Abstract
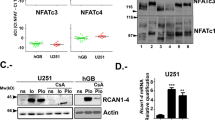
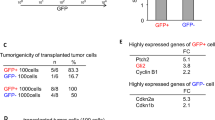
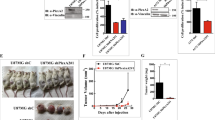
Earlier we used a glioma model to identify loci in the mouse genome, which were repeatedly targeted by platelet-derived growth factor (PDGF)-containing Moloney murine leukemia viruses. The gene Prkg2, encoding cyclic guanosine monophosphate (cGMP)-dependent protein kinase II, cGKII, was tagged by retroviral insertions in two brain tumors. The insertions were both situated upstream of the kinase domain and suggested creating a truncated form of the cGKII protein. We transfected different human glioma cell lines with Prkg2 and found an overall reduction in colony formation and cell proliferation compared with controls transfected with truncated Prkg2 (lacking the kinase domain) or empty vector. All glioma cells transfected with the cGKII phosphorylate vasodilator-stimulated phosphoprotein, VASP, after cGMP analog treatment. Glioma cell lines positive for the Sox9 transcription factor showed reduced Sox9 expression when Prkg2 was stably transfected. When cGKII was activated by cGMP analog treatment, Sox9 was phosphorylated, Sox9 protein expression was suppressed and the glioma cell lines displayed loss of cell adhesion, inhibition of Akt phosphorylation and G1 arrest. Sox9 repression by siRNA was similarly shown to reduce glioma cell proliferation. Expression analysis of stem and glial lineage cell markers also suggests that cGKII induces differentiation of glioma cell lines. These findings describe an anti-proliferative role of cGKII in human glioma biology and would further explain the retroviral tagging of the cGKII gene during brain tumor formation in PDGF-induced tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Butt E, Abel K, Krieger M, Palm D, Hoppe V, Hoppe J et al. (1994). cAMP- and cGMP-dependent protein kinase phosphorylation sites of the focal adhesion vasodilator-stimulated phosphoprotein (VASP) in vitro and in intact human platelets. J Biol Chem 269: 14509–14517.
Cen B, Deguchi A, Weinstein IB . (2008). Activation of protein kinase G increases the expression of p21CIP1, p27KIP1, and histidine triad protein 1 through Sp1. Cancer Res 68: 5355–5362.
Chen L, Daum G, Chitaley K, Coats SA, Bowen-Pope DF, Eigenthaler M et al. (2004). Vasodilator-stimulated phosphoprotein regulates proliferation and growth inhibition by nitric oxide in vascular smooth muscle cells. Arterioscler Thromb Vasc Biol 24: 1403–1408.
Cheung M, Briscoe J . (2003). Neural crest development is regulated by the transcription factor Sox9. Development 130: 5681–5693.
Chikuda H, Kugimiya F, Hoshi K, Ikeda T, Ogasawara T, Shimoaka T et al. (2004). Cyclic GMP-dependent protein kinase II is a molecular switch from proliferation to hypertrophic differentiation of chondrocytes. Genes Dev 18: 2418–2429.
Cook AL, Haynes JM . (2004). Protein kinase G II-mediated proliferative effects in human cultured prostatic stromal cells. Cell Signal 16: 253–261.
Deguchi A, Thompson WJ, Weinstein IB . (2004). Activation of protein kinase G is sufficient to induce apoptosis and inhibit cell migration in colon cancer cells. Cancer Res 64: 3966–3973.
Dromard C, Bartolami S, Deleyrolle L, Takebayashi H, Ripoll C, Simonneau L et al. (2007). NG2 and Olig2 expression provides evidence for phenotypic deregulation of cultured central nervous system and peripheral nervous system neural precursor cells. Stem Cells 25: 340–353.
Finzsch M, Stolt CC, Lommes P, Wegner M . (2008). Sox9 and Sox10 influence survival and migration of oligodendrocyte precursors in the spinal cord by regulating PDGF receptor alpha expression. Development 135: 637–646.
Gambaryan S, Palmetshofer A, Glazova M, Smolenski A, Kristjansson GI, Zimmer M et al. (2002). Inhibition of cGMP-dependent protein kinase II by its own splice isoform. Biochem Biophys Res Commun 293: 1438–1444.
Gudi T, Hong GK, Vaandrager AB, Lohmann SM, Pilz RB . (1999). Nitric oxide and cGMP regulate gene expression in neuronal and glial cells by activating type II cGMP-dependent protein kinase. FASEB J 13: 2143–2152.
Guha A, Dashner K, Black PM, Wagner JA, Stiles CD . (1995). Expression of PDGF and PDGF receptors in human astrocytoma operation specimens supports the existence of an autocrine loop. Int J Cancer 60: 168–173.
Hagerstrand D, Hesselager G, Achterberg S, Wickenberg Bolin U, Kowanetz M, Kastemar M et al. (2006). Characterization of an imatinib-sensitive subset of high-grade human glioma cultures. Oncogene 25: 4913–4922.
Hofmann F, Feil R, Kleppisch T, Schlossmann J . (2006). Function of cGMP-dependent protein kinases as revealed by gene deletion. Physiol Rev 86: 1–23.
Holland EC, Celestino J, Dai C, Schaefer L, Sawaya RE, Fuller GN . (2000). Combined activation of Ras and Akt in neural progenitors induces glioblastoma formation in mice. Nat Genet 25: 55–57.
Hou Y, Gupta N, Schoenlein P, Wong E, Martindale R, Ganapathy V et al. (2006). An anti-tumor role for cGMP-dependent protein kinase. Cancer Lett 240: 60–68.
Humphrey PA, Wong AJ, Vogelstein B, Friedman HS, Werner MH, Bigner DD et al. (1988). Amplification and expression of the epidermal growth factor receptor gene in human glioma xenografts. Cancer Res 48: 2231–2238.
Johansson FK, Brodd J, Eklof C, Ferletta M, Hesselager G, Tiger CF et al. (2004). Identification of candidate cancer-causing genes in mouse brain tumors by retroviral tagging. Proc Natl Acad Sci USA 101: 11334–11337.
Johansson FK, Goransson H, Westermark B . (2005). Expression analysis of genes involved in brain tumor progression driven by retroviral insertional mutagenesis in mice. Oncogene 24: 3896–3905.
Johansson Swartling F . (2008). Identifying candidate genes involved in brain tumor formation. Ups J Med Sci 113: 1–38.
Kordes U, Hagel C . (2006). Expression of SOX9 and SOX10 in central neuroepithelial tumor. J Neurooncol 80: 151–155.
Lahortiga I, Akin C, Cools J, Wilson TM, Mentens N, Arthur DC et al. (2008). Activity of imatinib in systemic mastocytosis with chronic basophilic leukemia and a PRKG2–PDGFRB fusion. Haematologica 93: 49–56.
Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI et al. (1997). PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275: 1943–1947.
Libermann TA, Nusbaum HR, Razon N, Kris R, Lax I, Soreq H et al. (1985). Amplification, enhanced expression and possible rearrangement of EGF receptor gene in primary human brain tumours of glial origin. Nature 313: 144–147.
Nister M, Libermann TA, Betsholtz C, Pettersson M, Claesson-Welsh L, Heldin CH et al. (1988). Expression of messenger RNAs for platelet-derived growth factor and transforming growth factor-alpha and their receptors in human malignant glioma cell lines. Cancer Res 48: 3910–3918.
Parsons DW, Jones S, Zhang X, Lin JC, Leary RJ, Angenendt P et al. (2008). An integrated genomic analysis of human glioblastoma multiforme. Science 321: 1807–1812.
Phillips HS, Kharbanda S, Chen R, Forrest WF, Soriano RH, Wu TD et al. (2006). Molecular subclasses of high-grade glioma predict prognosis, delineate a pattern of disease progression, and resemble stages in neurogenesis. Cancer Cell 9: 157–173.
Smolenski A, Burkhardt AM, Eigenthaler M, Butt E, Gambaryan S, Lohmann SM et al. (1998). Functional analysis of cGMP-dependent protein kinases I and II as mediators of NO/cGMP effects. Naunyn Schmiedebergs Arch Pharmacol 358: 134–139.
Smolenski A, Poller W, Walter U, Lohmann SM . (2000). Regulation of human endothelial cell focal adhesion sites and migration by cGMP-dependent protein kinase I. J Biol Chem 275: 25723–25732.
Sottile V, Li M, Scotting PJ . (2006). Stem cell marker expression in the Bergmann glia population of the adult mouse brain. Brain Res 1099: 8–17.
Steck PA, Pershouse MA, Jasser SA, Yung WK, Lin H, Ligon AH et al. (1997). Identification of a candidate tumour suppressor gene, MMAC1, at chromosome 10q23.3 that is mutated in multiple advanced cancers. Nat Genet 15: 356–362.
Stolt CC, Lommes P, Sock E, Chaboissier MC, Schedl A, Wegner M . (2003). The Sox9 transcription factor determines glial fate choice in the developing spinal cord. Genes Dev 17: 1677–1689.
Tanaka J, Markerink-van Ittersum M, Steinbusch HW, De Vente J . (1997). Nitric oxide-mediated cGMP synthesis in oligodendrocytes in the developing rat brain. Glia 19: 286–297.
Taylor MK, Ahmed R, Begley M, Uhler MD . (2002). Autoinhibition and isoform-specific dominant negative inhibition of the type II cGMP-dependent protein kinase. J Biol Chem 277: 37242–37253.
Uhrbom L, Hesselager G, Nister M, Westermark B . (1998). Induction of brain tumors in mice using a recombinant platelet-derived growth factor B-chain retrovirus. Cancer Res 58: 5275–5279.
Villalobo A . (2006). Nitric oxide and cell proliferation. FEBS J 273: 2329–2344.
Vindelov LL, Christensen IJ, Nissen NI . (1983). A detergent-trypsin method for the preparation of nuclei for flow cytometric DNA analysis. Cytometry 3: 323–327.
Walz C, Metzgeroth G, Haferlach C, Schmitt-Graeff A, Fabarius A, Hagen V et al. (2007). Characterization of three new imatinib-responsive fusion genes in chronic myeloproliferative disorders generated by disruption of the platelet-derived growth factor receptor beta gene. Haematologica 92: 163–169.
Wang H, Leav I, Ibaragi S, Wegner M, Hu GF, Lu ML et al. (2008). SOX9 is expressed in human fetal prostate epithelium and enhances prostate cancer invasion. Cancer Res 68: 1625–1630.
Wang H, McKnight NC, Zhang T, Lu ML, Balk SP, Yuan X . (2007). SOX9 is expressed in normal prostate basal cells and regulates androgen receptor expression in prostate cancer cells. Cancer Res 67: 528–536.
Wang SI, Puc J, Li J, Bruce JN, Cairns P, Sidransky D et al. (1997). Somatic mutations of PTEN in glioblastoma multiforme. Cancer Res 57: 4183–4186.
Yin D, Wang X, Konda BM, Ong JM, Hu J, Sacapano MR et al. (2008). Increase in brain tumor permeability in glioma-bearing rats with nitric oxide donors. Clin Cancer Res 14: 4002–4009.
Yoshioka A, Yamaya Y, Saiki S, Kanemoto M, Hirose G, Pleasure D . (2000). Cyclic GMP/cyclic GMP-dependent protein kinase system prevents excitotoxicity in an immortalized oligodendroglial cell line. J Neurochem 74: 633–640.
Acknowledgements
This work was supported by grants from the Swedish Cancer Foundation, the Children's Cancer Foundation, the Swedish Research Council and the Swedish Foundation for Strategic Research. We acknowledge Lisa Christiansson for skillful technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Swartling, F., Ferletta, M., Kastemar, M. et al. Cyclic GMP-dependent protein kinase II inhibits cell proliferation, Sox9 expression and Akt phosphorylation in human glioma cell lines. Oncogene 28, 3121–3131 (2009). https://doi.org/10.1038/onc.2009.168
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.168
Keywords
This article is cited by
-
ABCC4 suppresses glioblastoma progression and recurrence by restraining cGMP-PKG signalling
British Journal of Cancer (2024)
-
Identification of dysregulated pathways underlying HTLV-1-associated myelopathy/tropical spastic paraparesis through co-expression network analysis
Journal of NeuroVirology (2021)
-
SOX9 drives the epithelial–mesenchymal transition in non-small-cell lung cancer through the Wnt/β-catenin pathway
Journal of Translational Medicine (2019)
-
Guanylin and uroguanylin are produced by mouse intestinal epithelial cells of columnar and secretory lineage
Histochemistry and Cell Biology (2016)
-
Cyclic GMP-dependent protein kinase II is necessary for macrophage M1 polarization and phagocytosis via toll-like receptor 2
Journal of Molecular Medicine (2015)