Abstract
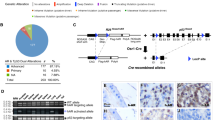
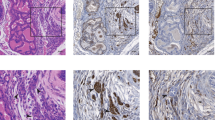
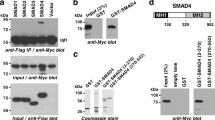
The high prevalence of TMPRSS2-ERG rearrangements (∼60%) in prostate cancer (CaP) leads to androgenic induction of the ETS-related gene (ERG) expression. However, the biological functions of ERG overexpression in CaP remain to be understood. ERG knockdown in TMPRSS2-ERG expressing CaP cells induced striking morphological changes and inhibited cell growth both in cell culture and SCID mice. Evaluation of the transcriptome and specific gene promoters in ERG siRNA-treated cells and investigation of gene expression signatures of human prostate tumors revealed ERG-mediated activation of C-MYC oncogene and the repression of prostate epithelial differentiation genes (PSA and SLC45A3/Prostein). Taken together, these data combining cell culture and animal models and human prostate tumors reveal that ERG overexpression in prostate tumor cells may contribute to the neoplastic process by activating C-MYC and by abrogating prostate epithelial differentiation as indicated by prostate epithelial specific markers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Baldus CD, Burmeister T, Martus P, Schwartz S, Gokbuget N, Bloomfield CD et al. (2006). High expression of the ETS transcription factor ERG predicts adverse outcome in acute T-lymphoblastic leukemia in adults. J Clin Oncol 24: 4714–4720.
De Marzo AM, Platz EA, Sutcliffe S, Xu J, Gronberg H, Drake CG et al. (2007). Inflammation in prostate carcinogenesis. Nat Rev Cancer 7: 256–269.
Demichelis F, Rubin MA . (2007). TMPRSS2-ETS fusion prostate cancer: biological and clinical implications. J Clin Pathol 60: 1185–1186.
Furusato B, Gao CL, Ravindranath L, Chen Y, Cullen J, McLeod DG et al. (2008). Mapping of TMPRSS2-ERG fusions in the context of multi-focal prostate cancer. Mod Pathol 21: 67–75.
Iljin K, Wolf M, Edgren H, Gupta S, Kilpinen S, Skotheim RI et al. (2006). TMPRSS2 fusions with oncogenic ETS factors in prostate cancer involve unbalanced genomic rearrangements and are associated with HDAC1 and epigenetic reprogramming. Cancer Res 66: 10242–10246.
Klezovitch O, Risk M, Coleman I, Lucas JM, Null M, True LD et al. (2008). A causal role for ERG in neoplastic transformation of prostate epithelium. Proc Natl Acad Sci USA 105: 2105–2110.
Korenchuk S, Lehr JE, Mclean L, Lee YG, Whitney S, Vessella R et al. (2001). VCaP, a cell-based model system of human prostate cancer. In Vivo 15: 163–168.
Lin B, Ferguson C, White JT, Wang S, Vessella R, True LD et al. (1999). Prostate-localized and androgen-regulated expression of the membrane-bound serine protease TMPRSS2. Cancer Res 59: 4180–4184.
Marcucci G, Mrozek K, Bloomfield CD . (2005). Molecular heterogeneity and prognostic biomarkers in adults with acute myeloid leukemia and normal cytogenetics. Curr Opin Hematol 12: 68–75.
Massie CE, Adryan B, Barbosa-Morais NL, Lynch AG, Tran MG, Neal DE et al. (2007). New androgen receptor genomic targets show an interaction with the ETS1 transcription factor. EMBO Rep 8: 871–878.
Meulia T, Krumm A, Spencer C, Groudine M . (1992). Sequences in the human c-myc P2 promoter affect the elongation and premature termination of transcripts initiated from the upstream P1 promoter. Mol Cell Biol 12: 4590–4600.
Oettgen P, Finger E, Sun Z, Akbarali Y, Thamrongsak U, Boltax J et al. (2000). PDEF, a novel prostate epithelium-specific ets transcription factor, interacts with the androgen receptor and activates prostate-specific antigen gene expression. J Biol Chem 275: 1216–1225.
Owczarek CM, Portbury KJ, Hardy MP, O'Leary DA, Kudoh J, Shibuya K et al. (2004). Detailed mapping of the ERG-ETS2 interval of human chromosome 21 and comparison with the region of conserved synteny on mouse chromosome 16. Gene 324: 65–77.
Petrovics G, Liu A, Shaheduzzaman S, Furasato B, Sun C, Chen Y et al. (2005). Frequent overexpression of ETS-related gene-1 (ERG1) in prostate cancer transcriptome. Oncogene 24: 3847–3852.
Scherf M, Epple A, Werner T . (2005). The next generation of literature analysis: integration of genomic analysis into text mining. Brief Bioinform 6: 287–297.
Seth A, Watson DK . (2005). ETS transcription factors and their emerging roles in human cancer. Eur J Cancer 41: 2462–2478.
Shaheduzzaman S, Vishwanath A, Furusato B, Cullen J, Chen Y, Banez L et al. (2007). Silencing of lactotransferrin expression by methylation in prostate cancer progression. Cancer Biol Ther 6: 1–8.
Tomlins SA, Laxman B, Dhanasekaran SM, Helgeson BE, Cao X, Morris DS et al. (2007). Distinct classes of chromosomal rearrangements create oncogenic ETS gene fusions in prostate cancer. Nature 448: 595–599.
Tomlins SA, Laxman B, Varambally S, Cao X, Yu J, Helgeson BE et al. (2008). Role of the TMPRSS2-ERG gene fusion in prostate cancer. Neoplasia 10: 177–188.
Tomlins SA, Rhodes DR, Perner S, Dhanasekaran SM, Mehra R, Sun XW et al. (2005). Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science 310: 644–648.
Turner DP, Watson DK . (2008). ETS transcription factors: oncogenes and tumor suppressor genes as therapeutic targets for prostate cancer. Expert Rev Anticancer Ther 8: 33–42.
Witte JS . (2007). Multiple prostate cancer risk variants on 8q24. Nat Genet 39: 579–580.
Acknowledgements
Authors express sincere thanks to Ms Lakshmi Ravindranath, Ms Atekelt Y Tadese, Ms Suma Ravulapalli and Mr Christopher Cook for their superb technical support of this study. The prostate tissue specimens used in this study were obtained under an IRB-approved protocol at Walter Reed Army Medical Center. This work was funded by the CPDR through an ongoing grant from the US Army Medical Research and Materiel Command, NIH Grants RO1 DK065977 to S.S and G.P. and RO1 CA106653 to S.S. and A.D. The views expressed in this manuscript are those of the authors and do not reflect the official policy of the Department of the Army, Department of Defense or the US Government.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Sun, C., Dobi, A., Mohamed, A. et al. TMPRSS2-ERG fusion, a common genomic alteration in prostate cancer activates C-MYC and abrogates prostate epithelial differentiation. Oncogene 27, 5348–5353 (2008). https://doi.org/10.1038/onc.2008.183
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.183
Keywords
This article is cited by
-
Mechanism-centric regulatory network identifies NME2 and MYC programs as markers of Enzalutamide resistance in CRPC
Nature Communications (2024)
-
The impact of fusion genes on cancer stem cells and drug resistance
Molecular and Cellular Biochemistry (2021)
-
Targeting the ERG oncogene with splice-switching oligonucleotides as a novel therapeutic strategy in prostate cancer
British Journal of Cancer (2020)
-
Centrosome loss results in an unstable genome and malignant prostate tumors
Oncogene (2020)
-
TMPRSS2-ERG activates NO-cGMP signaling in prostate cancer cells
Oncogene (2019)