Abstract
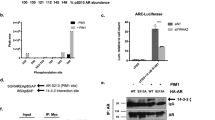
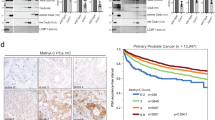
Phosphoinositide 3-OH kinases (PI3Ks) are a group of major intracellular signaling molecules. In our previous study, we found that inhibition of PI3K activity suppressed the androgen receptor (AR)-mediated gene expression in prostate cancer cells. The AR has been considered as a critical determinant for the development and progression of human prostate cancers. In this study, we sought to identify the PI3K isoforms involved in AR transactivation. Using a gene-specific small interference RNA (siRNA) approach, we determined that the regulatory isoform p85α and the catalytic isoform p110β, but not p110α, were required for androgen-stimulated AR transactivation and cell proliferation in prostate cancer cells. Consistently, overexpression of wild-type p110β but not p110α gene led to androgen-independent AR transactivation. Silencing p110β gene in prostate cancer cells abolished tumor growth in nude mice. Of the dual (lipid and protein) kinase activities, p110β’s lipid kinase activity was required for AR transactivation. Further analysis by a chromatin immunoprecipitation assay showed that p110β is indispensable for androgen-induced AR–DNA interaction. Finally, gene expression analysis of clinical specimens showed that both p85α and p110β were highly expressed in malignant prostate tissues compared to the nonmalignant compartments, and their expression levels correlated significantly with disease progression. Taken together, our data demonstrated that p85α and p110β are essential for androgen-stimulated AR transactivation, and their aberrant expression or activation might play an important role in prostate cancer progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- AR:
-
androgen receptor
- ARE:
-
androgen response element
- BrdU:
-
Bromodeoxyuridine
- ChIP:
-
chromatin IP
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- PAGE:
-
polyacrylamide
- PI3K:
-
phosphoinositide 3-OH kinase
- PKO:
-
protein kinase only
- PSA:
-
prostate-specific antigen
- PTEN:
-
phosphatase and tensin homologue deleted on chromosome 10
- RT:
-
reverse-transcription
- SEAP:
-
secreted alkaline phosphatase
- s.e.m.:
-
standard error of mean
- siRNA:
-
small interference RNA
References
Bader AG, Kang S, Zhao L, Vogt PK . (2005). Oncogenic PI3K deregulates transcription and translation. Nat Rev Cancer 5: 921–929.
Chini B, Parenti M . (2004). G-protein coupled receptors in lipid rafts and caveolae: how, when and why do they go there? J Mol Endocrinol 32: 325–338.
Cinar B, Mukhopadhyay NK, Meng G, Freeman MR . (2007). Phosphoinositide 3-kinase-independent non-genomic signals transit from the androgen receptor to Akt1 in membrane raft microdomains. J Biol Chem 282: 29584–29593.
Czauderna F, Santel A, Hinz M, Fechtner M, Durieux B, Fisch G et al. (2003). Inducible shRNA expression for application in a prostate cancer mouse model. Nucleic Acids Res 31: e127.
Engelman JA, Luo J, Cantley LC . (2006). The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat Rev Genet 7: 606–619.
Hayakawa M, Kaizawa H, Moritomo H, Koizumi T, Ohishi T, Okada M et al. (2006). Synthesis and biological evaluation of 4-morpholino-2-phenylquinazolines and related derivatives as novel PI3 kinase p110alpha inhibitors. Bioorg Med Chem 14: 6847–6858.
Heinlein CA, Chang C . (2004). Androgen receptor in prostate cancer. Endocr Rev 25: 276–308.
Holzbeierlein J, Lal P, LaTulippe E, Smith A, Satagopan J, Zhang L et al. (2004). Gene expression analysis of human prostate carcinoma during hormonal therapy identifies androgen-responsive genes and mechanisms of therapy resistance. Am J Pathol 164: 217–227.
Jackson SP, Schoenwaelder SM, Goncalves I, Nesbitt WS, Yap CL, Wright CE et al. (2005). PI 3-kinase p110beta: a new target for antithrombotic therapy. Nat Med 11: 507–514.
Kasbohm EA, Guo R, Yowell CW, Bagchi G, Kelly P, Arora P et al. (2005). Androgen receptor activation by G(s) signaling in prostate cancer cells. J Biol Chem 280: 11583–11589.
Latil A, Bieche I, Vidaud D, Lidereau R, Berthon P, Cussenot O et al. (2001). Evaluation of androgen, estrogen (ER alpha and ER beta), and progesterone receptor expression in human prostate cancer by real-time quantitative reverse transcription-polymerase chain reaction assays. Cancer Res 61: 1919–1926.
Lee SO, Lou W, Hou M, Onate SA, Gao AC . (2003). Interleukin-4 enhances prostate-specific antigen expression by activation of the androgen receptor and Akt pathway. Oncogene 22: 7981–7988.
Li B, Liao XB, Fujito A, Thrasher JB, Shen FY, Xu PY . (2007a). Dual androgen-response elements mediate androgen regulation of MMP-2 expression in prostate cancer cells. Asian J Androl 9: 41–50.
Li B, Sun A, Youn H, Hong Y, Terranova PF, Thrasher JB et al. (2007b). Conditional Akt activation promotes androgen-independent progression of prostate cancer. Carcinogenesis 28: 572–583.
Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI et al. (1997). PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275: 1943–1947.
Li P, Nicosia SV, Bai W . (2001). Antagonism between PTEN/MMAC1/TEP-1 and androgen receptor in growth and apoptosis of prostatic cancer cells. J Biol Chem 276: 20444–20450.
Liao X, Tang S, Thrasher JB, Griebling TL, Li B . (2005). Small-interfering RNA-induced androgen receptor silencing leads to apoptotic cell death in prostate cancer. Mol Cancer Ther 4: 505–515.
Liao X, Thrasher JB, Holzbeierlein J, Stanley S, Li B . (2004). Glycogen synthase kinase-3beta activity is required for androgen-stimulated gene expression in prostate cancer. Endocrinology 145: 2941–2949.
Lu ML, Schneider MC, Zheng Y, Zhang X, Richie JP . (2001). Caveolin-1 interacts with androgen receptor. A positive modulator of androgen receptor mediated transactivation. J Biol Chem 276: 13442–13451.
Majumder PK, Sellers WR . (2005). Akt-regulated pathways in prostate cancer. Oncogene 24: 7465–7474.
Mellinghoff IK, Vivanco I, Kwon A, Tran C, Wongvipat J, Sawyers CL . (2004). HER2/neu kinase-dependent modulation of androgen receptor function through effects on DNA binding and stability. Cancer Cell 6: 517–527.
Murga C, Laguinge L, Wetzker R, Cuadrado A, Gutkind JS . (1998). Activation of Akt/protein kinase B by G protein-coupled receptors. A role for alpha and beta gamma subunits of heterotrimeric G proteins acting through phosphatidylinositol-3-OH kinasegamma. J Biol Chem 273: 19080–19085.
Murillo H, Huang H, Schmidt LJ, Smith DI, Tindall DJ . (2001). Role of PI3K signaling in survival and progression of LNCaP prostate cancer cells to the androgen refractory state. Endocrinology 142: 4795–4805.
Scher HI, Sawyers CL . (2005). Biology of progressive, castration-resistant prostate cancer: directed therapies targeting the androgen-receptor signaling axis. J Clin Oncol 23: 8253–8261.
Shang Y, Myers M, Brown M . (2002). Formation of the androgen receptor transcription complex. Mol Cell 9: 601–610.
Shanmugam I, Cheng G, Terranova PF, Thrasher JB, Thomas CP, Li B . (2007). Serum/glucocorticoid-induced protein kinase-1 facilitates androgen receptor-dependent cell survival. Cell Death Differ 14: 2085–2094.
Sharma M, Chuang WW, Sun Z . (2002). Phosphatidylinositol 3-kinase/Akt stimulates androgen pathway through GSK3beta inhibition and nuclear beta-catenin accumulation. J Biol Chem 277: 30935–30941.
Steck PA, Pershouse MA, Jasser SA, Yung WK, Lin H, Ligon AH et al. (1997). Identification of a candidate tumour suppressor gene, MMAC1, at chromosome 10q23.3 that is mutated in multiple advanced cancers. Nat Genet 15: 356–362.
Sun A, Shanmugam I, Song J, Terranova PF, Thrasher JB, Li B . (2007a). Lithium suppresses cell proliferation by interrupting E2F-DNA interaction and subsequently reducing S-phase gene expression in prostate cancer. Prostate 67: 976–988.
Sun A, Tawfik O, Gayed B, Thrasher JB, Hoestje S, Li C et al. (2007b). Aberrant expression of SWI/SNF catalytic subunits BRG1/BRM is associated with tumor development and increased invasiveness in prostate cancers. Prostate 67: 203–213.
Sun M, Yang L, Feldman RI, Sun XM, Bhalla KN, Jove R et al. (2003). Activation of phosphatidylinositol 3-kinase/Akt pathway by androgen through interaction of p85alpha, androgen receptor, and Src. J Biol Chem 278: 42992–43000.
Yart A, Roche S, Wetzker R, Laffargue M, Tonks N, Mayeux P et al. (2002). A function for phosphoinositide 3-kinase beta lipid products in coupling beta gamma to Ras activation in response to lysophosphatidic acid. J Biol Chem 277: 21167–21178.
Zhao JJ, Liu Z, Wang L, Shin E, Loda MF, Roberts TM . (2005). The oncogenic properties of mutant p110alpha and p110beta phosphatidylinositol 3-kinases in human mammary epithelial cells. Proc Natl Acad Sci USA 102: 18443–18448.
Acknowledgements
We thank Dr Alen Gao at Roswell Park Cancer Institute (Buffalo, NY) for the construct of ARE-LUC reporter, Dr David Spencer (Baylor College of Medicine) for the construct of CMV-SEAP reporter and Dr Jorg Kaufmann for the pU6+2p110beta.shRNA construct. We also thank Mrs Marsha Danley (Department of Pathology, KUMC Hospital) and Dr Ilanchezhian Shanmugam for excellent technical assistance in IHC staining and ChIP assay, respectively. This study was supported by KU William L Valk Endowment and Kansas Masonic Foundation through KU Cancer Center pilot grant. This work was also partially supported by a KU-NIH COBRE grant (1P20RR15563) from the National Center for Research Resources (NIH-NCRR), Department of Defense New Investigator Award (DAMD17-03-1-0121) and Idea Development Award (W81XWH-04-1-0214) to Dr Benyi Li.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, Q., Youn, H., Tang, J. et al. Phosphoinositide 3-OH kinase p85α and p110β are essential for androgen receptor transactivation and tumor progression in prostate cancers. Oncogene 27, 4569–4579 (2008). https://doi.org/10.1038/onc.2008.91
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.91
Keywords
This article is cited by
-
PI3K isoforms in cell signalling and vesicle trafficking
Nature Reviews Molecular Cell Biology (2019)
-
Overexpression of PIK3R1 promotes hepatocellular carcinoma progression
Biological Research (2018)
-
Molecules in medicine mini-review: isoforms of PI3K in biology and disease
Journal of Molecular Medicine (2016)
-
Targeting the androgen receptor pathway in castration-resistant prostate cancer: progresses and prospects
Oncogene (2015)
-
RNAi-based functional selection identifies novel cell migration determinants dependent on PI3K and AKT pathways
Nature Communications (2014)