the European Community.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Bakiri, L., Reschke, M., Gefroh, H. et al. Functions of Fos phosphorylation in bone homeostasis, cytokine response and tumourigenesis. Oncogene 30, 1506–1517 (2011). https://doi.org/10.1038/onc.2010.542
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.542
Keywords
This article is cited by
-
KDM2B/FBXL10 targets c-Fos for ubiquitylation and degradation in response to mitogenic stimulation
Oncogene (2016)
-
Fra-1/AP-1 induces EMT in mammary epithelial cells by modulating Zeb1/2 and TGFβ expression
Cell Death & Differentiation (2015)