Abstract
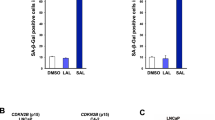
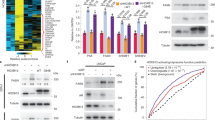
Androgen-withdrawal-induced apoptosis (AWIA) is deregulated in androgen refractory prostate cancer. Androgens have been shown to positively regulate expression of the antiapoptotic FADD-like interleukin-1β-converting enzyme (FLICE)-like inhibitory protein (FLIP), and reduced FLIP expression precedes apoptosis after androgen withdrawal. Here, we show that FLIP protein expression is downregulated in castrated rats, while in LNCaP cells, androgens regulate FLIP in a manner that is dependent on phosphoinositol-3-kinase (PI3K) and Akt signaling. Specifically, treatment of LNCaP cells with LY294002, or expression of either PTEN or a non-phosphorylatable form of FOXO3a (FOXO3aTM), downregulates FLIP protein and mRNA. Conversely, treatment with androgens in the absence of PI3/Akt signaling, or following expression of FOXO3aTM, leads to increased FLIP expression. A FOXO3a binding site was identified in the FLIP promoter and shown necessary for the combined effects of androgens and FOXO3a on FLIP transcription. FOXO3a binds the androgen receptor, suggesting that the transcriptional synergy depends on an interaction between these proteins. Finally, LNCaP cells are sensitized to TRAIL-induced apoptosis by PTEN or LY294002, and rescued by androgens. FOXO3aTM also sensitizes cells to androgen-inhibited TRAIL apoptosis. Androgen rescue was diminished when either FOXO3a or FLIP was reduced by siRNA. These data support a role for FOXO3a in AWIA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
References
Ayala G, Thompson T, Yang G, Frolov A, Li R, Scardino P et al. (2004). High levels of phosphorylated form of Akt-1 in prostate cancer and non-neoplastic prostate tissues are strong predictors of biochemical recurrence. Clin Cancer Res 10: 6572–6578.
Bertram J, Peacock JW, Fazli L, Mui AL, Chung SW, Cox ME et al. (2006). Loss of PTEN is associated with progression to androgen independence. Prostate 66: 895–902.
Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS et al. (1999). Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96: 857–868.
Cully M, You H, Levine AJ, Mak TW . (2006). Beyond PTEN mutations: the PI3K pathway as an integrator of multiple inputs during tumorigenesis. Nat Rev Cancer 6: 184–192.
Daskivich TJ, Oh WK . (2006). Recent progress in hormonal therapy for advanced prostate cancer. Curr Opin Urol 16: 173–178.
De la Taille A, Chen MW, Shabsigh A, Bagiella E, Kiss A, Buttyan R . (1999). Fas antigen/CD-95 upregulation and activation during castration-induced regression of the rat ventral prostate gland. Prostate 40: 89–96.
Furutani T, Watanabe T, Tanimoto K, Hashimoto T, Koutoku H, Kudoh M et al. (2002). Stabilization of androgen receptor protein is induced by agonist, not by antagonists. Biochem Biophys Res Commun 294: 779–784.
Gao S, Lee P, Wang H, Gerald W, Adler M, Zhang L et al. (2005). The androgen receptor directly targets the c-FLIP gene to promote the androgen-independent growth of prostate cancer cells. Mol Endocrinol 19: 1792–1802.
Gao S, Wang H, Lee P, Melamed J, Li CX, Zhang F et al. (2006). Androgen receptor and prostate apoptosis response factor-4 target the c-FLIP gene to determine survival and apoptosis in the prostate gland. J Mol Endocrinol 36: 463–483.
Hermans KG, van Alewijk DC, Veltman JA, van Weerden W, van Kessel AG, Trapman J . (2004). Loss of a small region around the PTEN locus is a major chromosome 10 alteration in prostate cancer xenografts and cell lines. Genes Chromosomes Cancer 39: 171–184.
Huang H, Muddiman DC, Tindall DJ . (2004). Androgens negatively regulate forkhead transcription factor FKHR (FOXO1) through a proteolytic mechanism in prostate cancer cells. J Biol Chem 279: 13866–13877.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C et al. (2006). Cancer statistics. CA Cancer J Clin 56: 106–130.
Kimura K, Markowski M, Bowen C, Gelmann EP . (2001). Androgen blocks apoptosis of hormone-dependent prostate cancer cells. Cancer Res 61: 5611–5618.
Krueger A, Schmitz I, Baumann S, Krammer PH, Kirchhoff S . (2001). Cellular FLICE-inhibitory protein splice variants inhibit different steps of caspase-8 activation at the CD95 death-inducing signaling complex. J Biol Chem 276: 20633–20640.
Kyprianou N, Isaacs JT . (1988). Activation of programmed cell death in the rat ventral prostate after castration. Endocrinology 122: 552–562.
Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI et al. (1997). PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275: 1943–1947.
Li P, Lee H, Guo S, Unterman TG, Jenster G, Bai W . (2003). AKT-independent protection of prostate cancer cells from apoptosis mediated through complex formation between the androgen receptor and FKHR. Mol Cell Biol 23: 104–118.
Li P, Nicosia SV, Bai W . (2001). Antagonism between PTEN/MMAC1/TEP-1 and androgen receptor in growth and apoptosis of prostatic cancer cells. J Biol Chem 276: 20444–20450.
Liao Y, Grobholz R, Abel U, Trojan L, Michel MS, Angel P et al. (2003). Increase of AKT/PKB expression correlates with gleason pattern in human prostate cancer. Int J Cancer 107: 676–680.
Longley DB, Wilson TR, McEwan M, Allen WL, McDermott U, Galligan L et al. (2006). c-FLIP inhibits chemotherapy-induced colorectal cancer cell death. Oncogene 25: 838–848.
Lynch RL, Konicek BW, McNulty AM, Hanna KR, Lewis JE, Neubauer BL et al. (2005). The progression of LNCaP human prostate cancer cells to androgen independence involves decreased FOXO3a expression and reduced p27KIP1 promoter transactivation. Mol Cancer Res 3: 163–169.
Majumder PK, Sellers WR . (2005). Akt-regulated pathways in prostate cancer. Oncogene 24: 7465–7474.
Marti A, Jaggi R, Vallan C, Ritter PM, Baltzer A, Srinivasan A et al. (1999). Physiological apoptosis in hormone-dependent tissues: involvement of caspases. Cell Death Differ 6: 1190–1200.
Martikainen P, Kyprianou N, Isaacs JT . (1990). Effect of transforming growth factor-beta 1 on proliferation and death of rat prostatic cells. Endocrinology 127: 2963–2968.
Matys V, Fricke E, Geffers R, Gossling E, Haubrock M, Hehl R et al. (2003). TRANSFAC: transcriptional regulation, from patterns to profiles. Nucleic Acids Res 31: 374–378.
Nastiuk KL, Kim JW, Mann M, Krolewski JJ . (2003). Androgen regulation of FLICE-like inhibitory protein gene expression in the rat prostate. J Cell Physiol 196: 386–393.
Nesterov A, Ivashchenko Y, Kraft AS . (2002). Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) triggers apoptosis in normal prostate epithelial cells. Oncogene 21: 1135–1140.
Nesterov A, Lu X, Johnson M, Miller GJ, Ivashchenko Y, Kraft AS . (2001). Elevated AKT activity protects the prostate cancer cell line LNCaP from TRAIL-induced apoptosis. J Biol Chem 276: 10767–10774.
Panka DJ, Mano T, Suhara T, Walsh K, Mier JW . (2001). Phosphatidylinositol 3-kinase/Akt activity regulates c-FLIP expression in tumor cells. J Biol Chem 276: 6893–6896.
Roth W, Reed JC . (2004). FLIP protein and TRAIL-induced apoptosis. Vitam Horm 67: 189–206.
Shaw G, Morse S, Ararat M, Graham FL . (2002). Preferential transformation of human neuronal cells by human adenoviruses and the origin of HEK 293 cells. FASEB J 16: 869–871.
Skurk C, Maatz H, Kim HS, Yang J, Abid MR, Aird WC et al. (2004). The Akt-regulated forkhead transcription factor FOXO3a controls endothelial cell viability through modulation of the caspase-8 inhibitor FLIP. J Biol Chem 279: 1513–1525.
Srikanth S, Kraft AS . (1998). Inhibition of caspases by cytokine response modifier A blocks androgen ablation-mediated prostate cancer cell death in vivo. Cancer Res 58: 834–839.
Sugihara A, Yamada N, Tsujimura T, Iwasaki T, Yamashita K, Takagi Y et al. (2001). Castration induces apoptosis in the male accessory sex organs of Fas-deficient lpr and Fas ligand-deficient gld mutant mice. In vivo 15: 385–390.
Suzuki A, Matsuzawa A, Iguchi T . (1996). Down regulation of Bcl-2 is the first step on Fas-mediated apoptosis of male reproductive tract. Oncogene 13: 31–37.
Tang ED, Nunez G, Barr FG, Guan KL . (1999). Negative regulation of the forkhead transcription factor FKHR by Akt. J Biol Chem 274: 16741–16746.
Yuan XJ, Whang YE . (2002). PTEN sensitizes prostate cancer cells to death receptor-mediated and drug-induced apoptosis through a FADD-dependent pathway. Oncogene 21: 319–327.
Zhang X, Jin TG, Yang H, DeWolf WC, Khosravi-Far R, Olumi AF . (2004). Persistent c-FLIP(L) expression is necessary and sufficient to maintain resistance to tumor necrosis factor-related apoptosis-inducing ligand-mediated apoptosis in prostate cancer. Cancer Res 64: 7086–7091.
Acknowledgements
This study was supported by grant PC030937 from the US Army Prostate Cancer Research Program. ANC was supported by a PHS training grant (5T32CA009054). We thank D Fruman, R Miesfeld, K Guan and M Greenberg for plasmids, and C Walsh and D Fruman for assistance with FACS analysis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Cornforth, A., Davis, J., Khanifar, E. et al. FOXO3a mediates the androgen-dependent regulation of FLIP and contributes to TRAIL-induced apoptosis of LNCaP cells. Oncogene 27, 4422–4433 (2008). https://doi.org/10.1038/onc.2008.80
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.80
Keywords
This article is cited by
-
The testosterone paradox of advanced prostate cancer: mechanistic insights and clinical implications
Nature Reviews Urology (2023)
-
Development of acquired resistance to lapatinib may sensitise HER2-positive breast cancer cells to apoptosis induction by obatoclax and TRAIL
BMC Cancer (2018)
-
A spatiotemporal hypothesis for the regulation, role, and targeting of AMPK in prostate cancer
Nature Reviews Urology (2017)
-
Nucleo-cytoplasmic transport as a therapeutic target of cancer
Journal of Hematology & Oncology (2014)
-
Estrogen receptor β upregulates FOXO3a and causes induction of apoptosis through PUMA in prostate cancer
Oncogene (2014)